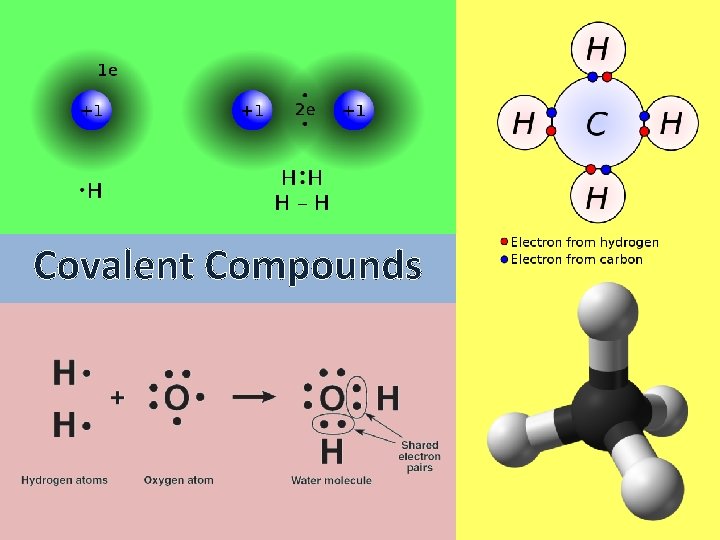
Covalent Compounds Nonmetals Chemical reactions occur when atoms










- Slides: 27

Covalent Compounds

Nonmetals Chemical reactions occur when atoms gain, lose, or share electrons. Sharing electrons is what creates covalent bonds! Nonmetals can _______ share electrons to form a covalent bond. molecule This creates a ______.

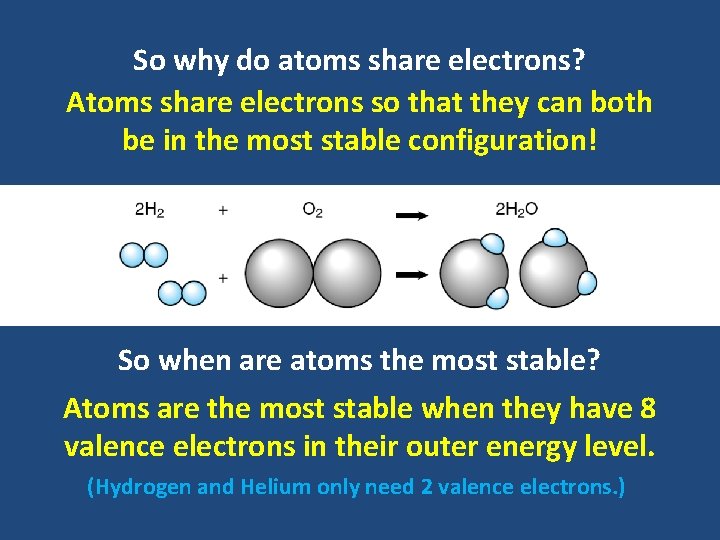
So why do atoms share electrons? Atoms share electrons so that they can both be in the most stable configuration! So when are atoms the most stable? Atoms are the most stable when they have 8 valence electrons in their outer energy level. (Hydrogen and Helium only need 2 valence electrons. )

Which group on the periodic table already has this stable configuration? Group 18 The Noble Gases They are inert gases. What does it mean to be inert? Inertia is a resistance to change. Inert substances do not want to change which means that they are very unreactive.

