Covalent Immobilization of Minor Actinide Selective Ligands onto

Covalent Immobilization of Minor Actinide. Selective Ligands onto Magnetic Nanoparticles 9 September 2015 UK-Korea Nuclear Consortium Kick-Off Meeting Ashfaq Afsar Petr Distler, Chad Edwards, Andreas Geist, Laurence M. Harwood, Mark E. Hodson, Michael J. Hudson, Jan John, Dominic M. Laventine, Elizabeth J. Shaw, Clint Sharrad, James Westwood, Roger Whitehead. 27 November 2020 © University of Reading 2008 www. reading. ac. uk
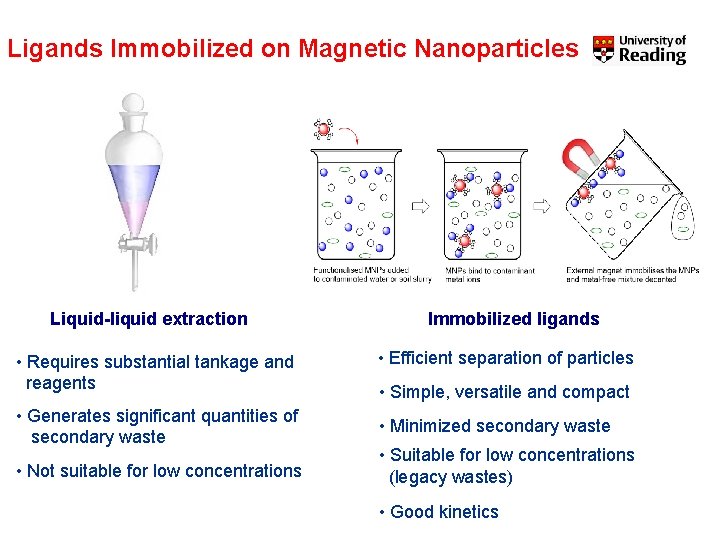
Ligands Immobilized on Magnetic Nanoparticles Liquid-liquid extraction • Requires substantial tankage and reagents • Generates significant quantities of secondary waste • Not suitable for low concentrations Immobilized ligands • Efficient separation of particles • Simple, versatile and compact • Minimized secondary waste • Suitable for low concentrations (legacy wastes) • Good kinetics

Ligands Immobilized on Magnetic Nanoparticles dispersed in water (colloidal solution). Nanoparticles extracted on application of magnetic field.
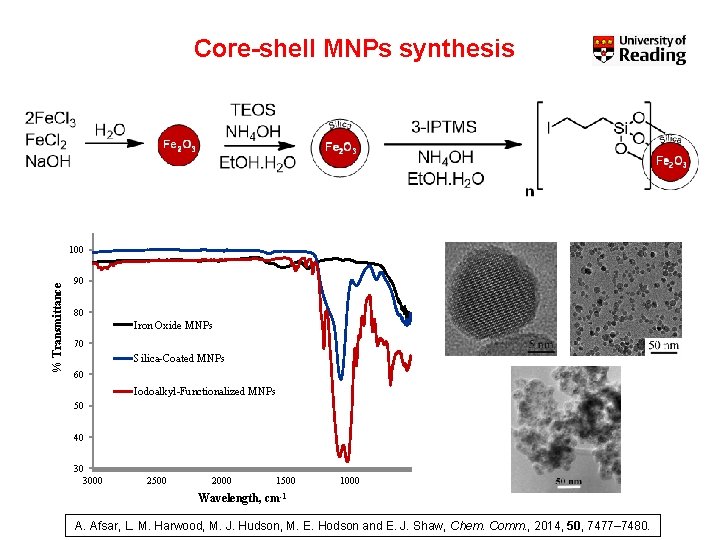
Core-shell MNPs synthesis % Transmittance 100 90 80 Iron Oxide MNPs 70 Silica-Coated MNPs 60 Iodoalkyl-Functionalized MNPs 50 40 30 3000 2500 2000 1500 1000 Wavelength, cm-1 A. Afsar, L. M. Harwood, M. J. Hudson, M. E. Hodson and E. J. Shaw, Chem. Comm. , 2014, 50, 7477– 7480.
