Covalent Ionic Metallic and Network Covalent Bonding Intramolecular

Covalent, Ionic, Metallic, and Network Covalent Bonding Intramolecular

Bonds q Forces that hold groups of atoms together and make them function as a unit. v Ionic bonds – transfer of electrons v Covalent bonds – sharing of electrons

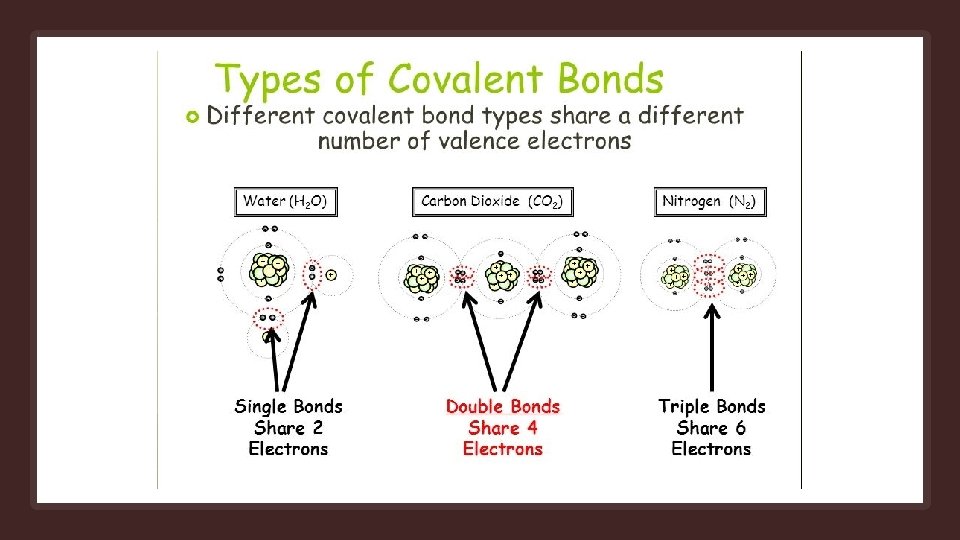
Covalent Bonds Occurs when pairs of electrons are shared by atoms Atoms will covalently bond with other atoms in order to gain more stability, which is gained by forming a full electron shell. By sharing their outer most (valence) electrons, atoms can fill up their outer electron shell and gain stability Nonmetals will readily form covalent bonds with other nonmetals