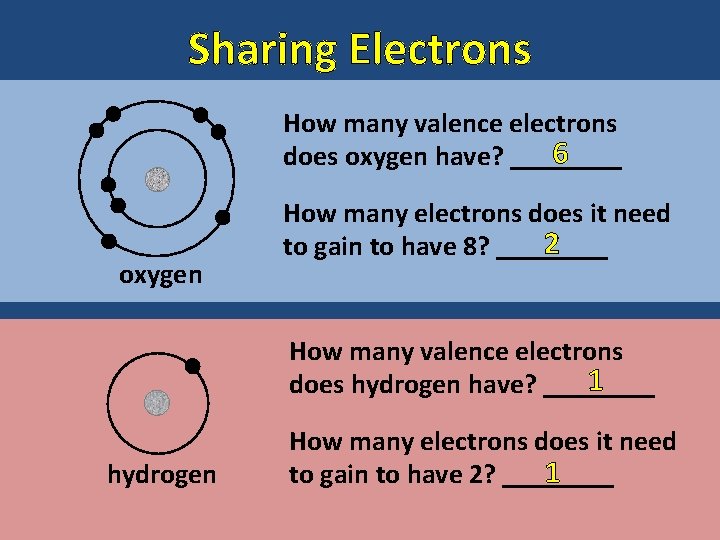
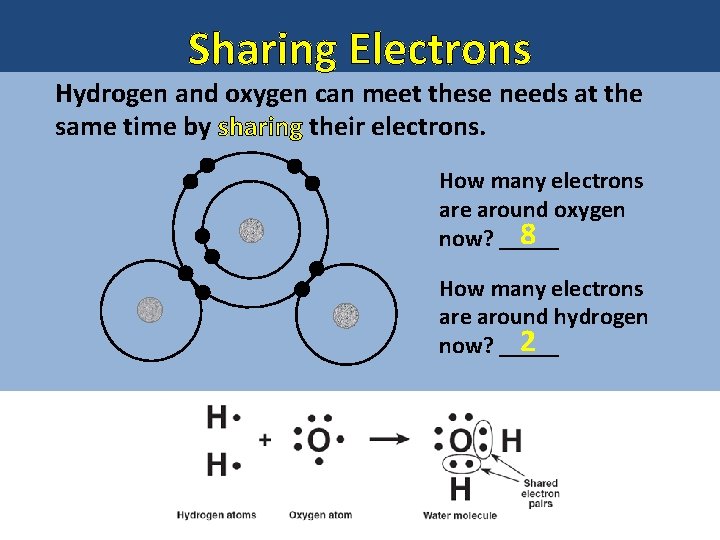
Sharing Electrons How many valence electrons 6 does oxygen have? ____ oxygen How many electrons does it need 2 to gain to have 8? ____ How many valence electrons 1 does hydrogen have? ____ hydrogen How many electrons does it need 1 to gain to have 2? ____

Sharing Electrons Hydrogen and oxygen can meet these needs at the same time by sharing their electrons. How many electrons are around oxygen 8 now? _____ How many electrons are around hydrogen 2 now? _____

If the substance is a covalent compound, the smallest particle will be a MOLECULE Water Molecule

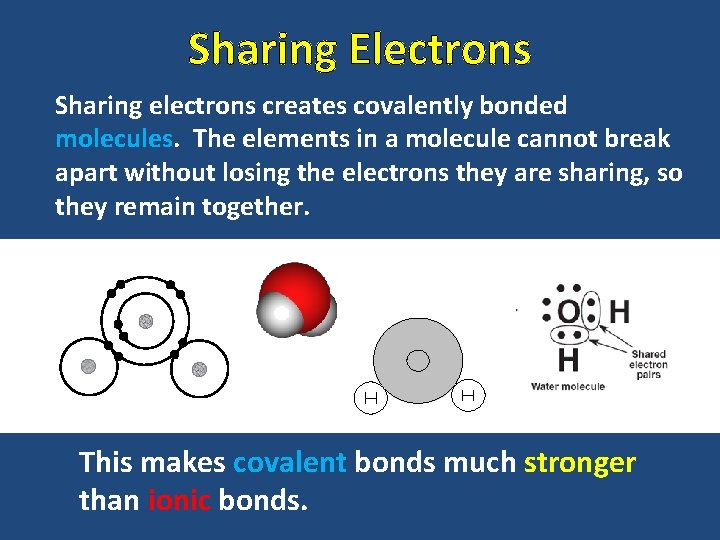
Sharing Electrons Sharing electrons creates covalently bonded molecules. The elements in a molecule cannot break apart without losing the electrons they are sharing, so they remain together. This makes covalent bonds much stronger than ionic bonds.

Deciding if a compound is ionic or covalent is easy. What elements do ionic compounds contain? Ionic compounds contain a metal and a nonmetal. What elements do covalent compounds contain? Covalent compounds contain only nonmetals.