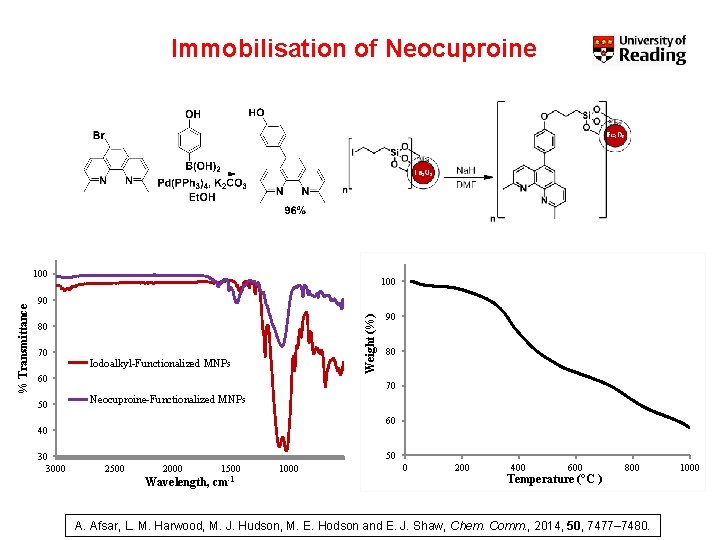
Immobilisation of Neocuproine 100 90 Weight (%) % Transmittance 100 80 70 Iodoalkyl-Functionalized MNPs 60 50 80 70 Neocuproine-Functionalized MNPs 60 40 30 3000 90 50 2500 2000 Wavelength, 1500 cm-1 1000 0 200 400 600 Temperature (°C ) 800 A. Afsar, L. M. Harwood, M. J. Hudson, M. E. Hodson and E. J. Shaw, Chem. Comm. , 2014, 50, 7477– 7480. 1000
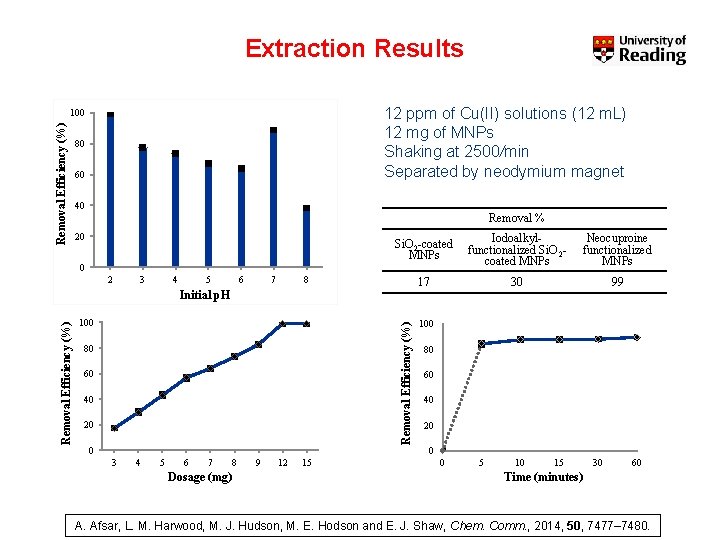
Extraction Results 12 ppm of Cu(II) solutions (12 m. L) 12 mg of MNPs Shaking at 2500/min Separated by neodymium magnet Removal Efficiency (%) 100 80 60 40 Removal % 20 Si. O 2 -coated MNPs Iodoalkylfunctionalized Si. O 2 coated MNPs Neocuproine functionalized MNPs 17 30 99 0 2 3 4 5 6 7 8 100 Removal Efficiency (%) Initial p. H 80 60 40 20 0 100 80 60 40 20 0 3 4 5 6 7 Dosage (mg) 8 9 12 15 0 5 10 15 30 60 Time (minutes) A. Afsar, L. M. Harwood, M. J. Hudson, M. E. Hodson and E. J. Shaw, Chem. Comm. , 2014, 50, 7477– 7480.
