Covalent Bonds Page 89 of INB Essential Question

Covalent Bonds Page 89 of INB

Essential Question • How does the Lewis dot structure help determine the bonding of an atom?

Covalent Bonds • Covalent Bonds: Atoms joined by covalent bonds share electrons. • Forms between 2 non-metals • Usually have low melting points, do not conduct electricity • Can Share more than one pair of electrons • Polar Bonds: created by the uneven sharing of electrons • Electrons are more drawn to the elements to the right and top of P. T.
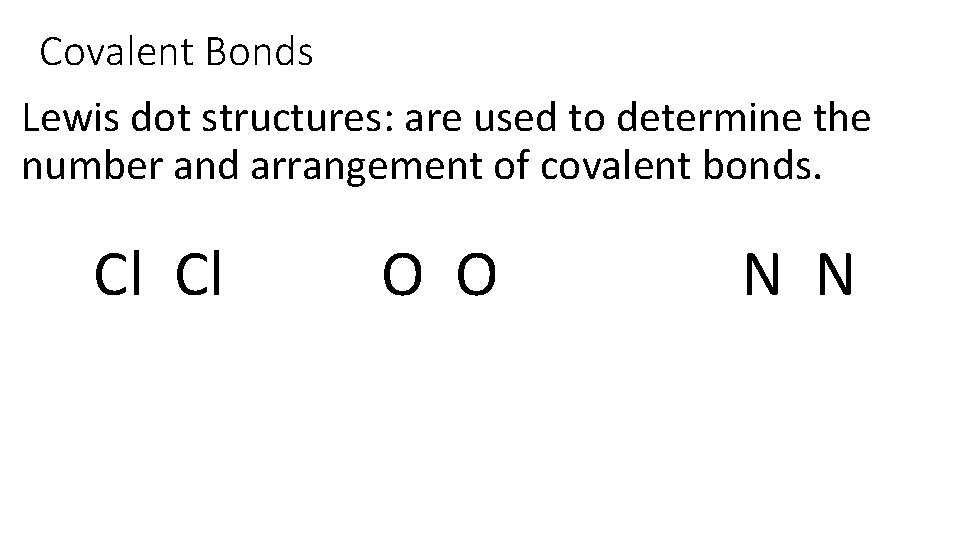
Covalent Bonds Lewis dot structures: are used to determine the number and arrangement of covalent bonds. Cl Cl O O N N

Naming and writing covalent compounds use prefixes to express the number of atoms or the ratio of atoms in the molecule • Rules: • If only 1 atom off the first element no prefix is used • Element furthest to the right s named second and ending is changed to -ide • Vowels at the end of the prefix are dropped if the element begins with a vowel.

1 2 3 4 5 6 7 8 9 Mono Di Tri Tetra Penta Hexa Hepta Octa Nano

Covalent Naming Practice: 1)Dinitrogen Tetroxide 2)Boron Trifluoride 3)Silicon Tetriodide 4)Se. O 2 5)As 2 O 5 6)P 4 S 3
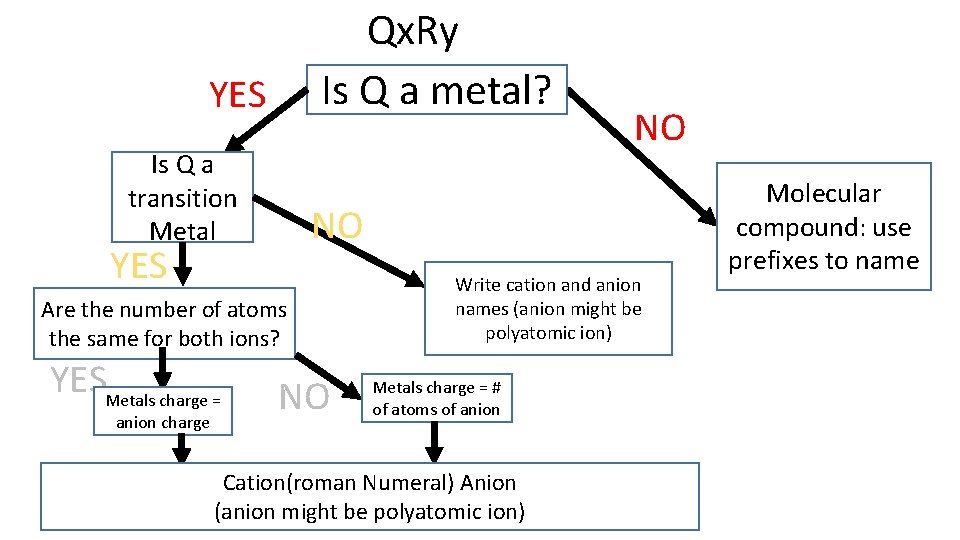
Qx. Ry Is Q a metal? YES Is Q a transition Metal NO YES Are the number of atoms the same for both ions? YESMetals charge = anion charge NO NO Write cation and anion names (anion might be polyatomic ion) Metals charge = # of atoms of anion Cation(roman Numeral) Anion (anion might be polyatomic ion) Molecular compound: use prefixes to name
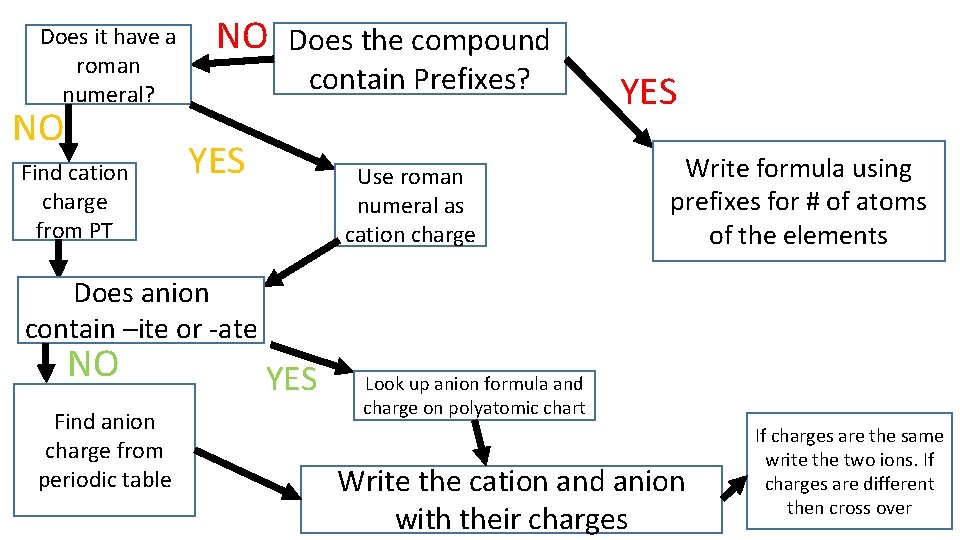
Does it have a roman numeral? NO Find cation charge from PT NO Does the compound contain Prefixes? YES Use roman numeral as cation charge YES Write formula using prefixes for # of atoms of the elements Does anion contain –ite or -ate NO Find anion charge from periodic table YES Look up anion formula and charge on polyatomic chart Write the cation and anion with their charges If charges are the same write the two ions. If charges are different then cross over
- Slides: 9