COVID19 vaccination programme QA session Vaccine Preventable Disease

COVID-19 vaccination programme Q&A session Vaccine Preventable Disease Programme Public Health Wales

The COVID-19 pandemic and novel vaccines

Key messages • The ongoing global COVID-19 pandemic has so far infected over 60 million people and over 1. 4 million deaths across the world. • A vaccine to prevent COVID-19 is the most effective way to control the pandemic. • Scientists across the world have worked to develop vaccines which have been rigorously tested for safety and efficacy, and independently assessed and authorised for use. • It is crucial that the COVID-19 vaccine is safely and effectively delivered to as many of those eligible as possible. • Those with a role in delivering the COVID-19 vaccine programme need to be knowledgeable, confident and competent in order to promote confidence in the vaccination programme and deliver the vaccine safely.

COVID-19 Vaccines • On 2 December 2020, the following vaccine was given authorisation for temporary supply by the UK DHSC and the MHRA: • Pfizer-Bio. NTech COVID-19 vaccine is a messenger ribonucleic acid (m. RNA) that contains the genetic sequence of the antigens found on the surface of the SARS-Co. V-2 virus • Another COVID-19 vaccine which may shortly be authorised for supply in the UK, subject to regulatory approval, is: • Astra. Zeneca COVID-19 vaccine which is a non-replicating viral vector vaccine. It uses a weakened adenovirus as a carrier to deliver the genetic sequence for part of the SARSCo. V-2 virus into the body

P r o v i s i o n a l – S u b j e c t t o r e v i s i o n – U Pfizer-Bio. NTech COVID-19 vaccine • The Pfizer-Bio. NTech COVID-19 vaccine is a messenger ribonucleic acid (m. RNA ) vaccine. • It contains the genetic sequence (m. RNA) for the spike protein which is found on the surface of the SARS-Co. V-2 virus, wrapped in a lipid envelope (referred to as a nanoparticle) to enable it to be transported into the cells in the body. • When injected, the m. RNA is taken up by the host’s cells which translate the genetic information and produce the spike proteins. • These are then displayed on the surface of the cell. This stimulates the immune system to produce antibodies and activate T-cells which prepare the immune system to respond to any future exposure to the SARS-Co. V-2 virus by binding to and disabling any virus encountered. • As there is no whole or live virus involved, the vaccine cannot cause disease. The m. RNA naturally degrades after a few days.

Astra. Zeneca COVID-19 vaccine • Astra. Zeneca COVID-19 vaccine is a viral vector vaccine which uses a weakened adenovirus as a carrier to deliver the SARS-Co. V-2 antigen. • The adenovirus has been modified so that it cannot replicate (grow and multiply by making copies of itself) in human cells and therefore cause any disease. • The genes that encode for the spike protein on the SARS-Co. V-2 virus have been inserted into the adenovirus's genetic code to make the vaccine. • When the vaccine is injected, it enters the host's cells which then manufacture the spike protein. • This then stimulates the immune system which reacts by producing antibodies and memory cells to the SARS-Co. V-2 virus without causing disease.

Regulatory approval and licencing • In the UK, vaccine manufacturers apply to the Medicines and Healthcare products Regulatory Agency (MHRA) or the European Medicines Agency (EMA) for a product licence but they may issue a temporary authorisation ahead of the full UK product licence. • In exceptional situations, the MHRA may enact Regulation 174. This enables them to temporarily authorise the supply of an unlicensed medicinal product in response to certain identified public health risks, such as the SARS-Co. V-2 pandemic. • The MHRA assess all of the available safety, quality and efficacy data and considers whether the evidence supports the use of the medicine or vaccine under the legal basis of regulation 174 until it can be licensed. • The process involves the same level of scrutiny as the usual licensing process and this exception is only used when strictly necessary to speed up access to a potentially lifesaving intervention.

The Welsh Immunisation System (WIS)

Welsh Immunisation System • The Welsh Immunisation System (WIS) is a data system developed by NWIS from CYPr. IS • WIS is designed to manage and administer COVID-19 immunisation sessions • It will keep a clinical record of everyone in Wales vaccinated with a COVID-19 vaccine • The WIS system holds patients demographic details and can identify those who are eligible for a vaccine by their age or conditions. • It generates appointments and produces invitation letters, and can send SMS text reminders

Welsh Immunisation System • WIS enables healthcare staff to input data at the point of service delivery to record clinical data • It also provides a link to the patient’s GP record which the vaccinator can view • The WIS system has two versions: • a WEB version to be used at immunisation sessions by vaccinators and support admin • a CORE version which is centrally based and will be used for setting up immunisation sessions, processing appointments, producing letters and general data management • WIS feeds all daily dashboards, surveillance outputs and is used for safety monitoring (via MHRA) and safety studies

WIS Entry Screen • Demographics pre-loaded • Entry screen for consultation details • Welsh GP Record lookup • Immunisation summary screen • Mandatory and optional fields. • Drop down menus. Text fields.

