COVALENT BONDS NONPOLAR AND POLAR Covalent Bonds Covalent

COVALENT BONDS: NONPOLAR AND POLAR

Covalent Bonds Covalent bonds form • when atoms share electrons to complete their outer shells. • between two nonmetal atoms. • between nonmetal atoms from Groups 14, 15, 16, and 17. 2

Covalent Bonds Definition 1) chemical bond in which two atoms share a pair of valence electrons 2) can be a single, double, or triple bond single, 2 e-’s (-); double, 4 e-’s (=); triple, 6 e-’s( ) 3)2 types of bonds - polar - non polar

Electrons are shared between atoms. Both elements are nonmetals. Molecules are formed with covalent bonds. Two types: Polar and non-polar Covalent bonded molecules are not conductors at any state Ex: H 2 O, O 2, C 2 H 6, Si. O 2, C 6 H 12 O 6 Covalent Bonds

MOLECULE STRUCTURE When atoms bond, they usually have to rearrange their electrons from the positions pictured in Lewis Diagrams. The goal is for every atom to have eight electrons around it (except for hydrogen which has only two electrons). HINT: Carbon, nitrogen, and sulfur are usually the central atom(s) (in the center) surrounded by terminal atoms (surrounding central). Carbon is always a central atom and hydrogen is always a terminal atom. When you draw a dot diagram for a molecule you start with the atom that is only in the formula once—it will be in the center of the molecule with the other atoms arranged around it.
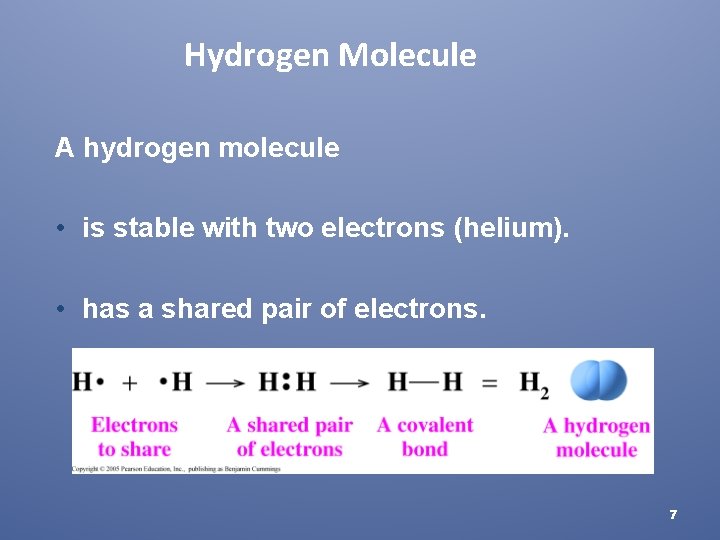
Hydrogen Molecule A hydrogen molecule • is stable with two electrons (helium). • has a shared pair of electrons. 7
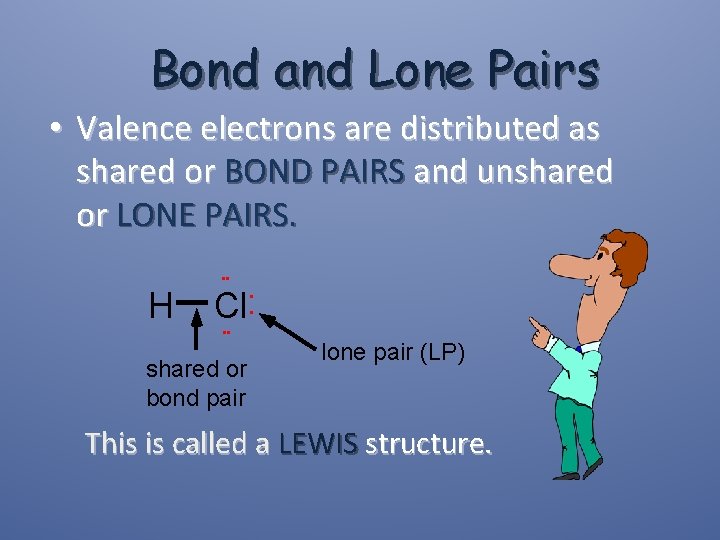
Bond and Lone Pairs • Valence electrons are distributed as shared or BOND PAIRS and unshared or LONE PAIRS. • • H Cl • • shared or bond pair • • lone pair (LP) This is called a LEWIS structure.