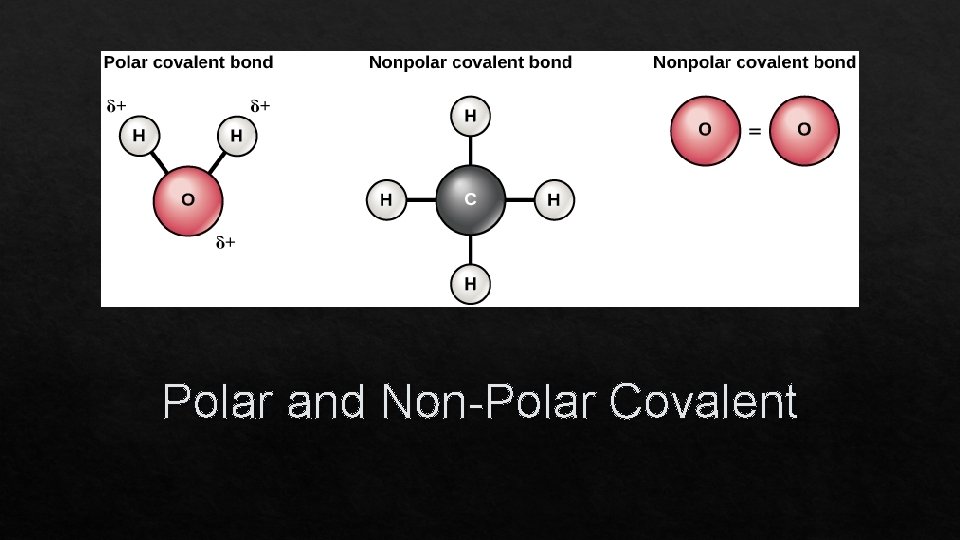
Polar and Non-Polar Covalent
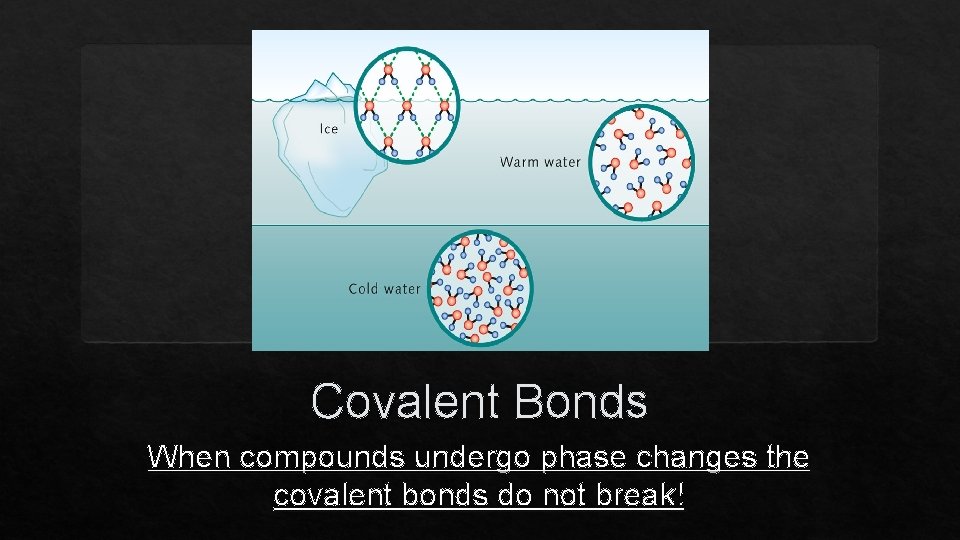
Covalent Bonds When compounds undergo phase changes the covalent bonds do not break!
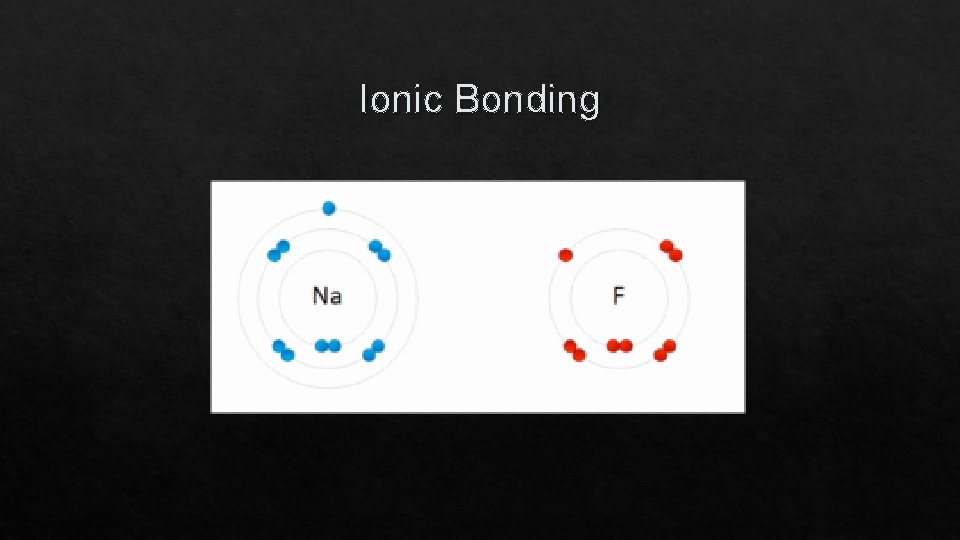
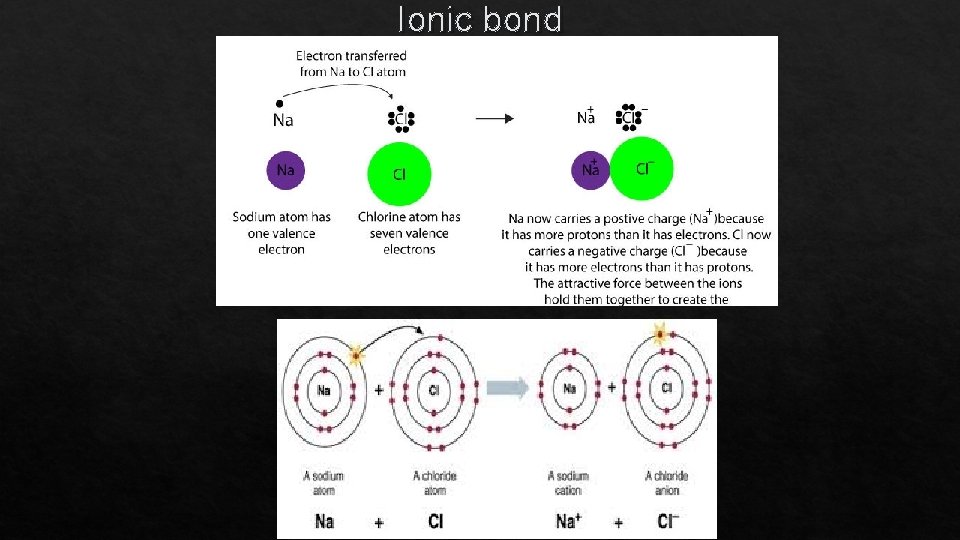
Ionic Bonding

The Octet Rule – Ionic Compounds Ionic compounds form so that each atom, by gaining or losing electrons, has an octet of electrons in its highest occupied energy level. Metals lose electrons to form positively-charged cations Nonmetals gains electrons to form negatively-charged anions

Ionic Bonding: The Formation of Sodium Chloride q Sodium has 1 valence electron q Chlorine has 7 valence electrons q An electron transferred gives each an octet Na: 1 s 22 p 63 s 1 Cl: 1 s 22 p 63 s 23 p 5
