Covalent Compounds Covalent Compounds Covalent bond atoms share

Covalent Compounds

Covalent Compounds Covalent bond: atoms share electrons so each has a full outer shell of 8 electrons Binary Covalent Compound: compound made from two non-metals that share electrons Non metal Covalent compound

Covalent Prefixes 1 = mono 2 = di 3 = tri 4 = tetra 5 = penta 6 = hexa 7 = hepta 8 = octa 9 = nona 10 = deca- Two notes: • No “mono” on the first element name • If oxygen/oxide: do NOT include the final “o” or “a” on the prefix

Covalent Compounds Formula to Name

Covalent Compounds Formula to Name • Formulas will have… • 2 non-metal elements • To name: 1. Name of the first element with the prefix indicating the number of atoms 2. Name of the second element with the prefix indicating the number of atoms and the suffix “ide”

Examples P 2 O 5 Si. O 2

Write the name for the following compounds Practice 1. CO 2 carbon dioxide 2. N 2 O 4 dinitrogen tetroxide 3. P 4 O 10 tetraphosphorus decoxide 4. CO carbon monoxide

Covalent Compounds Name to Formula

Covalent Compounds Name to Formula • Names will have… – covalent prefixes before the element name • To write: 1. Write the symbols of the first and second element 2. Use the covalent prefixes to determine the subscripts of each element (assume the first element is “ 1” if there is no prefix). Atoms do not form ions when bonding covalently…you DO NOT need to worry about charges with this type!

Example Dinitrogen Tetroxide

Example

Write the formula for the following compounds Practice 1. carbon monoxide CO 2. nitrogen dioxide NO 2 3. sulfur tetrachloride SCl 4

A Word of Caution… “di” and “bi” do not mean the same thing! di- bi- Stands for “ 2” in covalent compounds Means there’s a hydrogen in the polyatomic anion Carbon dioxide = CO 2 Sodium biphosphate = Na 2 HPO 4


Let’s Practice Example: Write the name for the following compounds CO 2 Carbon dioxide N 2 O 4 Dinitrogen tetraoxide P 4 O 10 Tetraphosphorus decaoxide CO Carbon monoxide
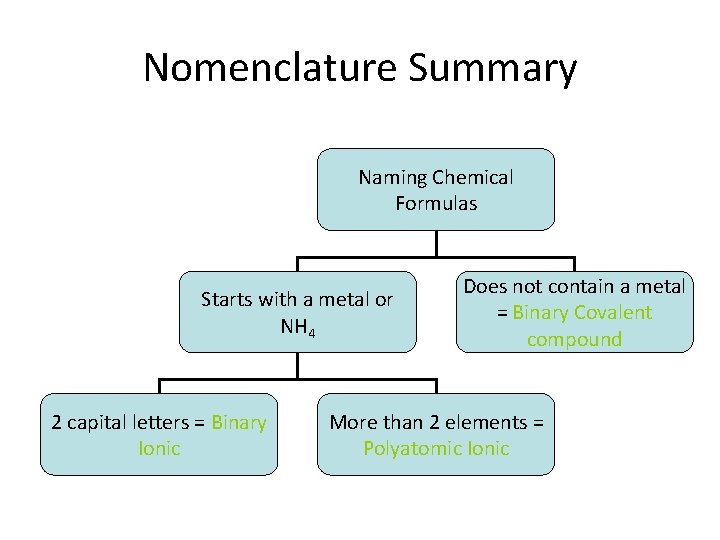
Nomenclature Summary Naming Chemical Formulas Starts with a metal or NH 4 2 capital letters = Binary Ionic Does not contain a metal = Binary Covalent compound More than 2 elements = Polyatomic Ionic

Mixed Practice – Ionic and Covalent Na 2 O Example: Write the name for the following compounds K 3 PO 4 Cu(OH)2 (NH 4)2 S Mg. Cl 2
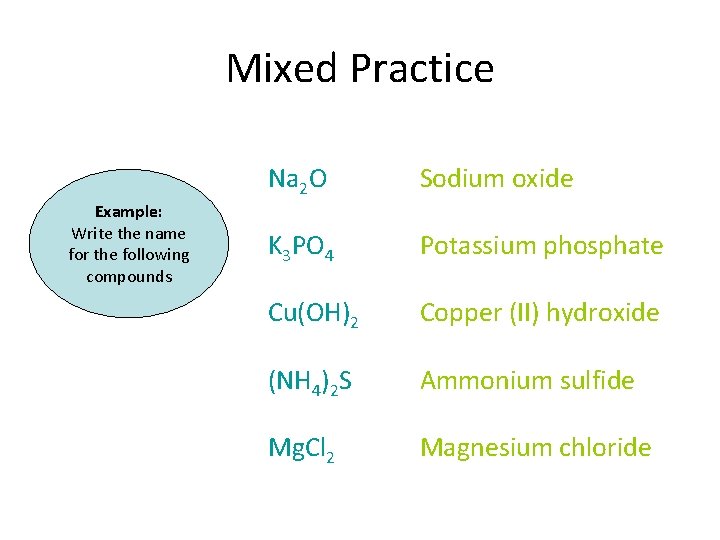
Mixed Practice Example: Write the name for the following compounds Na 2 O Sodium oxide K 3 PO 4 Potassium phosphate Cu(OH)2 Copper (II) hydroxide (NH 4)2 S Ammonium sulfide Mg. Cl 2 Magnesium chloride
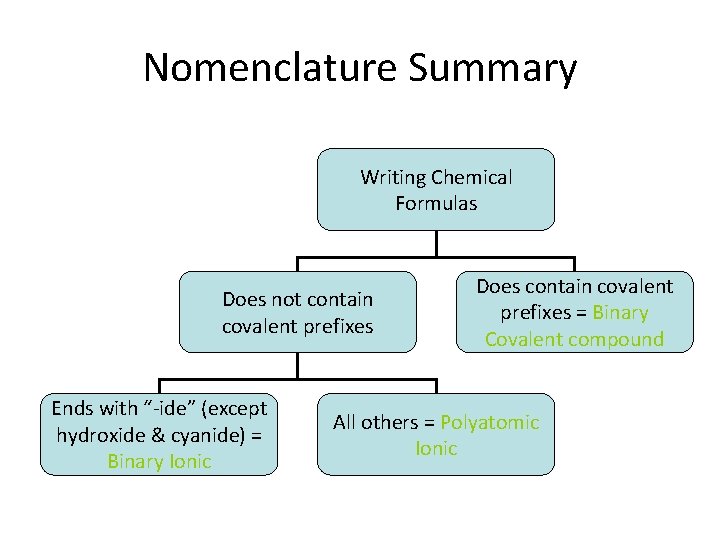
Nomenclature Summary Writing Chemical Formulas Does not contain covalent prefixes Ends with “-ide” (except hydroxide & cyanide) = Binary Ionic Does contain covalent prefixes = Binary Covalent compound All others = Polyatomic Ionic

Mixed Practice Example: Write the following chemical formulas Magnesium hydroxide Copper (II) nitrate Iron (III) oxide Nitrogen dioxide Sodium bicarbonate

Mixed Practice Example: Write the following chemical formulas Magnesium hydroxide Mg(OH)2 Copper (II) nitrate Cu(NO 3)2 Iron (III) oxide Fe 2 O 3 Nitrogen dioxide NO 2 Sodium bicarbonate Na. HCO 3
- Slides: 21