Decide whether the compounds on your notes are ionic or covalent. I I C I C C I

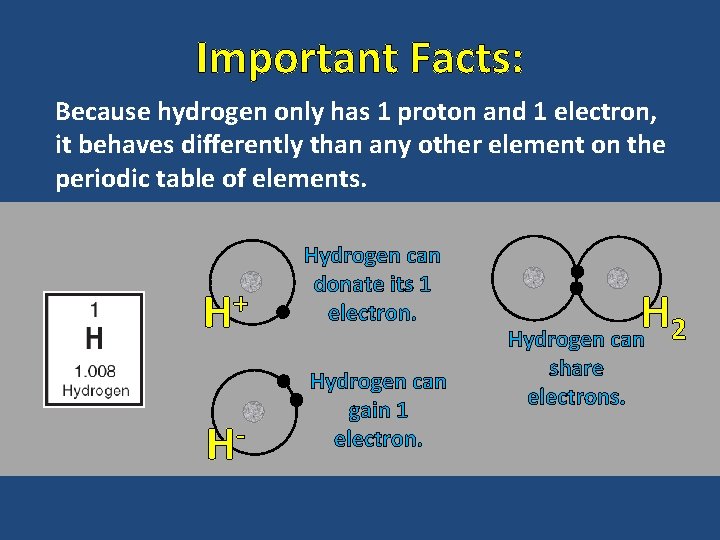
Important Facts: Because hydrogen only has 1 proton and 1 electron, it behaves differently than any other element on the periodic table of elements. H+ H- Hydrogen can donate its 1 electron. Hydrogen can gain 1 electron. H Hydrogen can 2 share electrons.

We can use Lewis structures to draw covalent compounds. We will do this often. H 2 H Notice that there are ____ electrons in a covalent bond. We will replace these electrons with a line to make things less cluttered. FF Electrons that are not involved in a covalent bond are placed around the element. These are called LONE PAIR ELECTRONS.

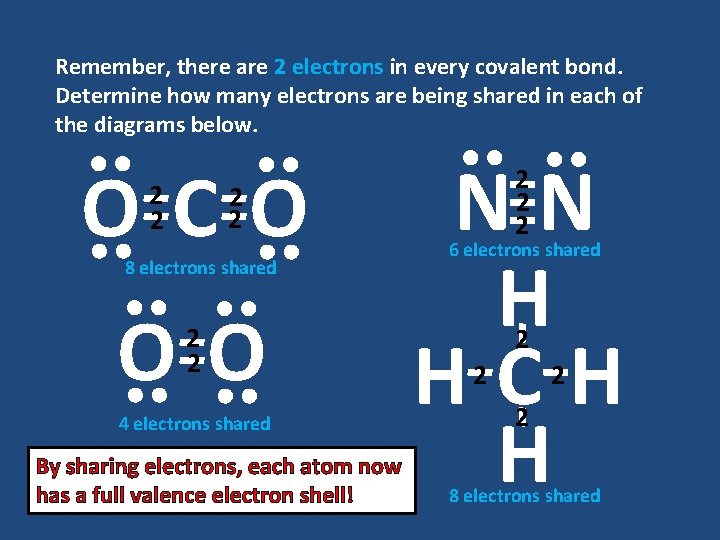
Remember, there are 2 electrons in every covalent bond. Determine how many electrons are being shared in each of the diagrams below. OCO 2 2 8 electrons shared OO 2 2 4 electrons shared By sharing electrons, each atom now has a full valence electron shell! NN H HCH H 2 2 2 6 electrons shared 2 2 8 electrons shared

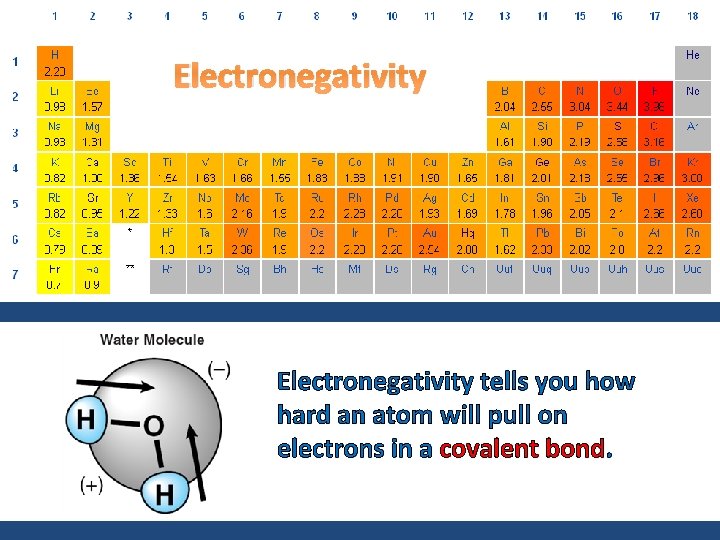
Electronegativity tells you how hard an atom will pull on electrons in a covalent bond.

