Covalent Bonds Review Reactivity Electron configuration Stability Chemical

Covalent Bonds

Review • Reactivity – Electron configuration – Stability • Chemical bond • Lewis dot symbol

Covalent bond • Bond in which two valence electrons are shared by two atoms – Occurs with elements close to each other on the periodic table – Ex: H 2 O, NH 3 (ammonia), CH 4 (methane) – Between a nonmetal and a nonmetal

3 Types of Covalent Bonds • Single Covalent Bond • Double Covalent Bond • Triple Covalent Bond

3 Pencil Demo for Single, Double and Triple Covent bonds • Make as many observations as you can i. e. facial expressions, body position, muscles used, noises made, force or energy exerted, etc.
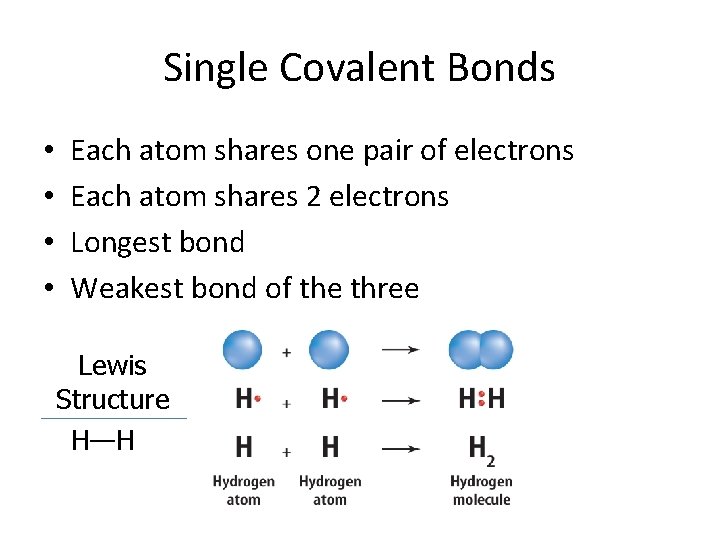
Single Covalent Bonds • • Each atom shares one pair of electrons Each atom shares 2 electrons Longest bond Weakest bond of the three Lewis Structure H—H
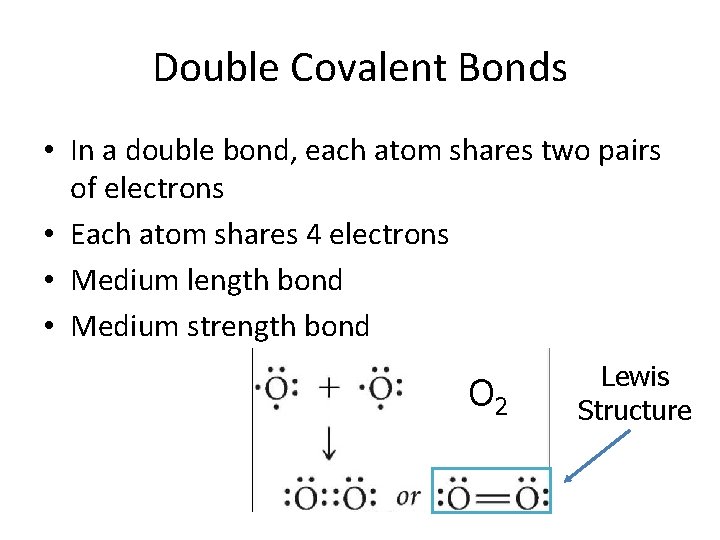
Double Covalent Bonds • In a double bond, each atom shares two pairs of electrons • Each atom shares 4 electrons • Medium length bond • Medium strength bond O 2 Lewis Structure

Triple Covalent Bond • In a triple covalent bond, each atom shares three pairs of electrons • Each atom shares 6 electrons • The shortest bond • The strongest bond • Carbon, nitrogen, oxygen, and sulfur can form double and triple covalent bonds Lewis N 2 Structure

Strength of Covalent Bond • Several factors control bond strength —Number of shared electrons-the more electrons shared, greater the bond —Size of the atom —Bond length – the greater the bond length, the weaker the bond
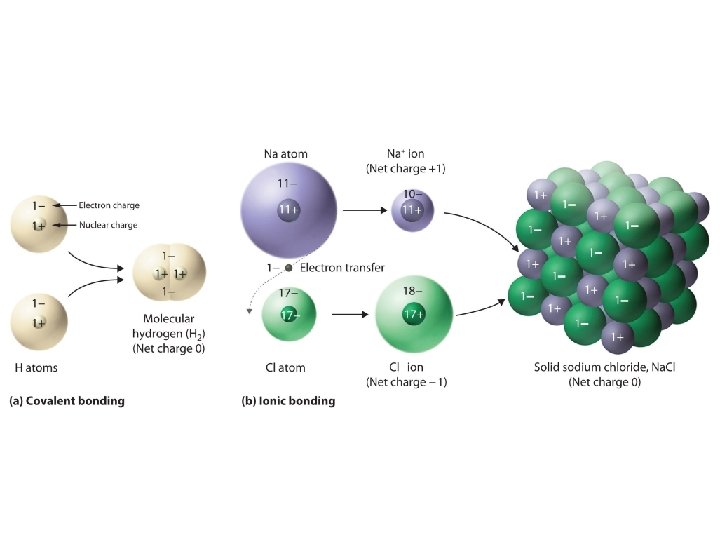