Electron distribution is depicted with Lewis electron dot structures This is how you decide how many atoms will bond covalently. G. N. Lewis 1875 - 1946 (In ionic bonds, it was decided with oxidation numbers)

Nonpolar Covalent Molecules

Multiple Bonds In nitrogen molecule, N 2, each N atom shares 3 electrons. each N attains has a full outer shell. the bond is a multiple bond called a triple bond. the name is the same as the element. 11
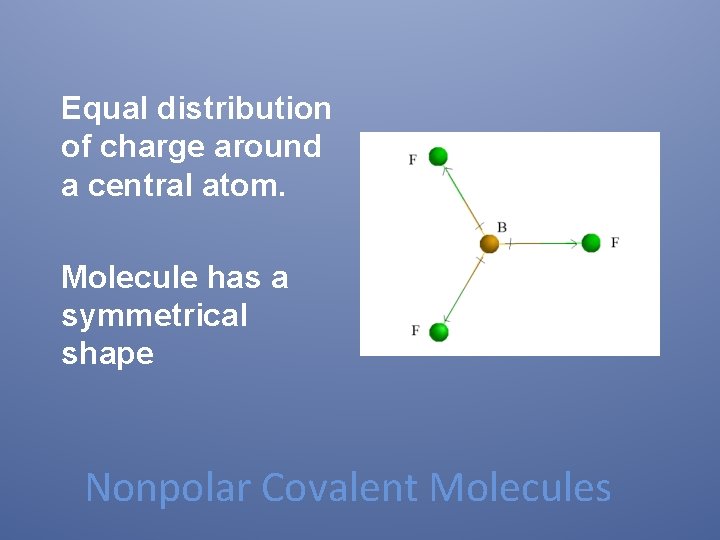
Equal distribution of charge around a central atom. Molecule has a symmetrical shape Nonpolar Covalent Molecules

In a chlorine molecule, each of the atoms shares a pair of electrons circled in GREEN, making the covalent bond. Since it’s just one pair being shared, this is a SINGLE COVALENT BOND. Each of the chlorine atoms also has three pairs each of UNSHARED ELECTRONS. THEY BOTH GET FULL OUTER SHELLS (AN OCTET) BY SHARING.
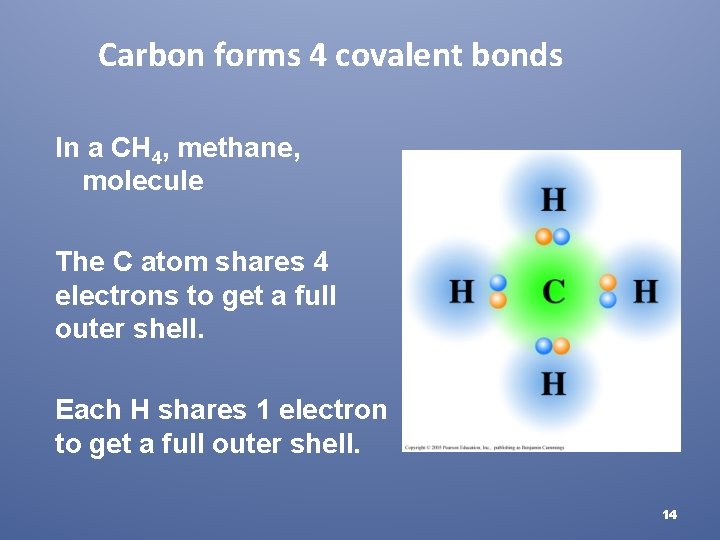
Carbon forms 4 covalent bonds In a CH 4, methane, molecule The C atom shares 4 electrons to get a full outer shell. Each H shares 1 electron to get a full outer shell. 14