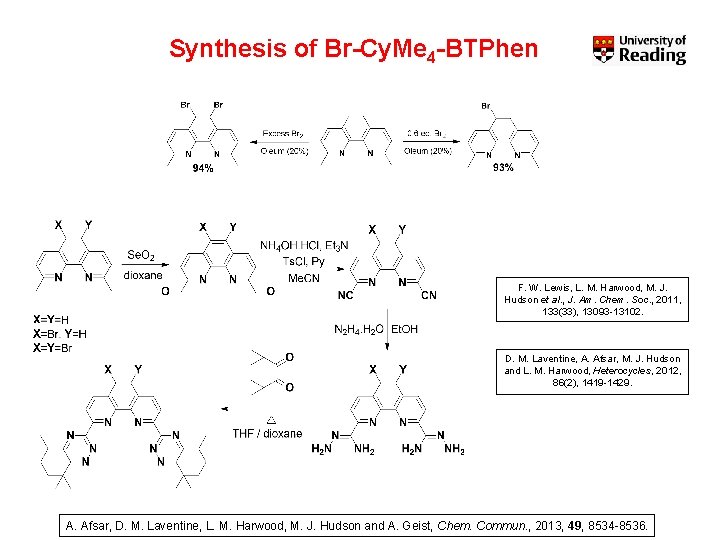
Synthesis of Br-Cy. Me 4 -BTPhen F. W. Lewis, L. M. Harwood, M. J. Hudson et al. , J. Am. Chem. Soc. , 2011, 133(33), 13093 -13102. D. M. Laventine, A. Afsar, M. J. Hudson and L. M. Harwood, Heterocycles, 2012, 86(2), 1419 -1429. A. Afsar, D. M. Laventine, L. M. Harwood, M. J. Hudson and A. Geist, Chem. Commun. , 2013, 49, 8534 -8536.
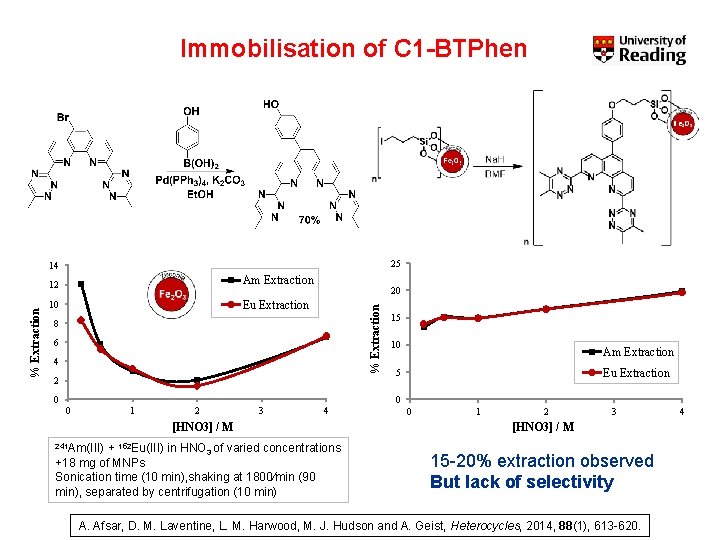
Immobilisation of C 1 -BTPhen 25 12 Am Extraction 10 Eu Extraction 20 % Extraction 14 8 6 4 2 0 15 10 Am Extraction Eu Extraction 5 0 0 1 2 3 4 [HNO 3] / M 241 Am(III) + 152 Eu(III) in HNO 3 of varied concentrations +18 mg of MNPs Sonication time (10 min), shaking at 1800/min (90 min), separated by centrifugation (10 min) 0 1 2 3 [HNO 3] / M 15 -20% extraction observed But lack of selectivity A. Afsar, D. M. Laventine, L. M. Harwood, M. J. Hudson and A. Geist, Heterocycles, 2014, 88(1), 613 -620. 4
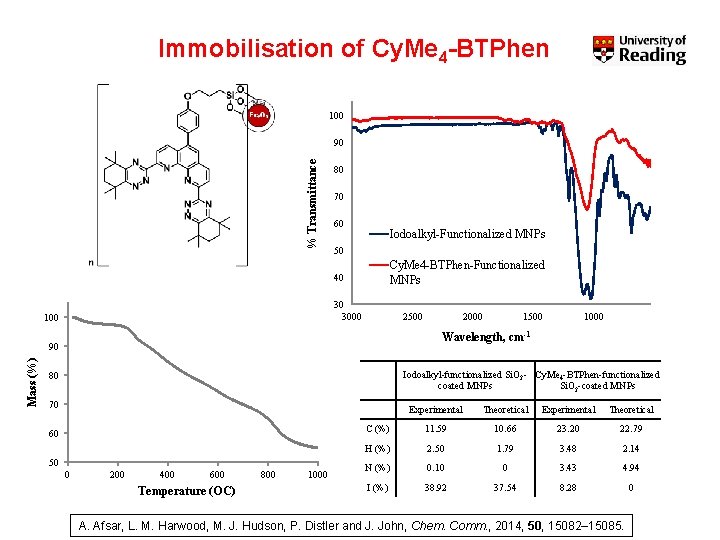
Immobilisation of Cy. Me 4 -BTPhen 100 % Transmittance 90 80 70 60 Iodoalkyl-Functionalized MNPs 50 Cy. Me 4 -BTPhen-Functionalized MNPs 40 30 3000 100 2500 1000 Wavelength, cm-1 90 Mass (%) 2000 Iodoalkyl-functionalized Si. O 2 - Cy. Me 4 -BTPhen-functionalized coated MNPs Si. O 2 -coated MNPs 80 70 60 50 0 200 400 600 Temperature (OC) 800 1000 Experimental Theoretical C (%) 11. 59 10. 66 23. 20 22. 79 H (%) 2. 50 1. 79 3. 48 2. 14 N (%) 0. 10 0 3. 43 4. 94 I (%) 38. 92 37. 54 8. 28 0 A. Afsar, L. M. Harwood, M. J. Hudson, P. Distler and J. John, Chem. Comm. , 2014, 50, 15082– 15085.