Substance Molecule Compound • Two or more atoms chemically bonded together • Two or more elements chemically bonded together
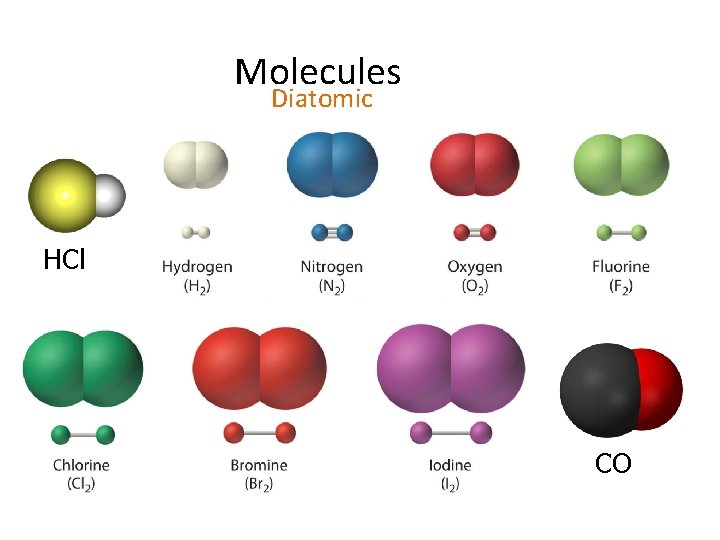
Molecules Diatomic HCl CO

Memeorize Diatomic Elements – elements that exist as two H 2, O 2, N 2, Cl 2, Br 2, I 2, F 2 – HONCl. Br. IF – Magnificent 7 -don’t forget H
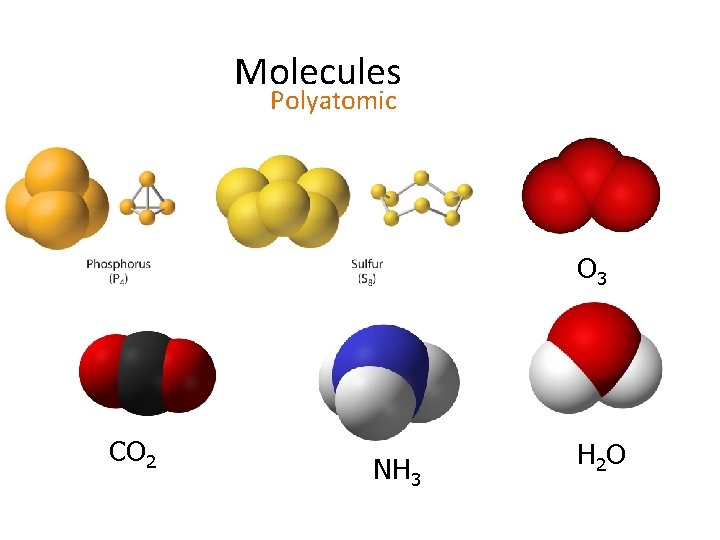
Molecules Polyatomic O 3 CO 2 NH 3 H 2 O
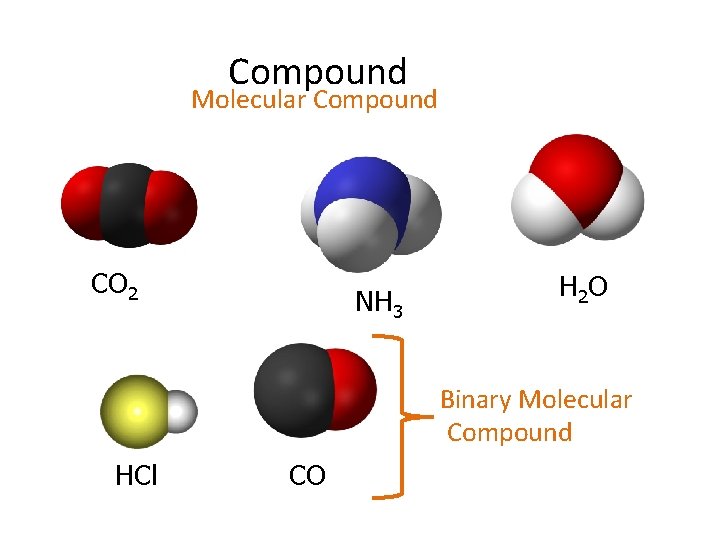
Compound Molecular Compound CO 2 NH 3 H 2 O Binary Molecular Compound HCl CO

Covalent Molecule Properties • Covalent molecular solids tend to be soft solids, liquids, or gases at room temperature • Low melting and boiling points • Poor conductors of heat and electricity • Non-electrolytes – do not conduct electricity in water
- Slides: 16