When electrons are written as a Lewis diagrams, they are usually paired. Electrons prefer being in pairs. When electrons are shared between atoms, one electron from each atom connects together, making a SINGLE BOND. Electrons not involved in bonding are mostly paired away from the bond. They are the “unshared” electrons. If they connect 2 or 3 pairs at a time, they make DOUBLE or TRIPLE BONDS.

When 2 oxygen atoms combine to form a molecule, each keeps two pairs of unshared electrons, but shares two pairs (in the middle) with each other. That is a DOUBLE COVALENT BOND

Diatomic Nitrogen makes a TRIPLE COVALENT BOND and each atom also has one pair of UNSHARED ELECTRONS.
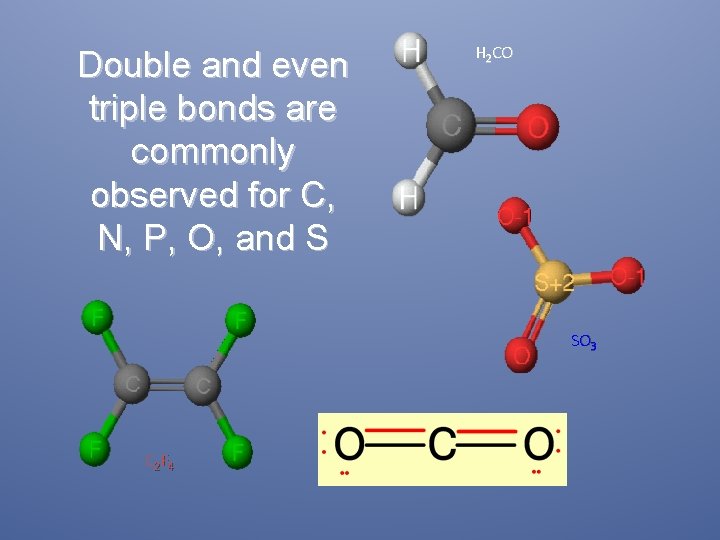
Double and even triple bonds are commonly observed for C, N, P, O, and S H 2 CO SO 3 C 2 F 4

Predicting Compounds using Lewis Dot Structures • Going back to the idea of Lewis dot configuration as a good way to keep track of valence electrons for predicting structure of ionic/covalent compounds. 19

Covalent Compounds • Covalent compound forms from oxygen and hydrogen • Step 1: Determine how many bonds are formed by oxygen • Step 2: Determine how many hydrogen atoms are in the chemical formula (hydrogen forms a single bond) • Step 3: Draw the structure • 20
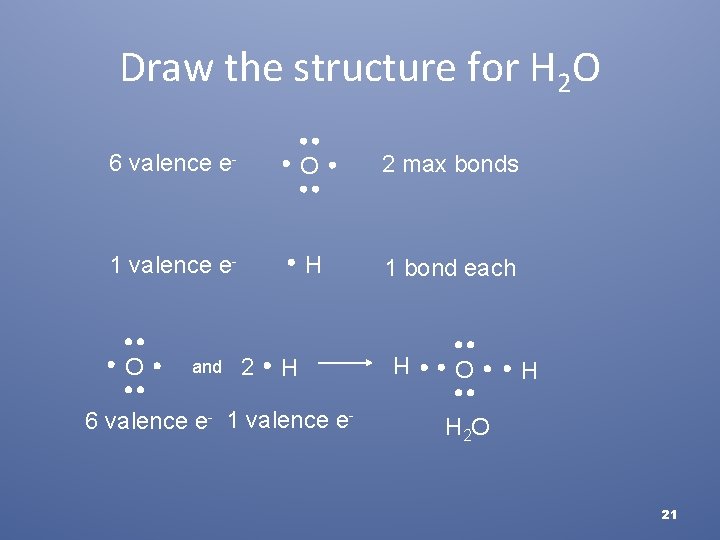
Draw the structure for H 2 O 6 valence e- O 2 max bonds 1 valence e- H 1 bond each and 2 H 6 valence e- 1 valence e- H O O H H 2 O 21

Chlorine forms a covalent bond with itself Cl 2

Cl Cl How will two chlorine atoms react?