Consent

Consent - same as for any vaccine/medicine • Paper records may be required for some locations • Template consent forms for staff and WIS back up forms • http: //nww. immunisation. wales. nhs. uk/covid-19 -and-immunisationprogrammes#Resources • Guidance document on consent processes including best interest decisions drafted by Dr Liam Taylor • Now finalised as version 2 • Will be shared with HBs and trusts

WIS paper back up

Staff paper consent form
WIS group reference

Vaccine specifics

Pfizer-Bio. NTech COVID-19 vaccine • The vaccine contains BNT 162 b 2 messenger RNA embedded in lipid nanoparticles • The vaccine does not contain preservative • No animal products are used in the manufacture of this vaccine • Product information about the Pfizer-Bio. NTech COVID-19 vaccine is available at https: //coronavirus-yellowcard. mhra. gov. uk/productinformation

Pfizer-Bio. NTech COVID-19 vaccine Presentation: • The vaccine packs contain 195 vials of vaccine (975 doses per pack as each vial contains 5 doses) • The vaccine is contained in a multidose clear glass vial. The vial has a rubber (bromobutyl) stopper, aluminium seal and a flip-off plastic cap. The stopper does not contain latex • Each vial contains 0. 45 ml of vaccine and should be diluted with 1. 8 ml of Sodium Chloride 0. 9% Solution for Injection (normal saline). • Once diluted, each reconstituted vaccine will supply 5 doses of 0. 3 ml (30 mcg) • There will be a small amount of vaccine left in the vial after drawing 5 doses: this should be discarded

Pfizer-Bio. NTech COVID-19 vaccine Vaccine dose and schedule: • A single dose is 0. 3 mls and two doses of vaccine are required *A minimum of 21 days interval between doses • * A 28 days interval is being recommended in Wales for ease of operational delivery • using a consistent interval between all two-dose COVID-19 vaccines simplifies the messaging to the public and arrangements within clinic settings where alternative vaccines may be supplied at short notice • Pfizer-Bio. NTech COVID-19 vaccine should not be given at the same time as any other vaccine. • If other vaccines are required, a 7 day interval should be left between the dose of COVID-19 m. RNA Vaccine BNT 162 b 2 and the other vaccine.

Pfizer-Bio. NTech COVID-19 vaccine Storage and transportation: • The vaccine will be delivered frozen to healthcare facilities with ultra low temperature (ULT) freezers • Vaccine packs will be shipped inside isothermic boxes (validated boxes which will maintain a constant temperature for a specified period of time) inside a cardboard box • The isothermic box will also contain dry ice which should be disposed of carefully following local protocols • Upon delivery, the vaccine packs should be removed from the isothermic box and transferred to a suitable ULT freezer to ensure ongoing storage between -75 o. C and +/-15 o. C • The vaccine should be kept upright, in its original packaging and away from prolonged light exposure • The vaccine packs must not be opened until the vaccine is going to be thawed • When required, frozen vials should be transferred to 2 o. C to 8 o. C to thaw; a 195 vial pack may take 3 hours to thaw • Alternatively, frozen vials may also be thawed for 30 minutes at temperatures up to 25 o. C for immediate use. once thawed, the vaccine cannot be re-frozen • shelf-life is 6 months at -80 o. C to -60 o. C

Pfizer-Bio. NTech COVID-19 vaccine Storage and transportation in a thawed state: • The vaccine may then be delivered to vaccine administration centres, thawed but refrigerated between +2 and +8 o. C • Refrigerated vaccine must be transferred immediately to a vaccine fridge on arrival and stored in a carefully monitored temperature range of +2 and +8 o. C • Stability data currently indicates the Pfizer-Bio. NTech COVID-19 vaccine will remain stable for 5 days (120 hours) when stored in a fridge with a temperature range of +2 and +8 o. C before reconstitution and for 6 hours at room temperature after reconstitution • The vaccine pack will have a yellow label on the front stating the time it was removed from the freezer into storage at +2 to +8 o. C and the date and time by which it must be discarded 5 days (120 hours) later if it has not been used • Vaccine should be stored in the original package to protect it from light. Exposure to room light should be minimised and exposure to direct sunlight and ultraviolet light should be avoided