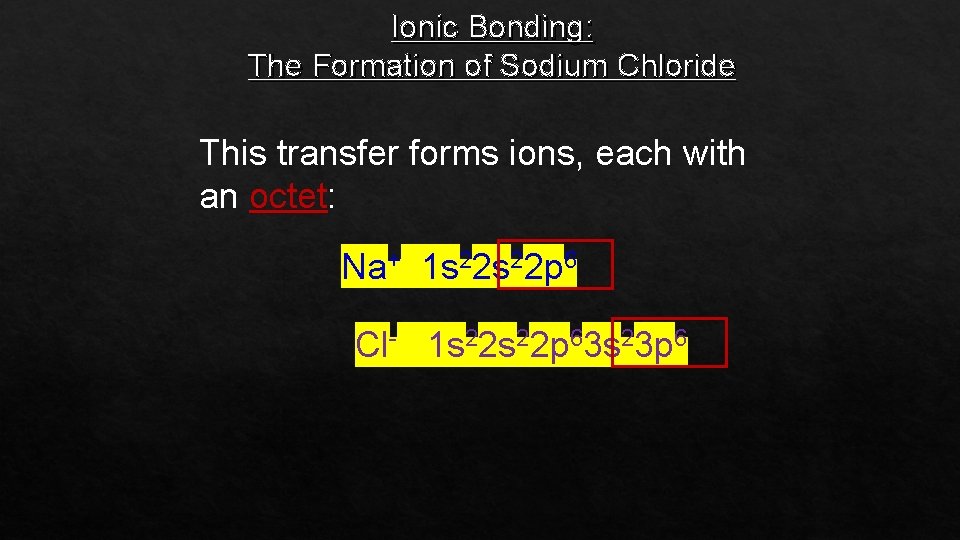
Ionic Bonding: The Formation of Sodium Chloride This transfer forms ions, each with an octet: Na+ 1 s 22 p 6 Cl- 1 s 22 p 63 s 23 p 6

Ionic Bonding: The Formation of Sodium Chloride The resulting ions come together due to electrostatic attraction (opposites attract): Na+ Cl. The net charge on the compound must equal zero
Ionic bond

Examples of Ionic compounds Mg 2+Cl-2 Magnesium chloride: Magnesium loses two electrons and each chlorine gains one electron Na+2 O 2 - Sodium oxide: Each sodium loses one electron and the oxygen gains two electrons Al 3+2 S 2 -3 Aluminum sulfide: Each aluminum loses two electrons (six total) and each sulfur gains two electrons (six total)