Cl Cl Each chlorine atom wants to gain one electron to achieve a full outer shell.

Cl Cl Neither atom will give up an electron chlorine is highly electronegative. What’s the solution – what can they do to achieve a full outer shell?

Cl Cl

Cl Cl

Cl Cl

Cl Cl

Cl Cl Full shell

Cl Cl Full shell Circle the shared electrons

Cl Cl The 8 in the outer shell (octet) is achieved by each atom sharing the electron pair in the middle We circle the shared electrons

Cl Cl Full shells are achieved by each atom sharing the electron pair in the middle

Cl Cl This is the bonding pair

Cl Cl It is a single bonding pair

Cl Cl It is called a SINGLE BOND We circle the electrons for each atom that completes their octets

Cl Cl Single bonds can be shown with a dash

Cl Cl This is the chlorine molecule, Cl 2

O 2 Oxygen is also one of the diatomic molecules

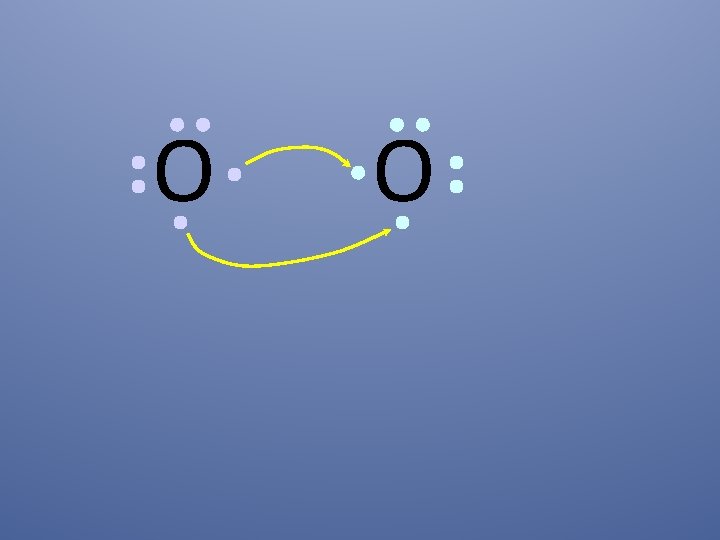
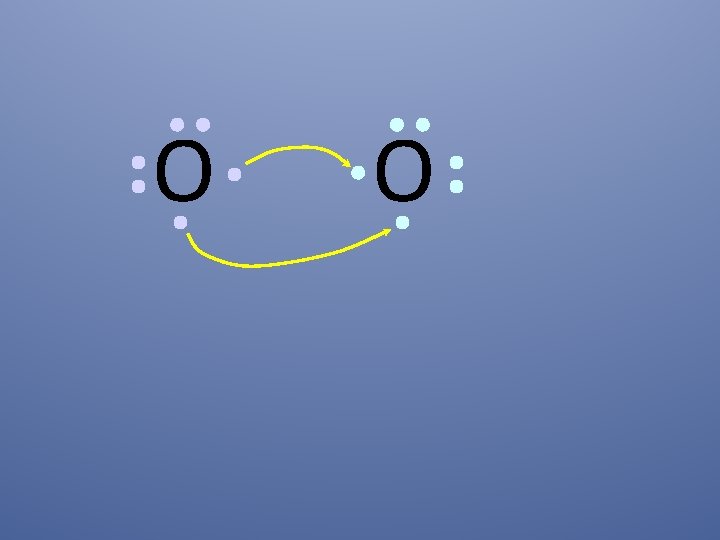
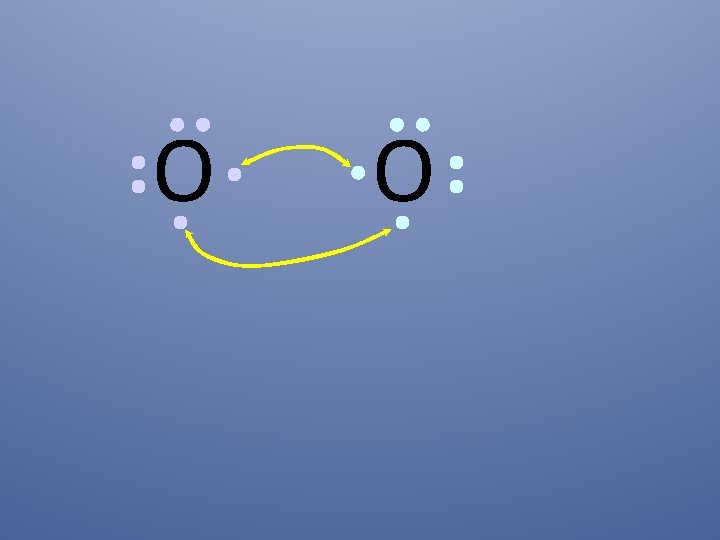
O O How will two oxygen atoms bond?
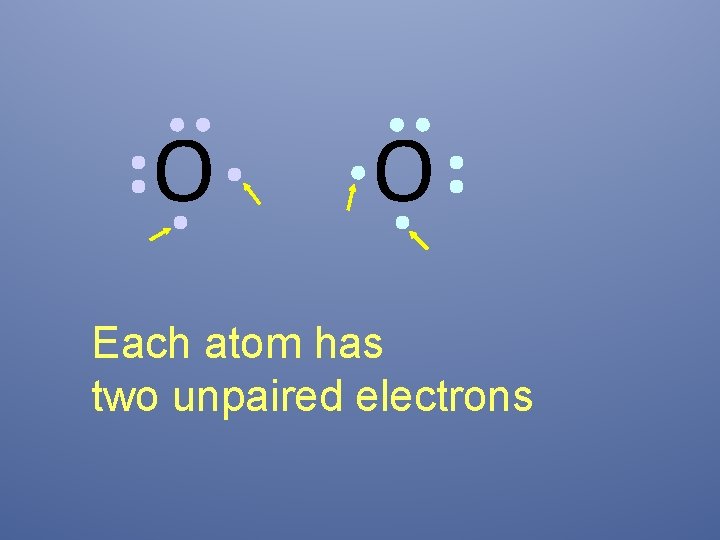
O O Each atom has two unpaired electrons
O O
O O
O O
O O
O O
O O

O O Oxygen atoms are highly electronegative, so both atoms want to gain two electrons.
O O