Storage and use of Pfizer-Bio. NTech COVID-19 vaccine • The Pfizer-Bio. NTech COVID-19 vaccine has very specific storage, reconstitution and 'use within' requirements • All those involved in the delivery of the COVID-19 vaccination programme must be aware of the recommended storage requirements The vaccine must not be given if you are not confident that it has been stored or reconstituted as recommended by the manufacturer or as advised by a vaccine expert • If the vaccine is stored incorrectly: Ø Label and isolate affected vaccines in the fridge and do not use until further notice Ø Seek advice from the manufacturer or a source of expert advice
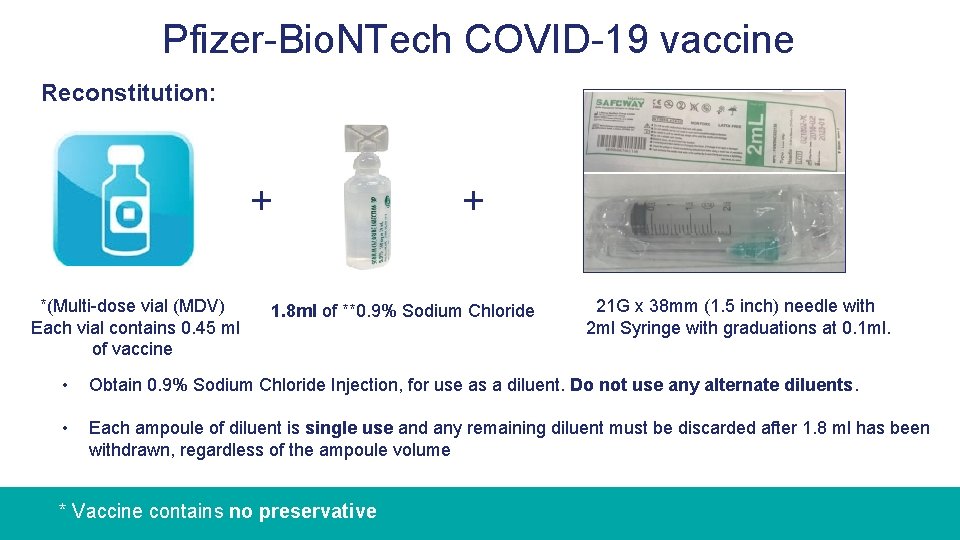
Pfizer-Bio. NTech COVID-19 vaccine Reconstitution: + *(Multi-dose vial (MDV) Each vial contains 0. 45 ml of vaccine + 1. 8 ml of **0. 9% Sodium Chloride 21 G x 38 mm (1. 5 inch) needle with 2 ml Syringe with graduations at 0. 1 ml. • Obtain 0. 9% Sodium Chloride Injection, for use as a diluent. Do not use any alternate diluents. • Each ampoule of diluent is single use and any remaining diluent must be discarded after 1. 8 ml has been withdrawn, regardless of the ampoule volume * Vaccine contains no preservative

Pfizer-Bio. NTech COVID-19 vaccine (1) Reconstituting: 1. Clean hands with alcohol-based gel or soap and water 2. Assemble one ampoule of Sodium Chloride 0. 9% Solution for Injection, a single use alcohol swab, a needle with a green hub and a 2 ml syringe 3. From cold storage, remove one vial of vaccine 4. If removing the multidose vaccine vial directly from a ULT freezer, allow the vaccine to defrost for 30 minutes at temperatures up to 25 o. C and reconstitute within 2 hours after thawing. Allow the thawed vial to come to room temperature and gently invert 10 times prior to dilution. Do not shake. 5. If removing the multidose vaccine vial from cold storage between +2 o. C and +8 o. C, check that it has not been stored there for longer than 5 days (120 hours) 6. Check the expiry date and the appearance of the vaccine. Prior to dilution, the vaccine should be an off-white solution with no particulates visible. Discard the vaccine if particulates or discolouration are present 7. Connect the needle with a green hub to the 2 ml syringe REG 174 INFORMATION FOR UK HEALTHCARE PROFESSIONALS

Pfizer-Bio. NTech COVID-19 vaccine (2) Reconstituting: 8. Clean the vial stopper with the single use alcohol swab and allow to air dry fully 9. Draw up 1. 8 ml of Sodium Chloride 0. 9% Solution for Injection, then discard the diluent ampoule and any remaining diluent in it. Do not use any other type of diluent 10. Add diluent to the vaccine vial. You may feel some pressure in the vial as you add the diluent. Equalise the vial pressure by withdrawing 1. 8 ml of air into the empty diluent syringe before removing the needle from the vial 11. Gently invert the diluted solution 10 times. Do not shake

Pfizer-Bio. NTech COVID-19 vaccine (3) 12. The diluted vaccine should be an off-white solution with no particulates visible. 13. Discard the diluted vaccine if particulates or discolouration are present 14. Dispose of green hub needle and syringe into yellow sharps bin 15. The diluted vial should be clearly labelled with the dilution date and time. Use immediately, and within 6 hours after dilution 16. Once reconstituted the MDV contains 5 doses of vaccine 17. The dosing solution concentration is 0. 1 mg/ml 18. A single dose is 0. 3 mls/ 30 micrograms 19. There will be a small amount of vaccine left in the vial after drawing 5 doses. 20. This is to ensure that 5 complete doses can be withdrawn. 21 Any vaccine remaining in the vial after this should be discarded

Pfizer-Bio. NTech COVID-19 vaccine Reconstituted vaccine can be returned to the fridge/cool box and stored between +2 o. C and +8 o. C but must be used within 6 hours following dilution