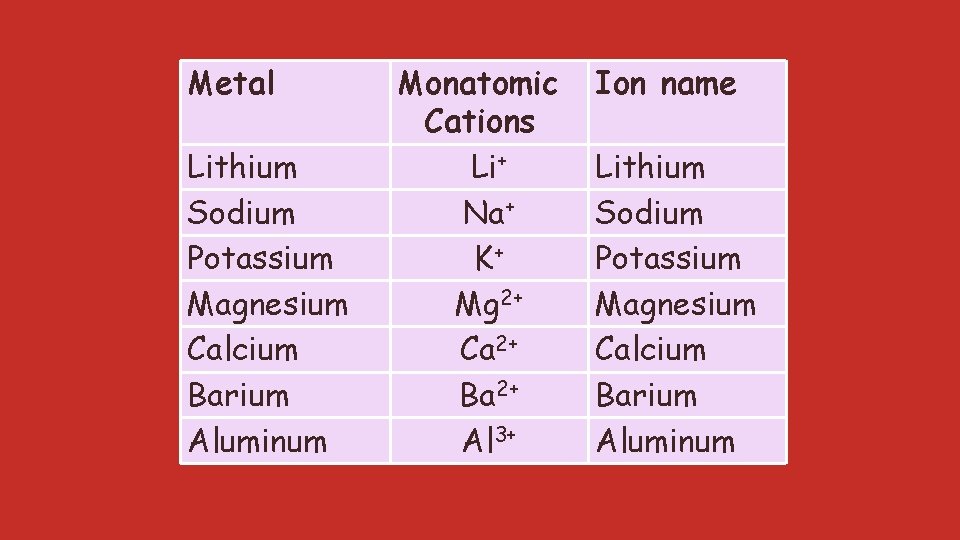
Metal Lithium Sodium Potassium Magnesium Calcium Barium Aluminum Monatomic Cations Li+ Na+ K+ Mg 2+ Ca 2+ Ba 2+ Al 3+ Ion name Lithium Sodium Potassium Magnesium Calcium Barium Aluminum
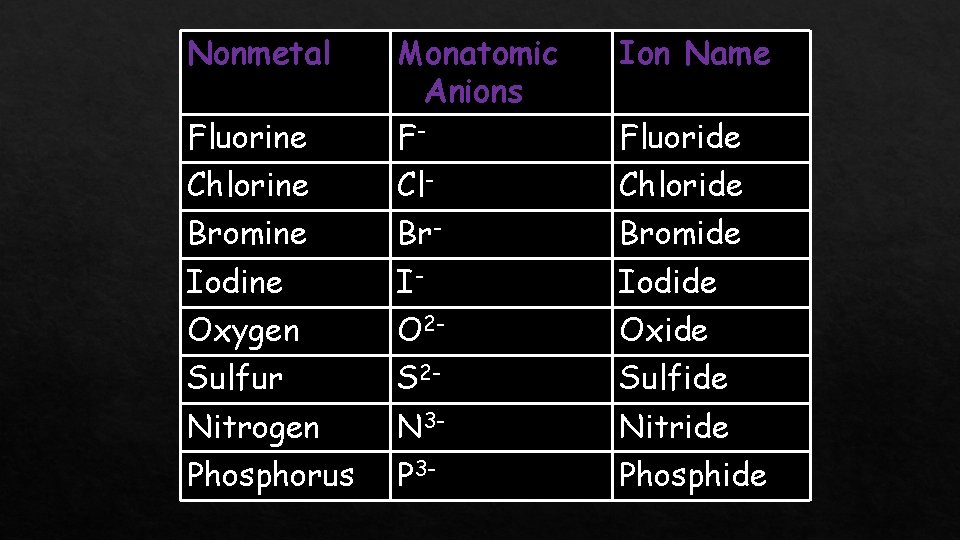
Nonmetal Ion Name Fluorine Monatomic Anions F- Chlorine Cl- Chloride Bromine Br- Bromide Iodine I- Iodide Oxygen O 2 - Oxide Sulfur S 2 - Sulfide Nitrogen N 3 - Nitride Phosphorus P 3 - Phosphide Fluoride

Sodium Chloride Crystal Lattice Ionic compounds form solid crystals at ordinary temperatures. Ionic compounds organize in a characteristic crystal lattice of alternating positive and negative ions. All salts are ionic compounds and form crystals.
