Covalent Bonds Sharing Electrons Atoms unit of a

Covalent Bonds Sharing Electrons
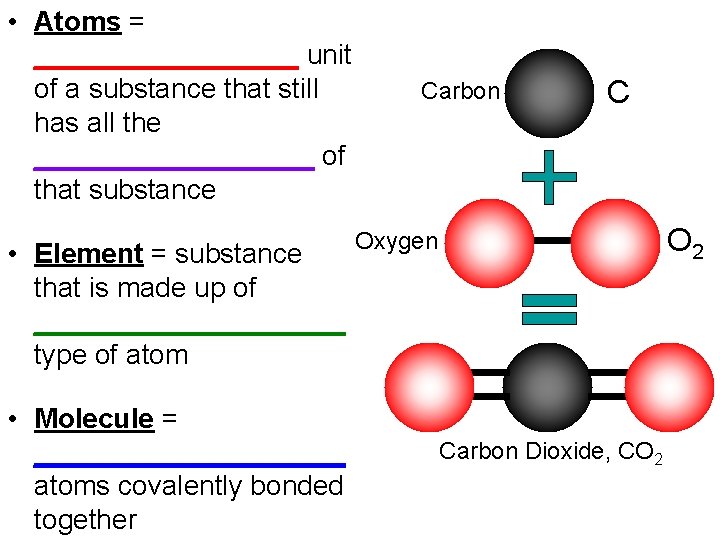
• Atoms = _________ unit of a substance that still has all the _________ of that substance Carbon C O 2 Oxygen • Element = substance that is made up of __________ type of atom • Molecule = __________ atoms covalently bonded together Carbon Dioxide, CO 2
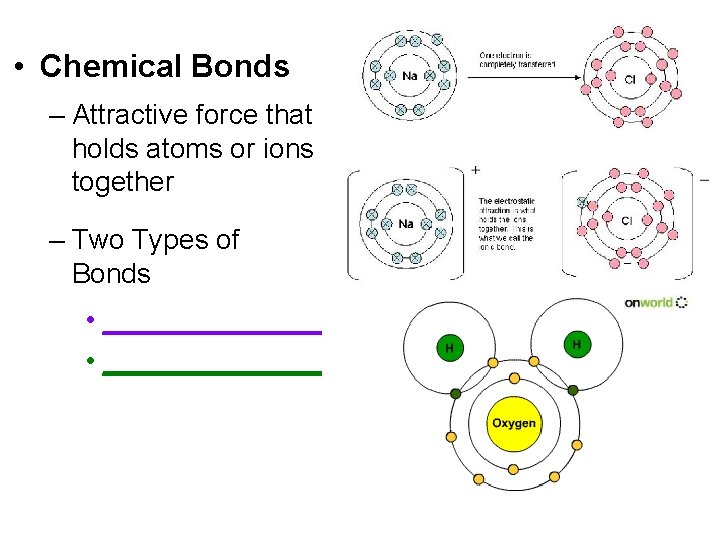
• Chemical Bonds – Attractive force that holds atoms or ions together – Two Types of Bonds • ______________