Preparing the syringes • Combined needle and syringe (CNS) sourced at UK level • GBUK product sourced which has 0. 1 ml graduations • Two sizes, 23 g and 25 g. Plan to use 23 g until exhausted and then switch to 25 g

Pfizer-Bio. NTech COVID-19 vaccine Dose preparation: • If the vaccine has previously been reconstituted, check that the time of reconstitution was within the last 6 hours • Clean top of vial with a single use alcohol swab and allow to air dry fully • Unwrap one of the 1 ml combined 23 g/25 mm blue hub needle and syringes provided (recommended needle length depends on body mass of patient. Longer length (38 mm) needles are recommended and being supplied for morbidly obese individuals to ensure the vaccine is injected into muscle). • Withdraw a dose of 0. 3 ml of diluted product for each vaccination. Ensure correct dose is drawn up • Any air bubbles should be removed before removing the needle from the vial in order to avoid losing any of the vaccine dose • It is not recommended that the needle between the vial and the patient is changed unless the needle is contaminated or damaged • For each additional dose, use a new sterile syringe and needle and ensure the vial stopper is cleansed with antiseptic swab before each withdrawal. Cleansing has to happen between each withdrawal even if they are done in close succession

Vaccine interchangeability • data is not yet available on the interchangeability of different COVID-19 vaccines. • therefore, every effort should be made to determine which vaccine the individual received and to complete with the same vaccine • for individuals who started the schedule and who attend for vaccination at a site where the same vaccine is not available, or where the first product received is unknown, it is reasonable to offer a single dose of the locally available product • this option is preferred if that individual is likely to be at immediate high risk or is considered unlikely to attend again • as the authorised Pfizer-Bio. NTech COVID-19 vaccine and the Astra. Zeneca vaccine which may be authorised for use in the UK are both based on the spike protein of the virus, it is likely that the second dose will help to boost the response to the first dose • further doses of vaccine are not required unless additional information becomes available

Pfizer-Bio. NTech COVID-19 vaccine Adverse events: Local reactions • In clinical trials, local reactions at the injection site were found to be fairly common. Over 80% of trial participants reported pain at the injection site. This occurred within 7 days after the injection and resolved after a few days. Systemic reactions • In clinical trials, the most frequently reported systemic reactions (reactions affecting the whole body) in participants were tiredness (>60%), headache (>50%), muscle aches (>30%), chills (>30%), joint pain (>20%) and a raised temperature (pyrexia) (>10%). These symptoms were usually mild or moderate in intensity and resolved within a few days after vaccination. • Vaccinated individuals should be advised that the COVID-19 vaccine may cause a mild fever which usually resolves within 48 hours. • This is a common, expected reaction and isolation is not required unless COVID-19 is suspected

Pfizer-Bio. NTech COVID-19 vaccine (1) Risk of allergic reactions Advice to Healthcare professionals This precautionary advice is being issued following two case reports of anaphylactoid reactions associated with administration of Pfizer Bio. Ntech COVID-19 vaccine. New advice: • Any person with a history of immediate-onset anaphylaxis to a vaccine, medicine or food, or anaphylaxis of unknown cause should not receive the Pfizer/Bio. NTech vaccine. A second dose of the Pfizer/Bio. NTech vaccine should not be given to those who have experienced anaphylaxis to the first dose of Pfizer/Bio. NTech vaccination • An anaphylaxis pack should be available at all times for all vaccinations. Vaccination should only be carried out in facilities where resuscitation measures are available. Please report any suspected adverse reactions via the Yellow Card scheme. To make a report or find out more about the Yellow Card COVID-19 reporting site please visit: Coronavirus Yellow Card reporting site

Pfizer-Bio. NTech COVID-19 vaccine (2) Risk of allergic reactions Advice to Healthcare professionals • Ensure the template clinical conversation script already provided is used. This includes a question on allergies. • Recipients of COVID-19 vaccine should be observed for a minimum of 15 minutes following vaccination for any immediate reactions. The MHRA have advised that individuals should be observed for longer than 15 minutes when indicated after a clinical assessment. • See FAQ 23 here http: //nww. immunisation. wales. nhs. uk/faqs-miscellaneous • An anaphylactic algorithm flow chart should be displayed in your observation or recovery area. The Resuscitation Council provide the following guidance (2008, reviewed 2016). • Emergency treatment of anaphylactic reactions: Guidelines for healthcare providers | Resuscitation Council UK • Ensure allergy history is accurately recorded on WIS, and that the details of any immediate vaccine reaction are added using the drop down definitions provided.

