Covalent Molecular Bonds Covalent Molecular Bond Definition Covalent

Covalent (Molecular) Bonds

Covalent (Molecular) Bond Definition • Covalent –“sharing, valence electrons” • Non-metals bonded to non-metals • Bond based on shared electrons between two atoms • Forms a molecule (hence the name)

Covalent (Molecular) Bond Properties • Very strong bond, however often not a strong force of attraction between molecules. • As a result molecular compounds have the lowest melting/boiling points

Types of Covalent Bonds • Single- one shared pair of e • Double- two shared pairs of e • Triple- three shared pairs of e. Strength: 3>2>1 Q: How do you know what types will form? A: Use VSEPR!

Drawing Covalent Bonds using V. S. E. P. R. Valence Shell Electron Pair Repulsion According to VSEPR theory, the repulsion between electron pairs causes molecular shapes to adjust so that the valenceelectron pairs stay as far apart as possible. -Like repels like (+ repels +)(- repels -)

VSEPR Steps: • Place either the single element or the C in the middle and arrange the others around it • Draw the Lewis Dot Diagram for each element • Draw a line connecting the valence electrons to represent one pair of electrons until each element fulfills the octet rule

Single Bond Example #1 • F 2 -Group 17 diatomic molecules are single bonds and formed this way!

Single Bond Example #2 • H 2 O

Single Bond Example #3 • CH 4

Double Bond Example #1 • CO 2

Double Bond Example #2 • NO

Triple Bond Example #1 • N 2

Triple Bond Example #1 • HCN

On notebook paper, use VSEPR to draw the following: Carbon tetrachloride (CCl 4) Ammonia (NH 3) Water (H 2 O) Nitrogen (N 2) Hydrogen (H 2) Phosphorous trichloride (PCl 3) Carbon dioxide (CO 2) Ethyne (C 2 H 2)
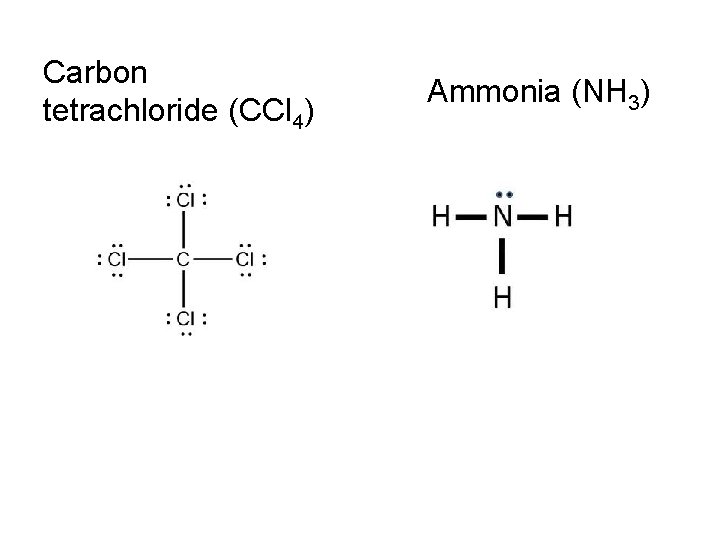
Carbon tetrachloride (CCl 4) Ammonia (NH 3)
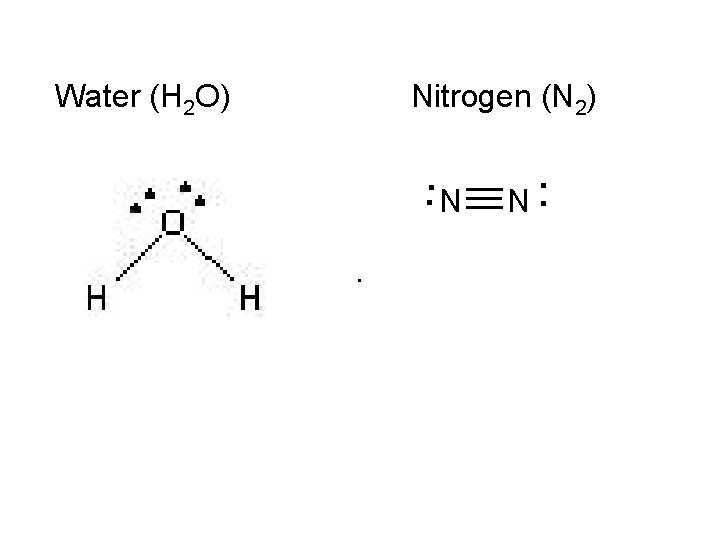
Water (H 2 O) Nitrogen (N 2) __. . . N __ __ N. .
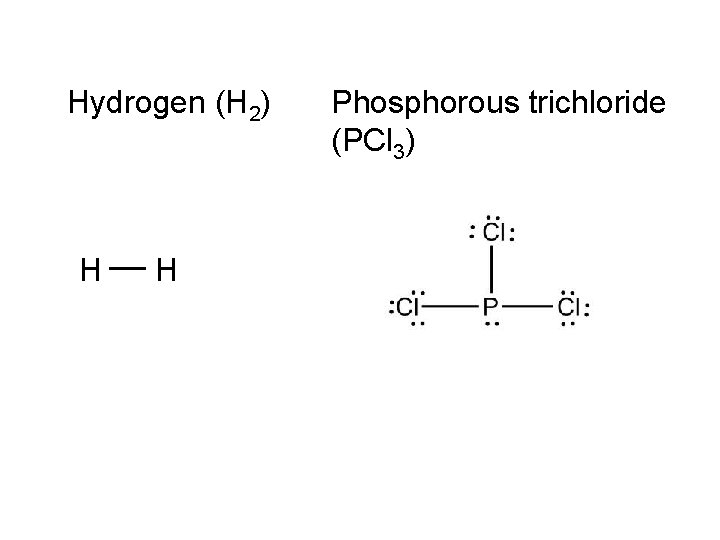
Hydrogen (H 2) H __ H Phosphorous trichloride (PCl 3)
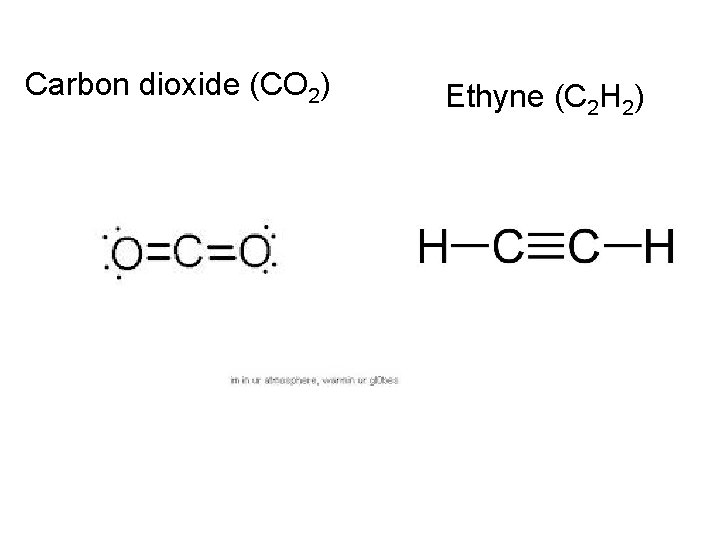
Carbon dioxide (CO 2) Ethyne (C 2 H 2)

