Covalent Molecules Review of Ionic Bonding Ionic bonds

Covalent Molecules

Review of Ionic Bonding Ionic bonds occur between a metal and a nonmetal, or between a positively charged particle and a negatively charged particle. n Ionic bonds involve a transfer of electrons. n n Can you identify which of the following are joined by ionic bonds? – Na. Cl – CO 2 - K 3 N - N 2 - Mg. Br 2 - Al 2 O 3

Covalent Bonding Covalent bonds occur between two nonmetals. n A molecule is formed when two or more atoms bond covalently. n Covalent bonds involve sharing of electrons. n

Diatomic Molecules Certain elements occur in nature as diatomic molecules and not as single atoms (more stable as the molecule) n Examples: n – H 2 , N 2 , O 2 , – F 2, Cl 2, Br 2, I 2 n Diatomic Molecules are bonded together by covalent bonds.
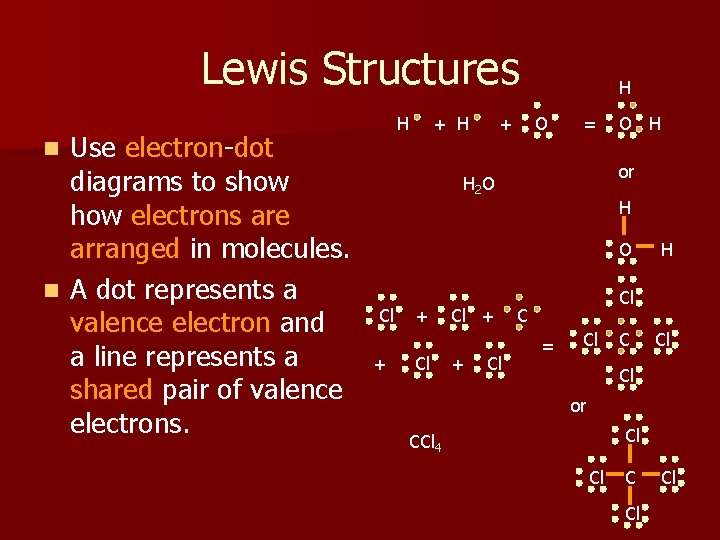
Lewis Structures Use electron-dot diagrams to show electrons are arranged in molecules. n A dot represents a valence electron and a line represents a shared pair of valence electrons. H + H H O + = n O H or H 2 O H O Cl + + Cl H Cl C = Cl Cl or Cl CCl 4 Cl Cl

Important Definitions n Chemical Formula: a representation, using symbols, showing the different types and number of atoms that make up a given compound or molecule n Subscript: small number, written behind an element, showing how many atoms of that element are found in a compound or molecule

Naming Molecular Compounds The first element written in the formula is always written first, using the element name. n The second element is named using the root of the element and the suffix “-ide”. n Prefixes are used to indicate the number of atoms of each type that are present in the compound. – Exception: Do not use the prefix mono- for the first element n Drop the final letter of the prefix when the element name begins with a vowel n

Table 1: Common Prefixes Used in Covalent Compounds Number of atoms Prefix 1 Mono- 6 Hexa- 2 Di- 7 Hepta- 3 Tri- 8 Octa- 4 Tetra- 9 Nona- 5 Penta- 10 Deca-

What Does Naming Look Like? 1. 2. 3. 4. CCl 4: Carbon tetrachloride CO: Carbon monoxide NF 3: Nitrogen trifluoride NO: Nitrogen monoxide

Now, you practice 1. 2. 3. 4. 5. 6. 7. 8. 9. 10. CO 2: Carbon monoxide ________________ Sulfur dioxide SO 2: ________________ Disulfur trioxide S 2 O 3: Dinitrogen ________________ monoxide N 2 O: ________________ Phosphorous pentachloride PCl 5: ________________ Oxygen difluoride OF 2: _______________ Dinitrogen tetroxide N 2 O 4: Tetrasulfur _______________ tetranitride S 4 N 4: _______________ Silicon tetrachloride Si. Cl 4: Boron _______________ trifluoride BF 3: ________________

What’s the formula? Se. F 6 Selenium hexafluoride: ______ Si 2 Br 6 2. Disilicon hexabromide: _____ B 2 Si 3. Diboron silicide: _____ IF 5 4. Iodine pentafluoride: ____ B 6 Si 5. Hexaboron silicide: ____ 6. Diboron tetrabromide: B_______ 2 Br 4 7. Diphosphorous pentoxide: P______ 2 O 5 4 8. Carbon tetrafluoride: CF ____ 4 S 5 9. Tetraphosphorous pentasulfide: P_______ S 6 O 10. Hexasulfur oxide: _____ 1.
- Slides: 11