COVID-19 programme and eligible groups

Priority Group Risk Group 1 Residents in a care home for older adults and their carers 2 All those 80 years of age and over. Frontline health and Social care Workers 3 All those 75 years and over 4 All those 70 years and over Clinically extremely vulnerable individuals (not including pregnant women and those under 16 years of age) 5 All those 65 years of age and over 6 All individuals aged 16 to 64 years with underlying health conditions which put them at higher risk of serious disease and mortality 7 All those 60 years of age and over 8 All those 55 years of age and over 9 All those 50 years of age and over JCVI advise on priority groups for coronavirus (COVID-19) vaccination [updated 2 nd December 2020]

Groups with underlying health conditions As well as age, other risk factors have been identified that place individuals at risk of serious disease or death from COVID-19. These include groups with certain underlying health conditions and may include people who have: • chronic (long-term) respiratory disease • chronic heart disease • chronic kidney disease • chronic liver disease • chronic neurological disease • diabetes • a weakened immune system due to disease or treatment • asplenia or dysfunction of the spleen • morbid obesity (defined as BMI of 40 and above) • severe mental illness • Detailed information giving examples of conditions in each of the clinical risk groups which would make individuals aged 16 years and over eligible for vaccination is provided in the COVID-19 Green Book chapter.

Contraindications • Any person with an anaphylactic reaction to a previous dose of the Pfizer Bio. NTech vaccine • Any person with a confirmed anaphylactic reaction to any components of the Pfizer Bio. NTech vaccine • Any person with a history of immediate-onset anaphylaxis to a vaccine, medicine or food should not receive the Pfizer Bio. NTech vaccine • Any person with previous unexplained anaphylaxis • Carrying an Adrenalin Auto-injector is itself not a contraindication • Anaphylaxis to an allergen not listed above, such as bee venom, is not a contraindication

Contraindications cont’d • The Pfizer Bio. NTech COVID-19 m. RNA Vaccine BNT 162 b 2 contains polyethylene glycol (PEG), a known allergen commonly found in medicines and also in household goods and cosmetics. • Known allergy to PEG is extremely rare but would contraindicate receipt of this vaccine. (Sellaturay P et al, 2020). Patients with undiagnosed PEG allergy may have a history of unexplained anaphylaxis or of anaphylaxis to multiple classes of drugs. • https: //assets. publishing. service. gov. uk/government/uploads/sy stem/uploads/attachment_data/file/943663/Greenbook_chapter _14 a_v 3. pdf

Precautions Postponements: • If acutely unwell postpone until recovered • Defer for 4 weeks after onset of Covid-19 symptoms or positive test • Having prolonged COVID-19 symptoms is not a contraindication to receiving COVID-19 vaccine but if the patient is seriously debilitated, still under active investigation, or has evidence of recent deterioration, deferral of vaccination may be considered to avoid incorrect attribution of any change in the person’s underlying condition to the vaccine.

Precautions - pregnancy • COVID-19 vaccines have not been trialled in pregnant women • It is therefore not currently recommended that COVID-19 vaccines are given to women at any stage of pregnancy or to women who are planning a pregnancy within three months of the first dose • Vaccination should be postponed until completion of pregnancy and breast feeding has stopped. It is unknown whether Pfizer-Bio. NTech COVID-19 vaccine is excreted in human milk. A risk to the newborn/infants cannot be excluded. • Vaccinated women should avoid pregnancy for 2 months following their 2 nd vaccine dose • Pregnant women at high risk (including health care workers) should be offered vaccine as soon as possible after completion of pregnancy • If a woman finds out she is pregnant after she has started a course of vaccine, she should complete her pregnancy before finishing the recommended schedule • In the event of an inadvertent administration of COVID-19 vaccine to a pregnant woman, reassurance should be given and termination of pregnancy should not be recommended MHRA has put forward a very precautionary approach.

Pregnancy - continued Implications for COVID-19 clinics in Wales • Routine questioning about last menstrual period and/or pregnancy testing is not required before offering the vaccine • Pregnancy and breastfeeding should be excluded before vaccination with the Pfizer. Bio. NTech COVID-19 vaccine. Excluding women who know they are pregnant or breastfeeding is relatively straightforward. Potentially more challenging are women who don’t know they are pregnant but who nevertheless need to be “excluded before vaccination”. Mass vaccination centres and GP surgeries will need to defer vaccination in any women who are uncertain if they are pregnant. • In the event of an inadvertent administration of COVID-19 vaccine to a pregnant woman, reassurance should be given as the vaccine cannot cause infection in the mother or the baby. Termination of pregnancy following inadvertent immunisation should not be recommended. • Surveillance of inadvertent administration in pregnancy is being conducted for the UK by the PHE Immunisation Department, to whom such cases should be reported (Tel: 020 8200 4400).

Legal framework to administer COVID-19 Vaccine(s)

Amendments to legislation, came into force 9 th November 2020 The Human Medicines Regulations 2012 have been amended to strengthen existing regulations that allow for the temporary authorisation of the supply of unlicensed medicines, including vaccines, in response to certain public health threats. And makes three key changes to ensure that the UK has the available workforce to administer the COVID-19 and influenza vaccines: 1. Expanding the scope of patient group directions to allow for them to be issued for the administration of any medicine, including COVID-19 vaccines and flu vaccines, the supply of which has been temporarily authorised under regulation 174; 2. Introducing a new type of national immunisation protocol (regulation 247 A), to be authorised by Welsh Ministers, which will allow those who are registered healthcare professionals who do not normally vaccinate, and people who are not registered health care professionals, to safely administer a licensed or temporarily authorised COVID-19 or influenza vaccine; 3. Expand the workforce legally allowed to administer vaccines under National Health Service (NHS) and local authority occupational health schemes, so that additional health care professionals in the occupational health workforce will be able to administer these particular vaccines.