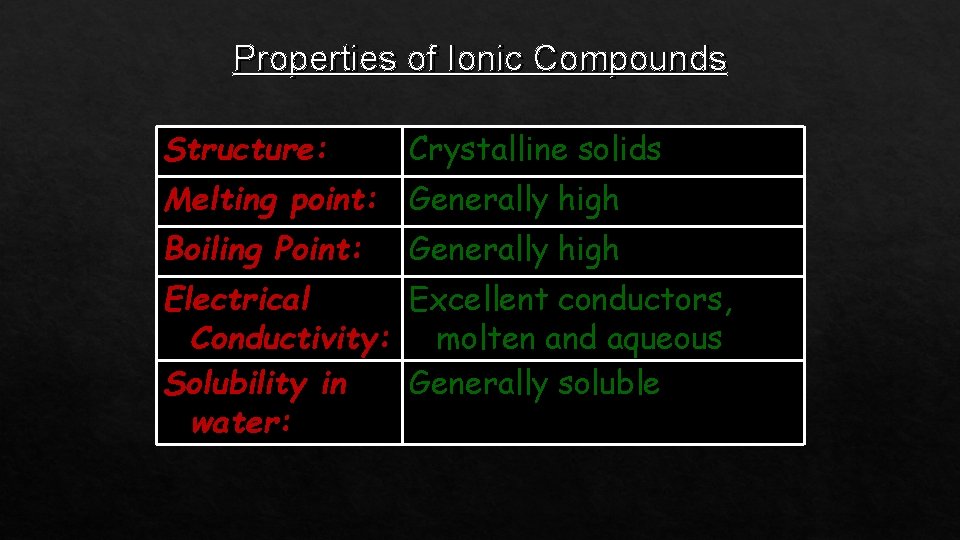
Properties of Ionic Compounds Structure: Crystalline solids Melting point: Generally high Boiling Point: Generally high Electrical Excellent conductors, Conductivity: molten and aqueous Solubility in Generally soluble water:

Strength of an Ionic Bond We can determine the strength of an ionic bond using a version of Coulomb’s Law: F= 2 Q 1 Q 2/r The stronger the bond, the higher the MP, BP, viscosity & vapor pressure.

Order the following from highest to lowest melting point: Na. F vs Mg. O Li. F vs Li. Cl Na. Cl vs Na. F Sr. S vs Ba. Se Be. S vs KCl K 2 P vs Ca 3 N 2

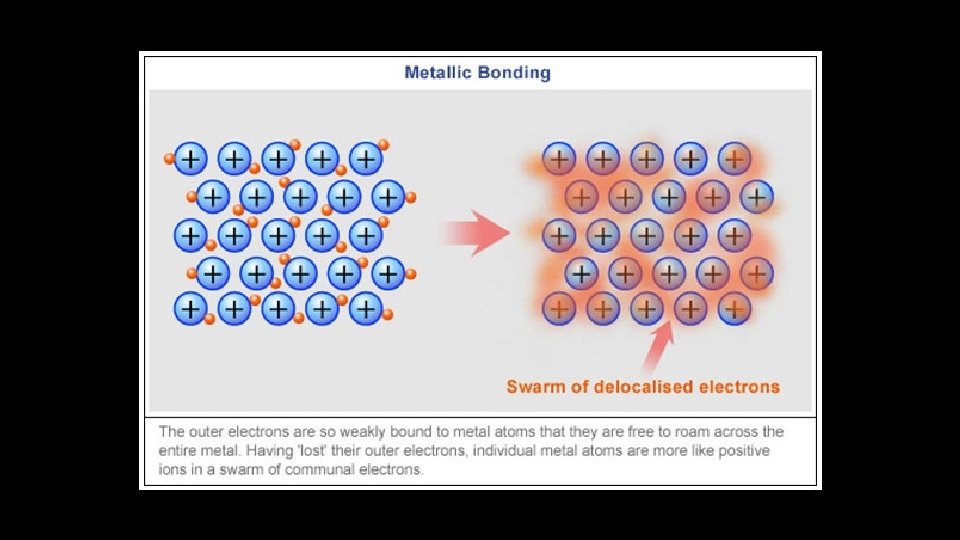
Metallic Bonding Strong forces of attraction are responsible for the high melting point of most metals.
