Covalent Compounds Section 6 1 Covalent Bond Shared

Covalent Compounds Section 6. 1

Covalent Bond • Shared electrons – Part of BOTH atoms’ valence shells • Occurs if BOTH atoms NEED valence electrons • Between NONMETALS
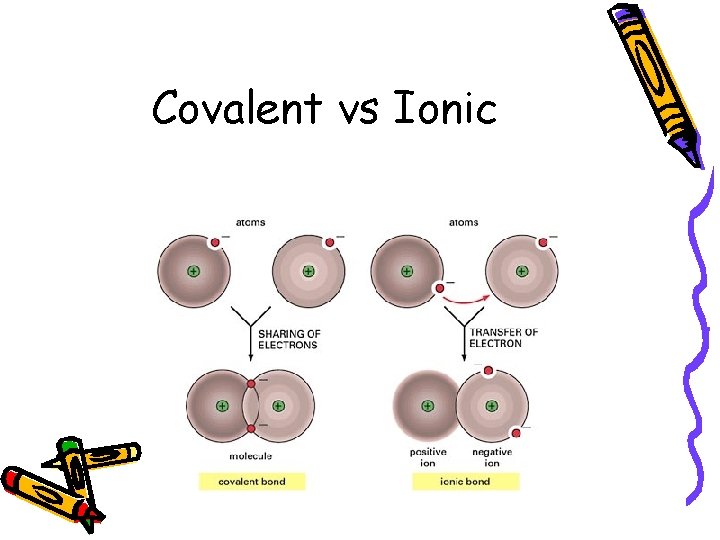
Covalent vs Ionic

Molecule • Formed when two or more atoms bond covalently

Diatomic Molecules • 2 atoms of the same element form a bond • Examples: – H 2 – O 2 – N 2 – Halogens: F 2, Cl 2, Br 2, I 2
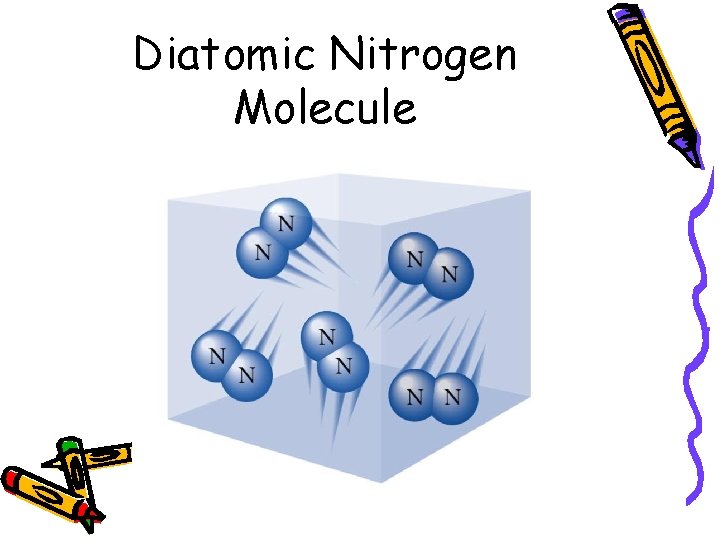
Diatomic Nitrogen Molecule

Binary Molecules • Contains only two different nonmetals • Do not contain metals or ions

3 Rules for Naming Binary Molecules 1) For the 1 st element in formula, use its name from periodic table 2) For the 2 nd element, use its root and the suffix –ide. 3) Give each element a prefix to represent its subscript

Prefixes • • • 1 – mono 2 – di 3 – tri 4 – tetra 5 – penta-

Prefixes • • • 6 – hexa 7 – hepta 8 – octa 9 – nona 10 – deca-

Prefixes • • • 11 – hendeca 12 – dodeca 13 – triskaideca 14 – tetradeca 15 – pentadeca-

Prefixes • • • 16 – hexadeca 17 – heptadeca 18 – octadeca 19 – ennedeca 20 – icosa-

Exception • Never use “mono” with first element – Ex: CO • Incorrect: Monocarbon monooxide. • Correct: Carbon monoxide.
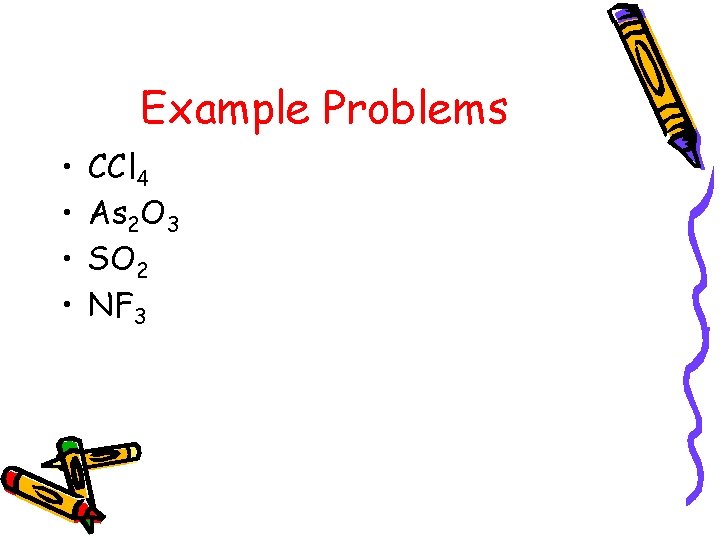
Example Problems • • CCl 4 As 2 O 3 SO 2 NF 3

2 Rules for Writing Formulas 1) For each name, write the element’s symbol 2) Assign each symbol a subscript using the prefixes (DO NOT REDUCE!!!)

Example Problems • • Carbon monofluoride Sulfur difluoride Disilicon tetraoxide Hexacarbon decahydride Hendecabromine icosachloride Octanitrogen dodecaiodide Triskaidecabromine hexadecasulfide

Common Names • Many binary compounds were discovered before the modern naming system

- Slides: 18