Using a Patient Group Direction (PGD) to give COVID 19 vaccine authorised under regulation 174 In response to certain public health threats, such as the current pandemic, the MHRA can temporarily authorise the supply of an unlicensed medicine or vaccine for use, under regulation 174 of The Human Medicines Regulations 2012, when it is satisfied that there is robust evidence to show the safety, quality and effectiveness of the medicine/vaccine. • In November 2020, new legislation amending The Human Medicines Regulations 2012 came into force. The Human Medicines (Coronavirus and Influenza) (Amendment) Regulations 2020 • Prior to this, PGDs could only be used for licensed medicines. The change to legislation allows medicines/vaccines which have been temporarily authorised for supply in the UK under regulation 174 to be used in accordance with a PGD. • Registered healthcare professionals who are allowed to work to a PGD may supply and administer COVID-19 vaccines, as temporarily authorised vaccines under Regulation 174. • The workforce that can administer under PGDs has not changed • Working with Welsh Medicines Information Centre (WMIC), Public Health Wales (PHW) will provide template PGDs for COVID-19 vaccines.

Protocols for the supply and/or administration of COVID-19 vaccine • Changes to the Human Medicines Regulations also brought about a new regulation 247 A. • While a disease is pandemic, regulation 247 A permits the supply or administration of a medicinal product used for vaccination or immunisation against coronavirus (or influenza) in accordance with a protocol that is approved by Welsh Ministers. • A national protocol will allow trained and competent non-registered healthcare workers (HCWs) as well as registered healthcare professionals who do not usually vaccinate, to administer COVID-19 vaccine. • The protocol will specify who can use it and will include information similar to that commonly found in PGDs. • The protocol will provide details of specific workforce requirements, alongside details of minimum training requirements • The provider is responsible for ensuring that its workforce are trained, competent and adequately supervised to safely deliver the activity they are employed to provide under the protocol. • It may be followed wholly from patient assessment through to post-vaccination by a single person. Alternatively, multiple healthcare workers may undertake different roles or stages in the patient vaccination pathway in accordance with the protocol. • The protocol template will be reviewed for use across Wales by PHW supported by WMIC, and provided to Welsh Government, to prepare advice for Welsh Ministers

Resources

Resources PHW are producing the following bilingual resources: • A 4 Information on Covid-19 vaccination for adults (4 page) • A 4 Covid-19 vaccination information for health and social care staff (4 page) • A 4 What to expect after your Covid-19 vaccination (4 page) • COVID-19 vaccination – information for adults DL leaflet • Why do I have to wait for my COVID-19 vaccine? DL leaflet • COVID-19 vaccine record card • Vaccine reward stickers These resources will be available soon to order and download from https: //phw. nhs. wales/services-and-teams/health-information-resources/ The leaflets can be downloaded now from the VPDP COVID-19 vaccination intranet page • Clinical conversation script (aide-mémoire) bilingual

A 4 Information leaflets 4 page bilingual tilt and turn

DL leaflets 50

Reward sticker Vaccine record card – size A 7 x 4 pages tilt and turn

Clinical conversation script http: //nww. immunisation. wales. nhs. uk/covid-19 -vaccination-programme#Resources

Accessibility & resources Over the coming weeks the COVID-19 vaccination – information for adults, What to expect after your COVID-19 vaccination, Why do I need to wait for my COVID-19 vaccination? leaflets and the clinical conversation script will also be available to download in: • Easy Read format • Large text • Approximately 10 other languages • British Sign Language video Other formats will be explored as the programme is rolled out

Sources of information • Green Book COVID-19 chapter 14 a www. gov. uk/government/collections/immunisation-againstinfectious-disease-the-green-book • PHW: http: //nww. immunisation. wales. nhs. uk/covid-19 -vaccination-programme (intranet only) • PHW micro-site: https: //phw. nhs. wales/topics/immunisation-and-vaccines/covid-19 -vaccinationinformation/ • Public Health England Coronavirus resources www. gov. uk/government/collections/immunisation • GOV. UK Coronavirus (COVID-19) in the UK https: //coronavirus. data. gov. uk/ • WHO COVID-19 Worldwide Dashboard https: //covid 19. who. int/? gclid=EAIa. IQob. Ch. MInr 6 P 36 Dc 7 AIVBWHm. Ch 3 Isw. IXEAAYASAAEg. IPT_D _Bw. E • LSHTM COVID-19 vaccine tracker https: //vac-lshtm. shinyapps. io/ncov_vaccine_landscape/ • Royal College of Nursing. Immunisation services and large-scale vaccination delivery during COVID-19. www. rcn. org. uk/clinical-topics/public-health/immunisation-services-andlarge-scale-vaccination-delivery-during-covid-19#planningandriskassessmentprocess

