COVALENT NOMENCLATURE Writing Names and Formulas H 2









- Slides: 12

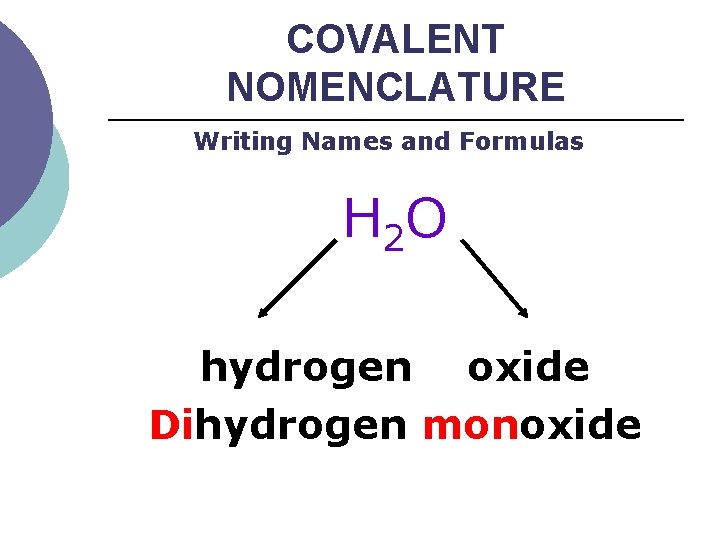
COVALENT NOMENCLATURE Writing Names and Formulas H 2 O hydrogen oxide Dihydrogen monoxide

MOLECULAR COMPOUNDS Another word for covalent molecules ¡ usually binary; called a molecule ¡ usually contain two nonmetals; solid, liquid or gas ¡ Sometimes contains a metalloid and a nonmetal.

NAMING MOLECULAR COMPOUNDS Rules: 1. Use prefixes to identify the number of atoms present. 2. If only one of the first atom (nonmetal), then NO prefix is used. 3. Last element ends in “ide”. * Can combine in different ratios so MUST use prefixes.

NAMING MOLECULAR COMPOUNDS Prefixes: 1 - mono 2 - di 3 - tri 4 – tetra 5 - penta 6 - hexa 7 - hepta 8 - octa 9 - nona 10 - deca

NAMING MOLECULAR COMPOUNDS Examples: ¡ CO 2 carbon dioxide ¡ PCl 3 phosphorus trichloride ¡ N 2 O dinitrogen monoxide ¡ P 2 O 3 diphosphorus trioxide

PRACTICE 1. NCl 3 2. CS 2 3. Si 2 Br 6 4. S 4 N 4 5. SF 6 6. N 2 O 5

WRITING MOLECULAR COMPOUNDS 1. Write the symbol of the first element adding a subscript of the number of atoms present. If only one of the first atom (nonmetal), then NO number is used. 2. Write the symbol of the second element adding a subscript of the number of atoms present.

PRACTICE A. boron trifluoride B. silicon tetrachloride C. iodine heptafluoride D. tetraphosphorus decoxide E. chlorine trifluoride F. sulfur dichloride

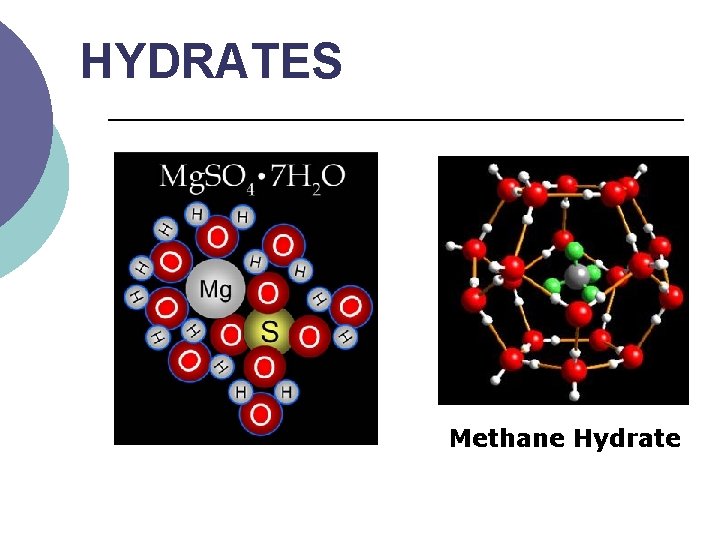
HYDRATES ¡ ¡ ¡ A crystalline compound in which its ions are attached to one or more water molecules. Hydrates are solids with water molecules trapped in them. The “ ” means that waters are attached.

HYDRATES Methane Hydrate

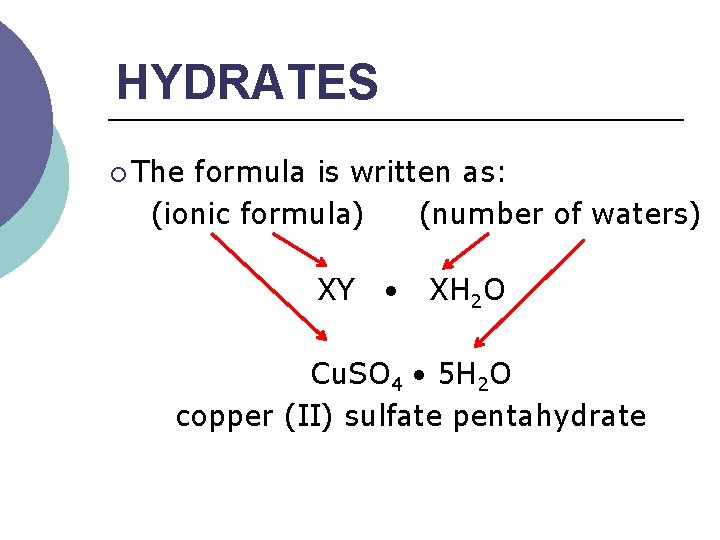
HYDRATES ¡ The formula is written as: (ionic formula) (number of waters) XY XH 2 O Cu. SO 4 5 H 2 O copper (II) sulfate pentahydrate

HYDRATES Use the same prefixes as with covalent compounds: mono – 1 penta – 5 nona -9 di - 2 hexa – 6 deca - 10 tri - 3 hepta -7 tetra - 4 octa - 8 Practice Naming ¡ Na 3 PO 4 2 H 2 O ¡ Sr. Cl 2 6 H 2 O ¡ Ba(OH)2 8 H 2 O