O O
O O

O O

O O
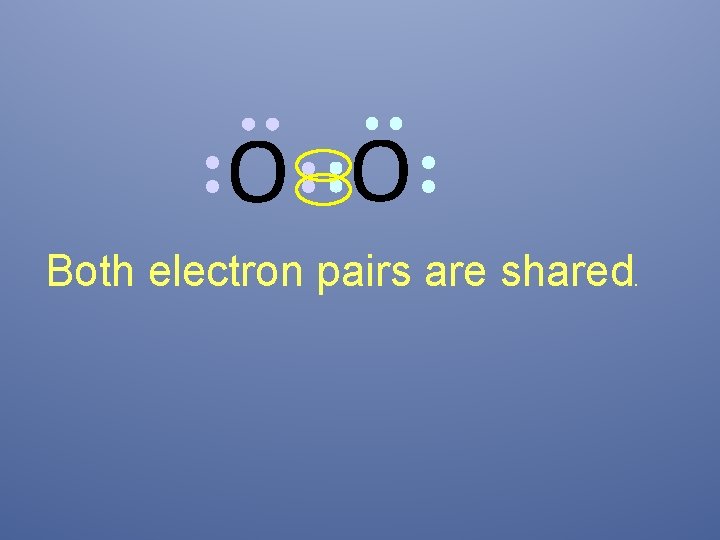
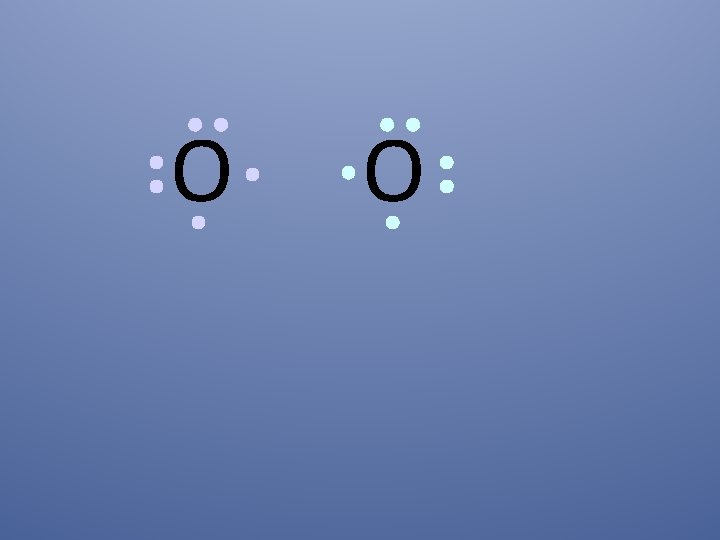
O O Both electron pairs are shared.
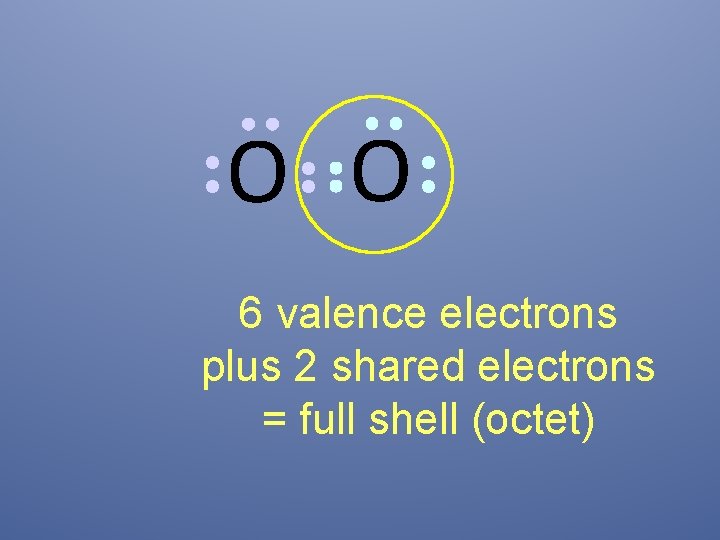
O O 6 valence electrons plus 2 shared electrons = full shell (octet)