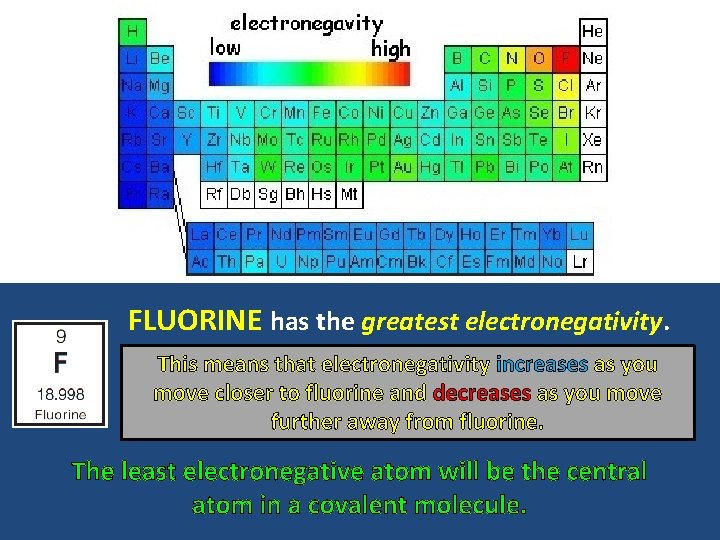
FLUORINE has the greatest electronegativity. This means that electronegativity increases as you move closer to fluorine and decreases as you move further away from fluorine. The least electronegative atom will be the central atom in a covalent molecule.

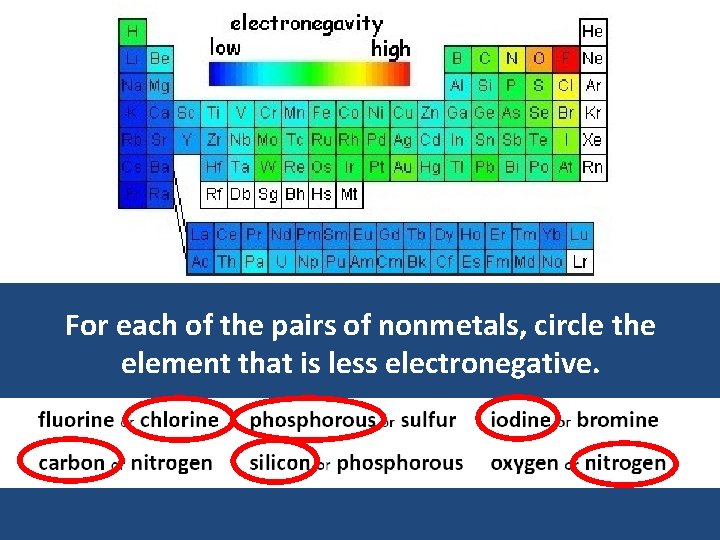
For each of the pairs of nonmetals, circle the element that is less electronegative.

Creating a Lewis Dot Structure for a Covalent Compound Step 1: Add together the total number of valence electrons for each of the elements. Example: NH 3 = 5 + 3(1) = 8 e CBr 4 = 4 + 4(7) = 32 e

Creating a Lewis Dot Structure for a Covalent Compound Step 2: Write out the elements so that the least electronegative element is in the middle of the compound. (H is NEVER the central atom. ) Example: H N H H Br Br C Br Br

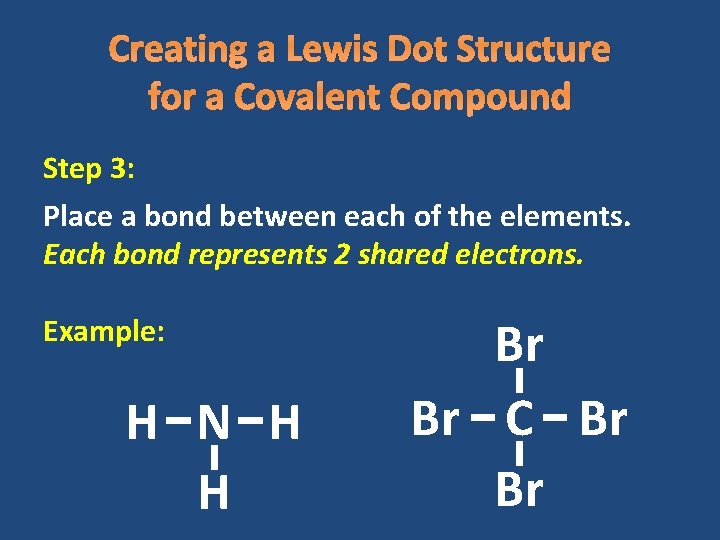
Creating a Lewis Dot Structure for a Covalent Compound Step 3: Place a bond between each of the elements. Each bond represents 2 shared electrons. Example: H N H H Br Br C Br Br