![Distribution ratio, log 10 Separation factor [HNO 3] / M Distribution ratio, log 10 Separation factor [HNO 3] / M](http://slidetodoc.com/presentation_image_h/b06c52e3b25135bc10a0280b15455159/image-10.jpg)
Distribution ratio, log 10 Separation factor [HNO 3] / M
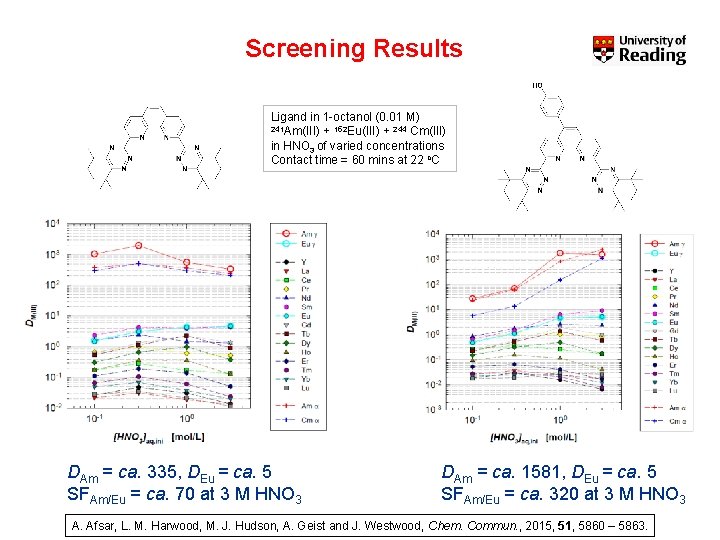
Screening Results Ligand in 1 -octanol (0. 01 M) 241 Am(III) + 152 Eu(III) + 244 Cm(III) in HNO 3 of varied concentrations Contact time = 60 mins at 22 o. C DAm = ca. 335, DEu = ca. 5 SFAm/Eu = ca. 70 at 3 M HNO 3 DAm = ca. 1581, DEu = ca. 5 SFAm/Eu = ca. 320 at 3 M HNO 3 A. Afsar, L. M. Harwood, M. J. Hudson, A. Geist and J. Westwood, Chem. Commun. , 2015, 51, 5860 – 5863.