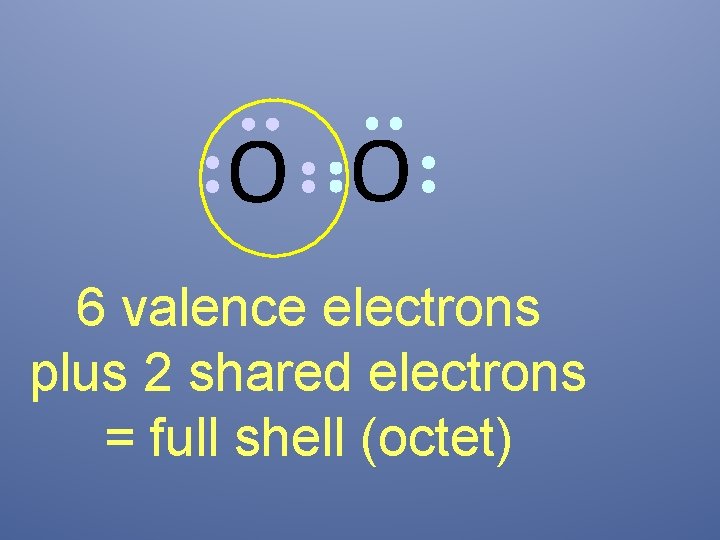
O O 6 valence electrons plus 2 shared electrons = full shell (octet)
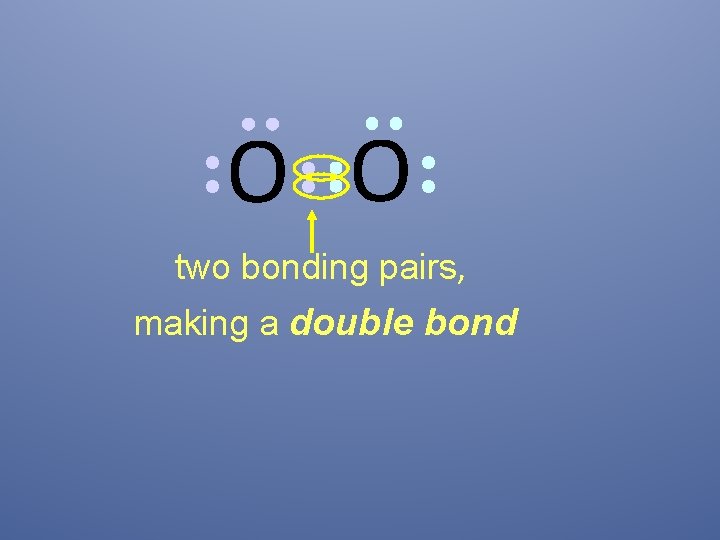
O O two bonding pairs, making a double bond