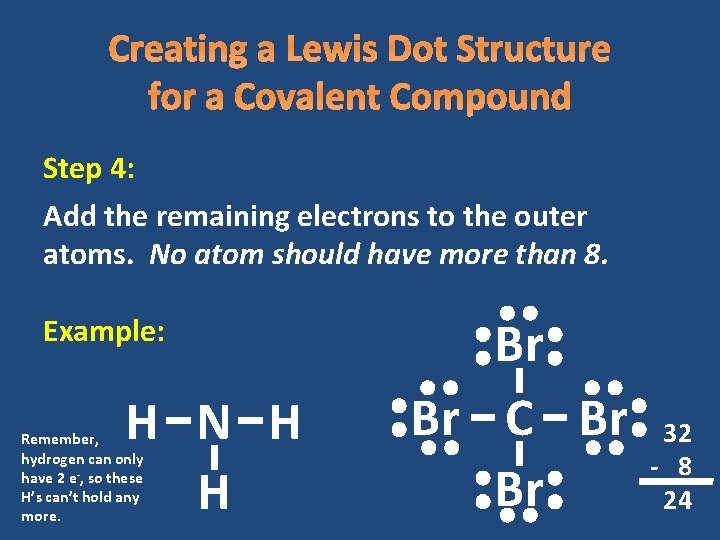
Creating a Lewis Dot Structure for a Covalent Compound Step 4: Add the remaining electrons to the outer atoms. No atom should have more than 8. Example: H N H H Remember, hydrogen can only have 2 e-, so these H’s can’t hold any more. Br Br C Br Br 32 - 8 24

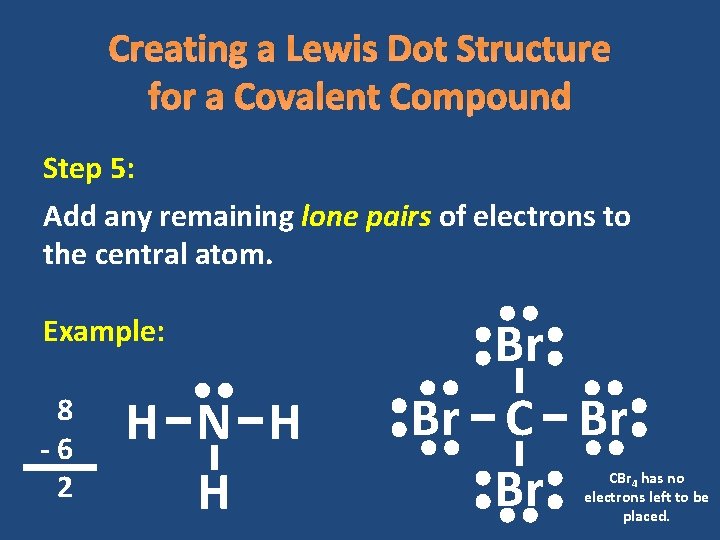
Creating a Lewis Dot Structure for a Covalent Compound Step 5: Add any remaining lone pairs of electrons to the central atom. Example: 8 -6 2 H N H H Br Br CBr 4 has no electrons left to be placed.

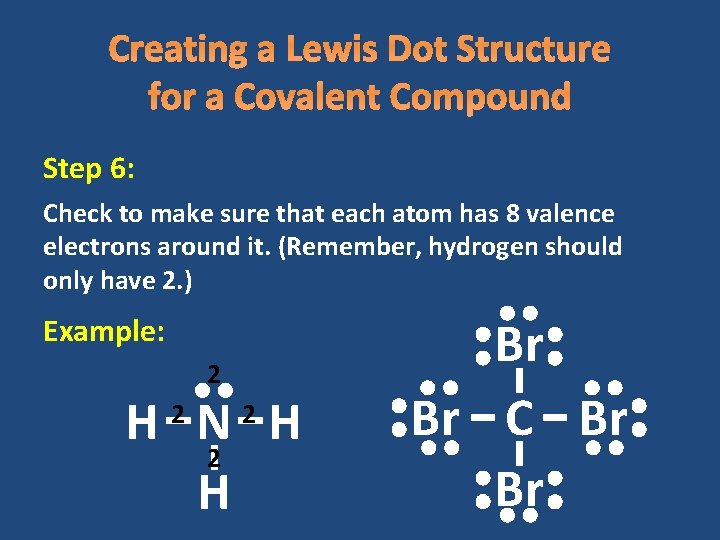
Creating a Lewis Dot Structure for a Covalent Compound Step 6: Check to make sure that each atom has 8 valence electrons around it. (Remember, hydrogen should only have 2. ) Example: 2 H N H 2 2 Br Br C Br Br