Further information • UK vaccine policy can be found in the online publication commonly referred to as the "Green Book". This can be found on the Immunisation page of the GOV. UK website • Green Book recommendations are based upon JCVI’s expert opinion and should always be followed, even when they differ from those in the vaccine manufacturer. • PHE COVID-19 immunisation programme Information for healthcare practitioners This document provides additional information, answers to frequently asked questions and actions to take in the event of inadvertent errors. • It is important to read this document before you start vaccinating and also to refer to it regularly as it will be updated as more information becomes available and to address any issues or frequently arising questions as COVID-19 vaccines are delivered more widely.

Training overview

Pre-requisites to administering COVID-19 vaccine Before administering COVID-19 vaccine, you should have: • Undertaken training in the management of anaphylaxis and Basic Life Support as specified by policy for your local area • Undertaken any additional statutory and mandatory training as required by your employer • Received COVID-19 vaccine training through a taught session and/or the e. Learning for Healthcare COVID-19 vaccine elearning programme • Received practical training in COVID-19 vaccine preparation and administration • Completed the COVID-19 vaccinator competency assessment tool • An appropriate legal framework to supply and administer COVID-19 vaccine in place e. g. patient specific prescription, Patient Specific Direction (PSD), Patient Group Direction (PGD) or Protocol • Accessed and familiarised yourself with the following key documents: Green Book COVID-19 chapter and the PHE ‘COVID-19 immunisation programme information for healthcare practitioners’ document

• The COVID-19 vaccination e. Learning programme consists of a core knowledge session and a vaccine-specific session with accompanying assessment sessions for each. • More vaccine-specific sessions will be added as and when more COVID-19 vaccines become available and authorised for supply in the UK. • All those undertaking this e-learning should complete the core knowledge session as this is designed to provide essential knowledge about COVID-19 and the key principles of immunisation needed to deliver the vaccine. • Learners should then complete the vaccine specific session(s) for the vaccine(s) they are going to give as these provide more detailed information. The assessment sessions should be completed after each knowledge session. • Further information available here: www. wales. nhs. uk/immslearning

Competency assessment tool The competency assessment tool is divided into three areas 1. Knowledge 2. Core clinical skills 3. The clinical process/procedure for vaccine administration It should be completed by those who are new to or returning to immunisation before sharing with their supervisor. Experienced immunisers can use it as a self-assessment tool. The supervisor carrying out the assessment should: i. review the self-assessment, discussing any areas identified as ‘need to improve’ ii. observe performance in the provision of immunisations/advice and indicate whether each competency is ‘met’ or ‘needs to improve’ in the ‘supervisor review’ column iii. if improvement is needed, help to develop an action plan to enable the achievement of the required level of competence and plan a further assessment iv. sign off the section at the bottom of the assessment when competency is agreed COVID-19 Vaccinator competency assessment tool [PHE, 2020]

COVID-19 vaccinator competency assessment tool

Acknowledgements • Colleagues in Public Health England • Vaccine Preventable Disease Programme team

Additional slides FAQs

Protocol for “Red Care Homes”: Care Home Staff booked into mass testing facilities: • If asymptomatic care home staff testing picks up a positive, repeat testing to clarify if possible false positive to be done as soon as possible – if second test negative and no other positives care home staff can proceed to attend the mass vaccination clinic to be vaccinated. Deferral of appointment may be necessary whilst repeat testing is undertaken. • New confirmed positive in a specific care home staff member: staff member and close contacts MUST NOT attend a mass vaccination clinic for vaccination. A risk assessment may be conducted to determine if there are members of care home staff within that care home, who can be confidently identified as not being significant contacts of the cases and who have negative test results to assess if it safe for these staff members to attend. If however there is evidence of spread of infection to residents / an incident or outbreak is declared then none of the care home staff should attend for vaccination. • For a care home that has been under incident / outbreak conditions if there have been no new cases for 20 days with two rounds of negative staff testing, staff may be invited for vaccination. FAQs now available here: http: //nww. immunisation. wales. nhs. uk/faqs-covid-19

Protocol for “Red Care Homes”: Staff attending care homes to deliver vaccinations: • If a care home has an ongoing outbreak / incident residents who are positive or close contacts would not be eligible for vaccination at this time. Other residents and staff within the care home could be offered vaccination. Staff attending to deliver the vaccinations would be required to wear FRSM mask, eye protection (if risk assessed as required) , gloves and aprons according to PHW advice note for PPE in care homes appendix table 1 phw. nhs. wales/Social. Care. PPEAdvisory. Note FAQs now available here: http: //nww. immunisation. wales. nhs. uk/faqs-covid-19