O O O=O For convenience, the double bond can be shown as two dashes.
O=O this is so cool!! This is the oxygen molecule, O 2
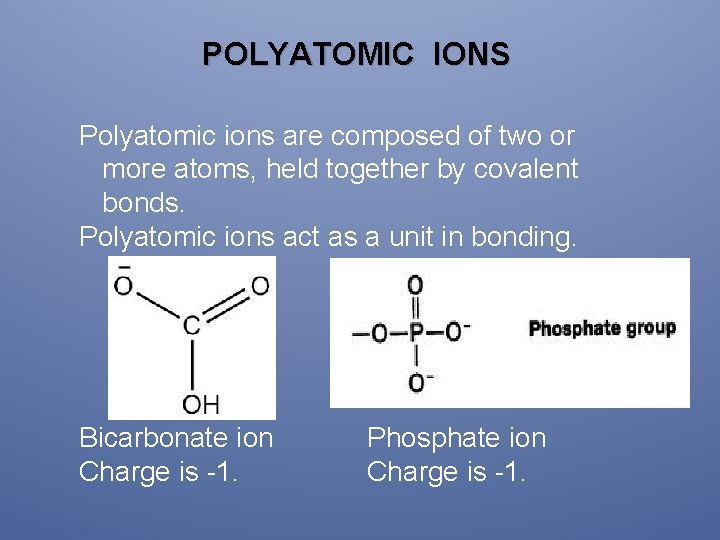
POLYATOMIC IONS Polyatomic ions are composed of two or more atoms, held together by covalent bonds. Polyatomic ions act as a unit in bonding. Bicarbonate ion Charge is -1. Phosphate ion Charge is -1.

Covalent Bonds Can be polar or nonpolar Non Polar bonded atoms that share e-’s equally true when same atoms bonded ex. Cl – Cl: Cl 2 Polar bonded atoms that do not share e-’s equally different atoms bonded, depends on shape, too ex: NH 3 H H–N–H

Polar Bonds HCl is POLAR because it has a positive end a negative end. (difference in electronegativity) Cl has a greater share of the bonding electron’s time than does H Cl end has slight negative charge and H end has slight positive charge.