• Covalent Bond – bond formed when atoms ________ one or more pairs of electrons. – covalent compounds can be ________________
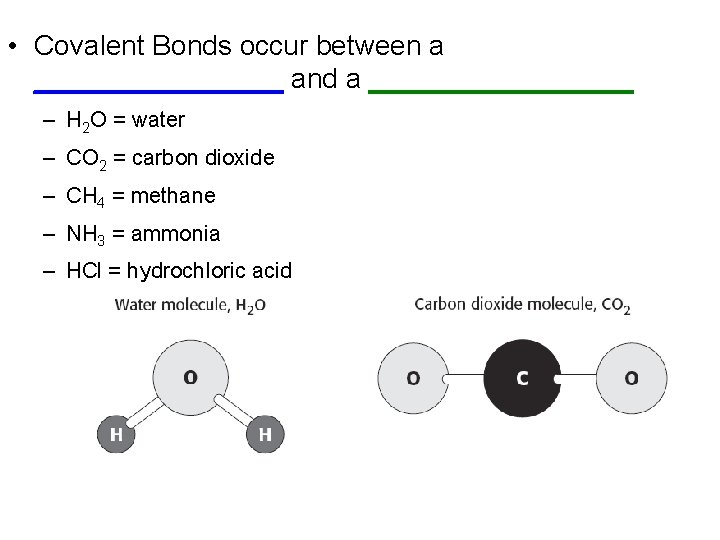
• Covalent Bonds occur between a ________ and a _________ – H 2 O = water – CO 2 = carbon dioxide – CH 4 = methane – NH 3 = ammonia – HCl = hydrochloric acid
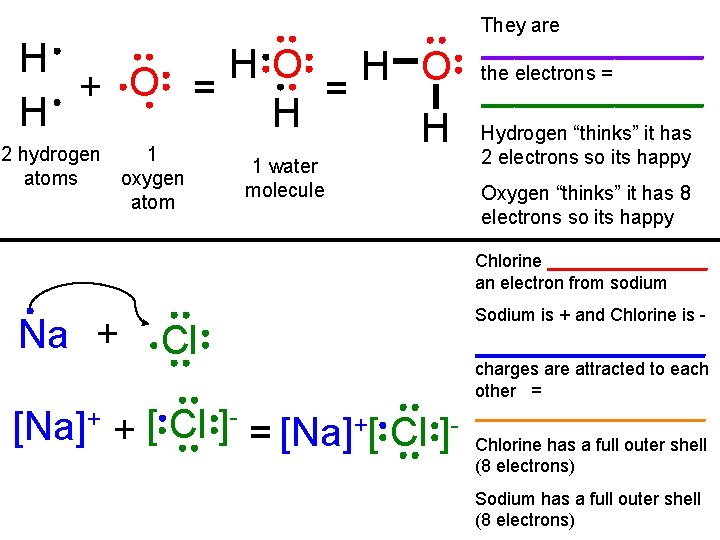
H H O + O = = H H H 2 hydrogen 1 atoms oxygen atom 1 water molecule They are __________ the electrons = __________ Hydrogen “thinks” it has 2 electrons so its happy Oxygen “thinks” it has 8 electrons so its happy Chlorine ________ an electron from sodium Na + Cl [Na]+ + [ Cl ]- = [Na]+[ Cl ]- Sodium is + and Chlorine is ____________ charges are attracted to each other = ____________ Chlorine has a full outer shell (8 electrons) Sodium has a full outer shell (8 electrons)
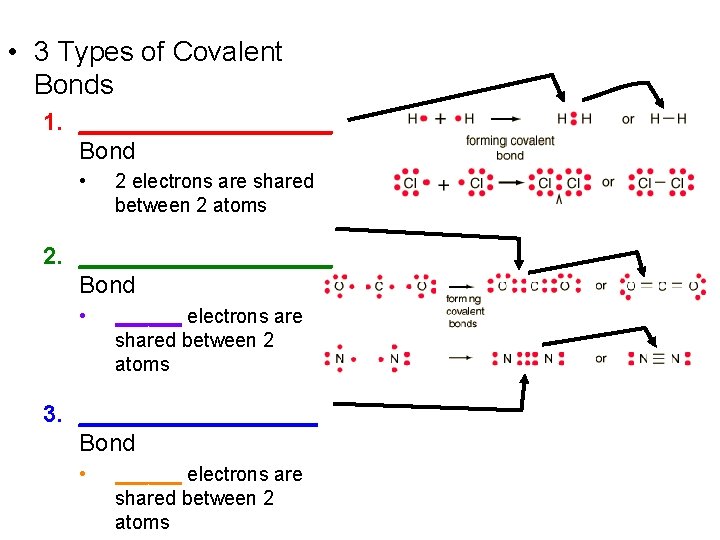
• 3 Types of Covalent Bonds 1. __________ Bond • 2 electrons are shared between 2 atoms 2. __________ Bond • ______ electrons are shared between 2 atoms 3. _________ Bond • ______ electrons are shared between 2 atoms

- Slides: 8