Covalent Compounds Covalent bond bond that results between


Covalent Compounds • Covalent bond: bond that results between nonmetallic elements that share electrons. • Molecule: compound formed when 2 or more elements are covalently bonded. • Bonds can be single, double, or triple • Single bond = 2 electrons shared • Double bond = 4 electrons shared • Triple bond = 6 electrons shared
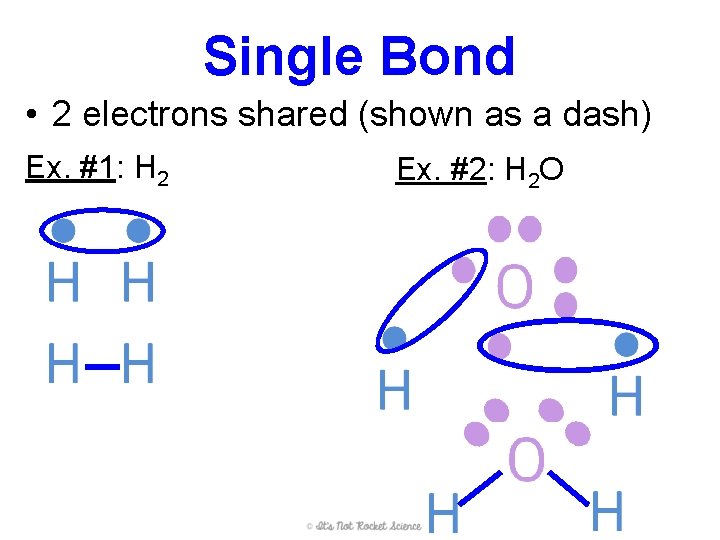
Single Bond • 2 electrons shared (shown as a dash) Ex. #1: H 2 Ex. #2: H 2 O
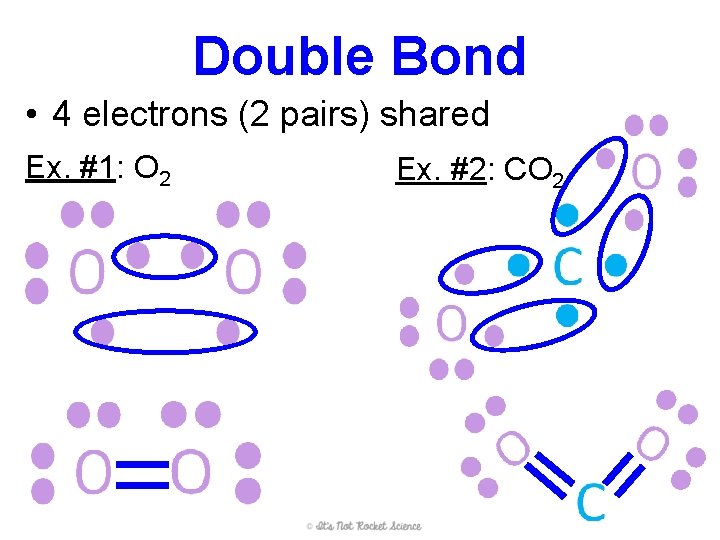
Double Bond • 4 electrons (2 pairs) shared Ex. #1: O 2 Ex. #2: CO 2
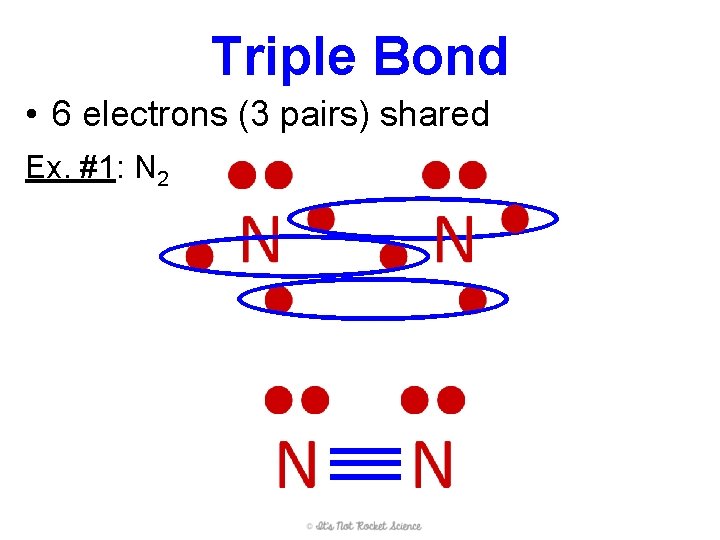
Triple Bond • 6 electrons (3 pairs) shared Ex. #1: N 2

Properties of Covalent Compounds • Made up of nonmetals sharing e • Molecules that can be solid, liquid, or gas • Low melting and boiling points • Cannot conduct electricity when dissolved in water

Properties of Ionic Compounds • Made up of (+) metallic ions and (-) nonmetallic ions after metal transferred eto nonmetal • Arranged in regular repeating patterns called crystals, therefore tend to be solids • High melting and boiling points • Can conduct electricity when dissolved in water

How to determine the NAME for a Covalent Compound Formula Name 1. Name first element with prefix to indicate how many. Exception: don’t use mono for “ 1” for 1 st element 2. Name second element with prefix to indicate how many AND add an –ide ending.

Prefixes to Know: mono – 1 di – 2 tri – 3 tetra – 4 penta – 5 hexa – 6 hepta – 7 octa – 8 nona – 9 deca – 10 Example: Name the compound P 2 O 5 diphosphorous pentoxide

Practice Time! Write the name of the covalent compounds based on the chemical formulas below. 1. 2. 3. 4. 5. CO 2 H 2 O CH 4 PCl 3 P 4 O 10 Carbon dioxide Dihydrogen monoxide Carbon tetrahydride Phosphorous trichloride Tetraphosphorous decaoxide

How to determine the Formula for a Covalent Compound Name Formula 1. Use prefixes to determine subscripts. NO CRISS CROSSES OR SIMPLIFYING IN COVALENT!!! Example: What is the formula for dichlorine monoxide? Cl 2 O

Practice Time! Determine the chemical formula of the covalent compounds based on the names below. 1. 2. 3. 4. 5. Dicarbon tetraoxide Hydrogen monosulfide Pentaphosphorous trinitride Sulfur pentoxide Silicon tetrafluoride C 2 O 4 HS P 5 N 3 SO 5 Si. F 4

- Slides: 13