![Separation factor Distribution ratio, log 10 Am(III)/Cm(III) Separation [HNO 3] / M Separation factor Distribution ratio, log 10 Am(III)/Cm(III) Separation [HNO 3] / M](http://slidetodoc.com/presentation_image_h/b06c52e3b25135bc10a0280b15455159/image-12.jpg)
Separation factor Distribution ratio, log 10 Am(III)/Cm(III) Separation [HNO 3] / M
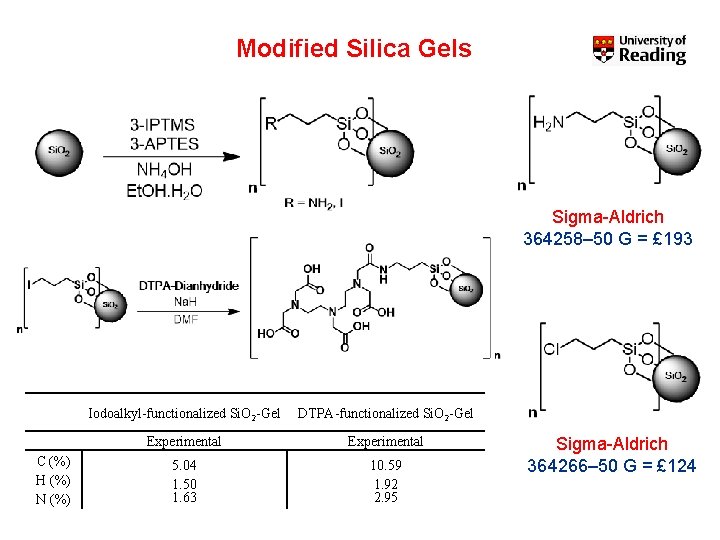
Modified Silica Gels Sigma-Aldrich 364258– 50 G = £ 193 C (%) H (%) N (%) Iodoalkyl-functionalized Si. O 2 -Gel DTPA-functionalized Si. O 2 -Gel Experimental 5. 04 1. 50 1. 63 10. 59 1. 92 2. 95 Sigma-Aldrich 364266– 50 G = £ 124
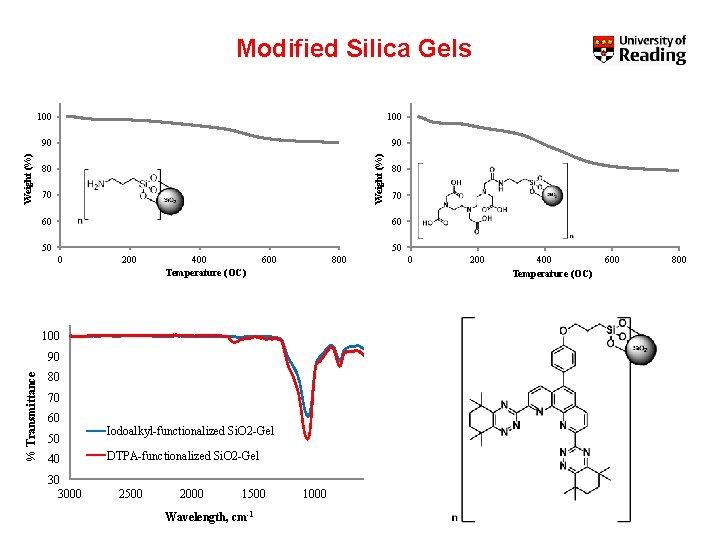
Modified Silica Gels 90 90 Weight (%) 100 80 70 60 50 50 0 200 400 Temperature (OC) 600 800 100 90 % Transmittance 80 80 70 60 50 40 30 3000 Iodoalkyl-functionalized Si. O 2 -Gel DTPA-functionalized Si. O 2 -Gel 2500 2000 1500 Wavelength, cm-1 1000 0 200 400 Temperature (OC) 600 800

Achievements and Conclusions Iron oxide (Fe 2 O 3) MNPs coated with silica and functionalized with Cy. Me 4 BTPhen have the ability to extract various metal ions from aqueous solution probably as 1: 1 (metal: ligand complexes), leading to a totally new extraction profile compared to liquid-liquid extraction systems. Adding substituents to the phenanthroline backbone at the 5 - and 5, 6 positions of BTPhens, enable these ligands to be fine-tuned in order to enhance the selectivity of Am(III) from Ln(III) and Cm(III). This has opened up a whole new range of extraction possibilities leading to very high Am 3+ – Eu 3+ and workable Am 3+ – Cm 3+ separations that are dependent upon nitric acid concentration.

Acknowledgments Chemistry group Soil science group Extraction studies group Laurence Harwood Michael Hudson Ashfaq Afsar Joe Cowell James Westwood Mark Hodson Liz Shaw David Hughes Andreas Geist (KIT-INE Karlsruhe) Jan John (CTU Prague) Petr Distler(CTU Prague) UCLan Gary Bond Harry Eccles Supporting staff Peter Harris (TEM) Peter Health (NMR) Martin Reeves (MS) Ogla Khutoryanskaya (TGA) Michael Andrews (XRD) Anne Dudley (AA)

OK dragon! Come out of that cave and fight!!
- Slides: 17