rill Decide whether each substance is composed of covalent or ionic bonds. 1. Methane CH 4 2. Carbon Dioxide CO 2 3. Potassium Bromide KBr

Naming Covalent Bonds

Steps to Naming Covalent Bonds • Write name of first element – Use prefix if there’s more than 1 • Write name of second element, changing ending to “ide” – Use prefix always

Prefixes 1 mono 2 di 3 tri 4 tetra 5 penta 6 hexa 7 hepta 8 octa 9 nona 10 deca-
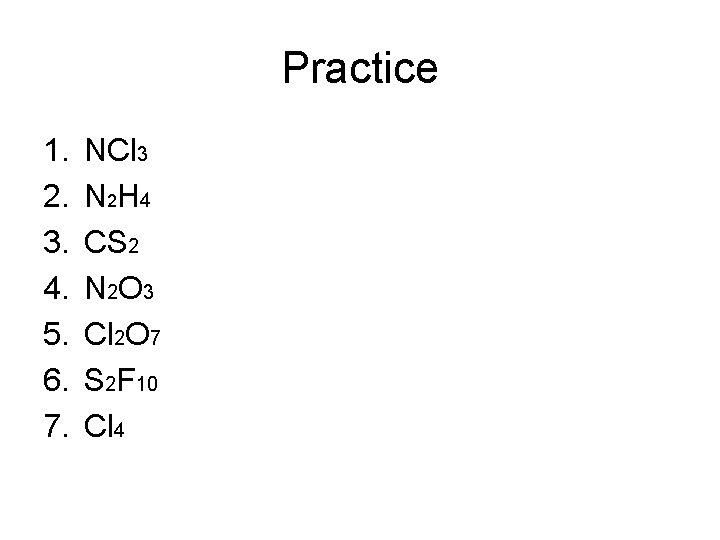
Practice 1. 2. 3. 4. 5. 6. 7. NCl 3 N 2 H 4 CS 2 N 2 O 3 Cl 2 O 7 S 2 F 10 Cl 4

Practice-ANSWERS 1. 2. 3. 4. 5. 6. 7. NCl 3 Nitrogen trichloride N 2 H 4 dinitrogen tetrahydride CS 2 carbon disulfide N 2 O 3 dinitrogen trioxide Cl 2 O 7 dichlorine heptoxide S 2 F 10 disulfur decafluoride Cl 4 tetrachloride
- Slides: 24