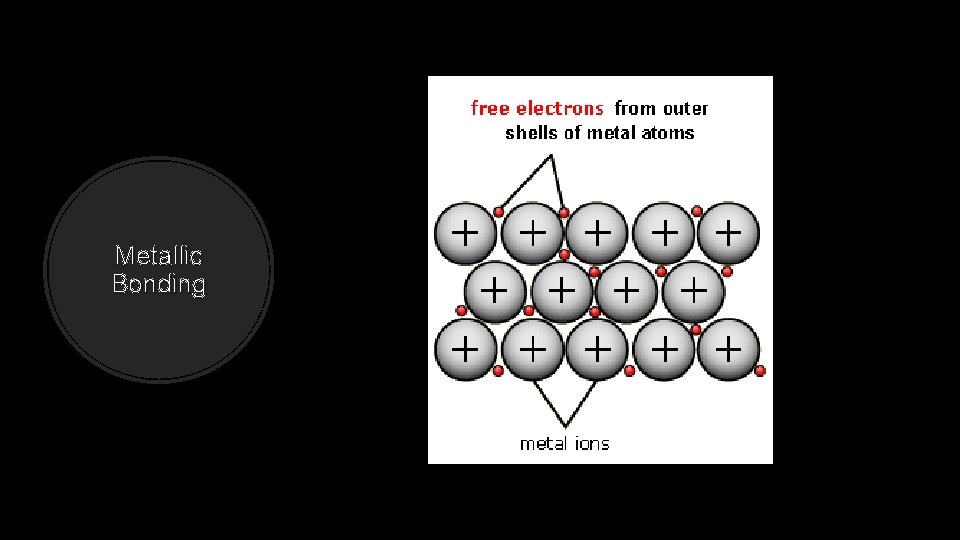
Metallic Bonding
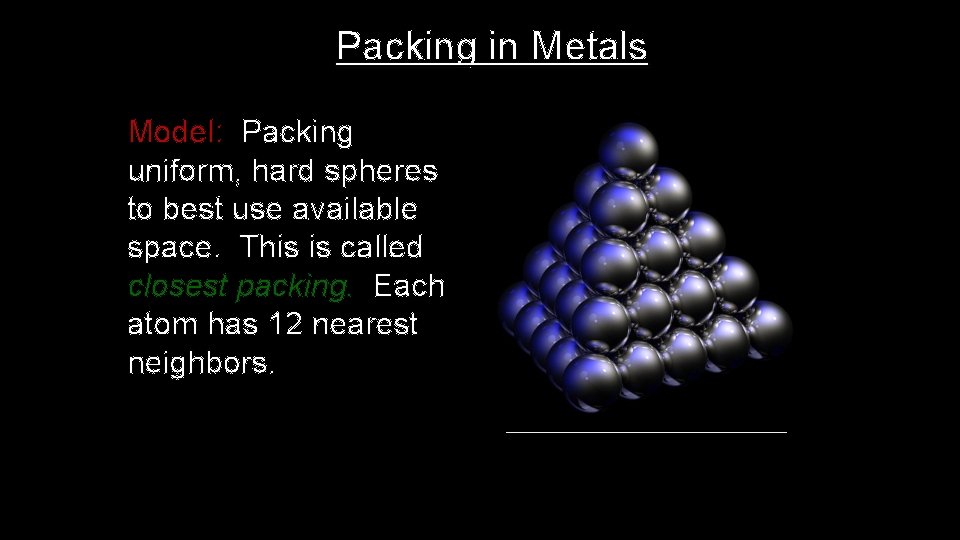
Packing in Metals Model: Packing uniform, hard spheres to best use available space. This is called closest packing. Each atom has 12 nearest neighbors.
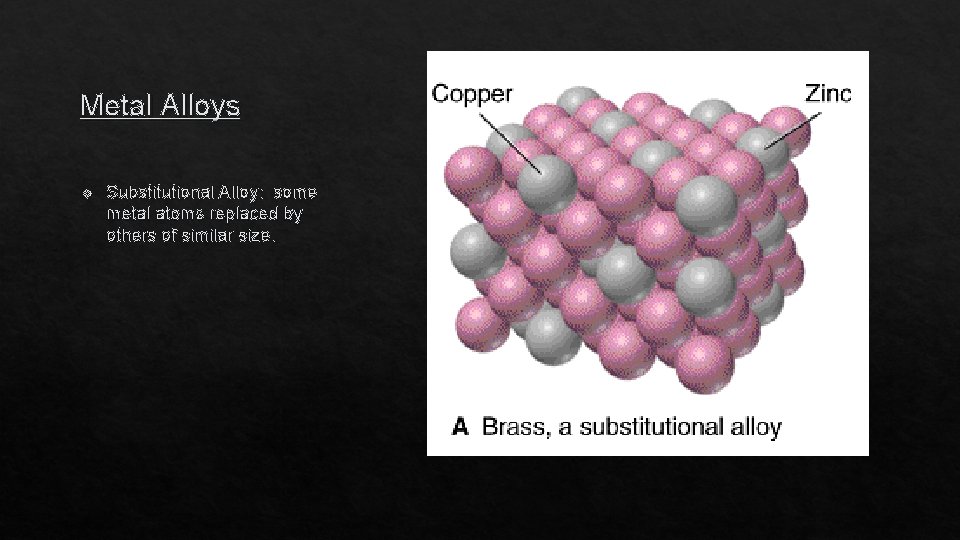
Metal Alloys v Substitutional Alloy: some metal atoms replaced by others of similar size.