Polar Bonds This is why oil and water will not mix! Oil is nonpolar, and water is polar. The two will repel each other, and so you can not dissolve one in the other

Polar Bonds “Like Dissolves Like” Polar dissolves Polar Nonpolar dissolves Nonpolar

Polar Covalent Molecules

Formed when 2 atoms with different electro-negativities form a covalent bond. The atoms DO NOT SHARE electrons equally. The electrons are more strongly attracted to the MOST electronegative atom. Molecules are asymmetrical.
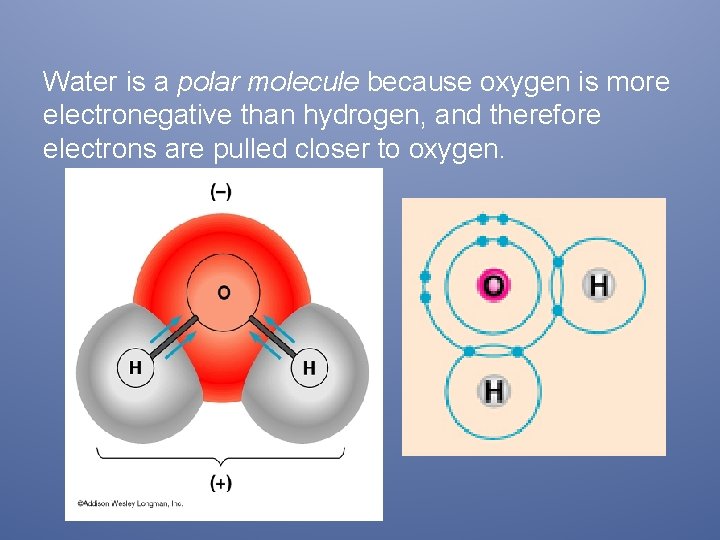
Water is a polar molecule because oxygen is more electronegative than hydrogen, and therefore electrons are pulled closer to oxygen.

A molecule can also be polar, if the electrons are moved about by polar bonds giving the WHOLE MOLECULE a positive and a negative side. The opposite sides of polarity are called dipoles, which means 2 poles, one positive and the other end negative. The oxygen side is negative since it takes the electrons, the hydrogen side is more positive because it lost the electrons.

Naming Covalent Compounds

Naming Covalent Compounds Cont. Rules for the prefix system 1. Less electronegative (leftmost) element is given first. It is given a prefix only if there’s more than one atom in the molecule. 2. The second element gets the ide ending and a prefix indicating the number of atoms contributed ex. Monoxide or pentoxide

To name compounds of different elements: Name the first element Change the last element’s last syllable to –ide Add prefixes: mon/mono di tri tetra penta 1 2 3 4 5 hexa hepta octa nona deca 6 7 8 9 10 Covalent Naming of 2 Nonmetals

Naming Covalent Compounds What is the name of SO 3? 1. The first nonmetal is S sulfur. 2. The second nonmetal is O named oxide. 3. The subscript 3 of O is shown as the prefix tri. SO 3 sulfur trioxide The subscript 1 (for S) or mono is understood.

Naming Covalent Compounds Name P 4 S 3. 1. The first nonmetal P is phosphorus. 2. The second nonmetal S is sulfide. 3. The subscript 4 of P is shown as tetra. The subscript 3 of O is shown as tri. P 4 S 3 tetraphosphorus trisulfide

Fill in the blanks to complete the following names of covalent compounds. CO carbon ______oxide Si. O 2 silicon ______oxide PCl 3 phosphorus _______chloride CCl 4 carbon ____chloride N 2 O _____nitrogen ___oxide P 2 O 5 ___phosphorus ____oxide Learning Check

CO carbon monoxide CO 2 carbon dioxide PCl 3 phosphorus trichloride CCl 4 carbon tetrachloride N 2 O dinitrogen monoxide P 2 O 5 diphosphorus pentoxide Solution
- Slides: 75