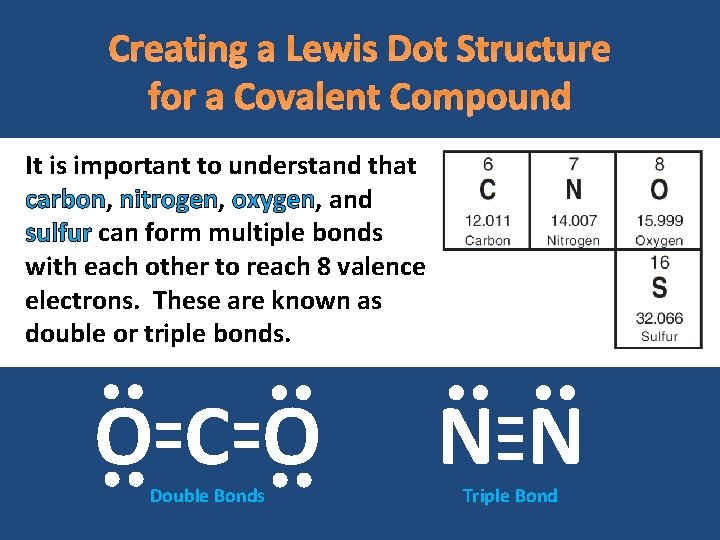
Creating a Lewis Dot Structure for a Covalent Compound It is important to understand that carbon, nitrogen, oxygen, and sulfur can form multiple bonds with each other to reach 8 valence electrons. These are known as double or triple bonds. OCO Double Bonds NN Triple Bond

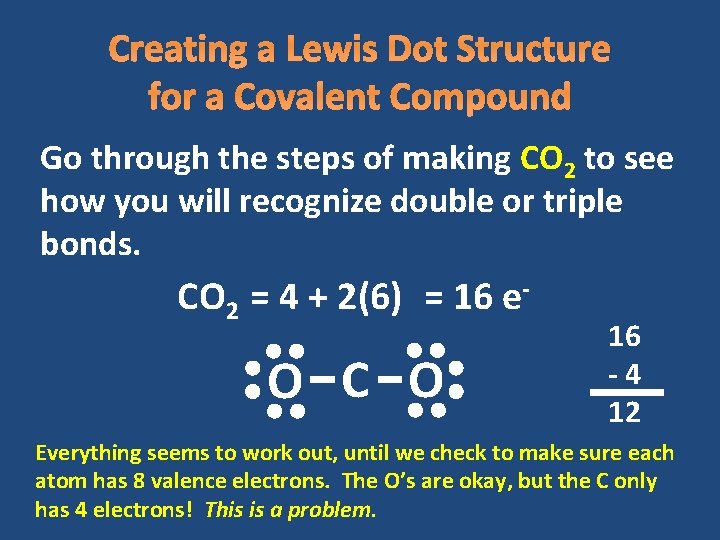
Creating a Lewis Dot Structure for a Covalent Compound Go through the steps of making CO 2 to see how you will recognize double or triple bonds. CO 2 = 4 + 2(6) = 16 e- O C O 16 -4 12 Everything seems to work out, until we check to make sure each atom has 8 valence electrons. The O’s are okay, but the C only has 4 electrons! This is a problem.

Creating a Lewis Dot Structure for a Covalent Compound To fix this problem, we remove a pair of electrons from around each oxygen and add two more bonds. CO 2 = 4 + 2(6) = 16 e- O C O Now EVERY atom in the compound has 8 valence electrons. We use double and triple bonds to make this happen in compounds with C, N, O and S.

Creating a Lewis Dot Structure for a Covalent Compound There are two major exceptions to the rule of 8 valence electrons that you need to know! Boron often creates compounds so that it only has 6 valence electrons! Phosphorous often creates compounds so that is has 10 valence electrons!

Creating a Lewis Dot Structure for a Covalent Compound These compounds are made following the same set of rules; you just have to realize that you will not end up with 8 electrons around these elements! BH 3 = 3 + 3(1) = 6 e. There are no more electrons to add. Boron is simply left with 6 valence electrons. H B H H 6 - 6 0