What types of waste may be generated and how should it be disposed of? Description of Waste • Personal contact and contaminated waste from routine vaccination: • PPE to vaccinate, prepare the vaccine and any contact with the individual being vaccinated. • Any other PPE worn by the vaccinator or support staff or the person being vaccinated • Used cotton balls/arm swabs and surgical tape, any blood stained items (except sharps), vial swabs • Used tissues, disposable pulp products Requirement • Place in the offensive waste stream “tiger stripe bag” – a yellow bag with a black stripe. Secure when ¾ full with swan neck and zip tie or tape • Store for disposal/collection as per local arrangements Note • Where possible bodily fluids e. g. vomit shall be flushed to sewer otherwise provision to solidify required e. g. gel agent. Liquid waste must not be placed in waste bag. How to manage healthcare waste from mass vaccination clinics – COVID-19 – PHW/SOP

What types of waste may be generated and how should it be disposed of? Description of Waste Clinical Sharps and Injecting Equipment • Needles, syringes, any other sharp • Vaccine Drug Vials • Diluent vial i. e. “Sodium Chloride 0. 9% for injection” (there will be excess diluent per vial) Requirement • Place in a yellow lidded sharp box assembled and labelled according to guidance • Use temporary closure mechanism between use and session • Complete labelling and use final closure to seal when sharp ¾ fill line is reached • Store for disposal/collection as per local arrangements Note • Place absorbent material in the base of the sharps box on assembly e. g. supplied gel pad. If not available, consider other absorbent materials e. g. paper roll /hand towel or other gel agent • Sharps boxes are not leak proof • Do not squirt diluent into sinks • Sodium Chloride 0. 9% is being used as the diluent, and only 1. 8 ml will be used for each vaccine, the remaining contents must be discarded. How to manage healthcare waste from mass vaccination clinics – COVID-19 – PHW/SOP

What types of waste may be generated and how should it be disposed of? Description of Waste: Vaccine packaging & Inserts • Trays for vaccine vials (described as pizza trays) • Outer wrapping of the vial trays • Information inserts on the vaccine Requirement • Deface and obscure any labelling using a permanent marker pen or similar • Cut up the plastic trays into several pieces so they are destroyed and rendered unusable • Deface and obscure inserts and cut up • All packaging (cardboard boxes and plastic trays, inserts) must be rendered unusable / destroyed. • Where available and a shredder suitable then packaging should be shredded then placed in suitable recycling; • If no shredders are available then place the packaging waste in an offensive waste bag, “tiger stripe bag” – a yellow bag with a black stripe. Secure when ¾ full with swan neck and zip tie or tape This is interim advice only and we are awaiting confirmation from Cabinet cell on this issue

What types of waste may be generated and how should it be disposed of? General Waste • Outer packaging including plastic wrappings, general paper, paper hand towels following hand hygiene. • Vigilance is required with waste management of commercially sensitive material e. g. packaging, is potentially a target/concern in relation to counterfraud. Specialist advice is being taken to mitigate this risk. • Disposable surface disinfectant wipes and cleaning wipes/cloths empty hand sanitiser bottles, disposable cups/bottle and other general waste items • Place in the general waste in a black bag. Secure when ¾ full with swan neck and zip tie or tape • Please check locally with your waste manager for recycling arrangements e. g. empty hand sanitiser bottles, clean cardboard packaging etc. • Other Clinical Waste If you are generating any clinical wastes not covered above then please liaise with the Health Board Waste Manager and Waste Contractor further help • Welsh Health Technical Memorandum. 07 -01: Safe management of healthcare waste. This guidance can be found here: https: //nwssp. nhs. wales/ourservices/specialist-estatesservices/publications-andinformation/welsh-health-technicalmemoranda-whtms-health-technical-memoranda-htms/ How to manage healthcare waste from mass vaccination clinics – COVID-19 – PHW/SOP

Administering COVID-19 vaccine to individuals with a bleeding disorder • Individuals with bleeding disorders may be vaccinated intramuscularly if, in the opinion of a doctor familiar with the individual's bleeding risk, vaccines or similar small volume intramuscular injections can be administered with reasonable safety by this route. • If the individual receives medication/treatment to reduce bleeding, for example treatment for haemophilia, intramuscular vaccination can be scheduled shortly after such medication/treatment is administered. • A fine needle (23 or 25 gauge) should be used for the vaccination, followed by firm pressure applied to the site (without rubbing) for at least 2 minutes (ACIP, 2019). The individual/carer should be informed about the risk of haematoma from the injection.

Administering COVID-19 vaccine to individuals taking anticoagulants • Individuals on stable anticoagulation therapy, including individuals on warfarin who are up-to-date with their scheduled INR testing and whose latest INR was below the upper threshold of their therapeutic range, can receive intramuscular vaccination. • If in any doubt, consult with the clinician responsible for prescribing or monitoring the individual’s anticoagulant therapy.
- Slides: 70