Metallic Bonding Melting point increases across a period from left to right as charge increases due to increasing # of valence electrons participating in metallic bonding

Properties of Metals q Metals are good conductors of heat and electricity q Metals are malleable q Metals are ductile q Metals have high tensile strength q Metals have luster
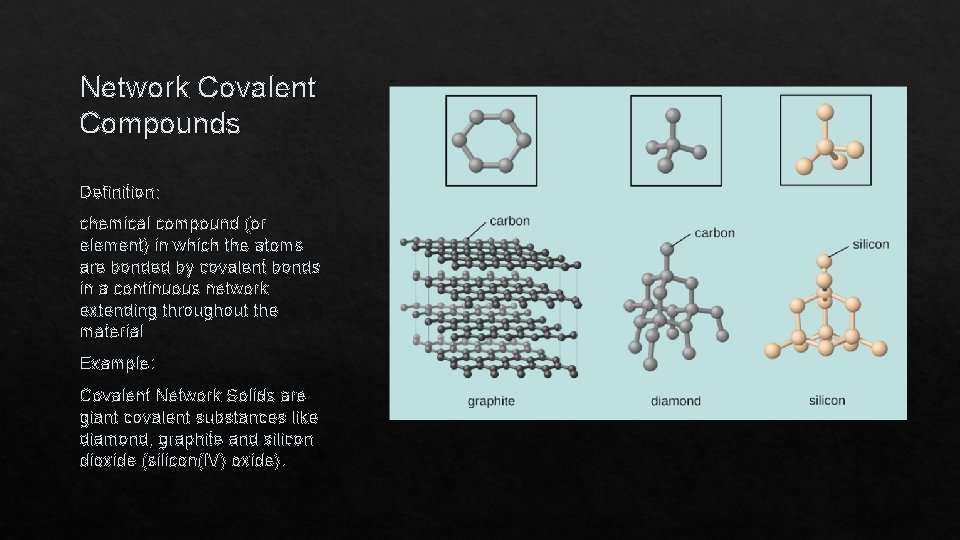
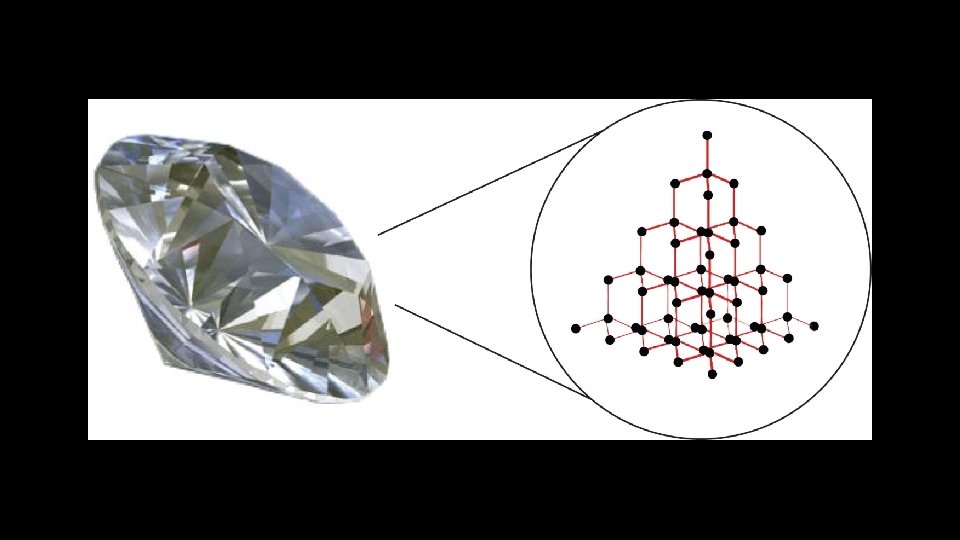
Network Covalent Compounds Definition: chemical compound (or element) in which the atoms are bonded by covalent bonds in a continuous network extending throughout the material Example: Covalent Network Solids are giant covalent substances like diamond, graphite and silicon dioxide (silicon(IV) oxide).
- Slides: 28