Covalent Bonds Molecules IV Electron Sharing Produces Molecules

Covalent Bonds Molecules IV

Electron Sharing Produces Molecules • Covalent bonds are formed when atoms produce stable octets by sharing valence electrons. Covalently bonded atoms form Molecules. • In a covalent bond, atoms share electrons and neither atom has an ionic charge. IV

Electron Sharing Produces Molecules • A molecule is an uncharged group of two or more atoms held together by covalent bonds. • Ethanol, also known as ethyl alcohol, is a typical covalent compound. IV

Bonding Video
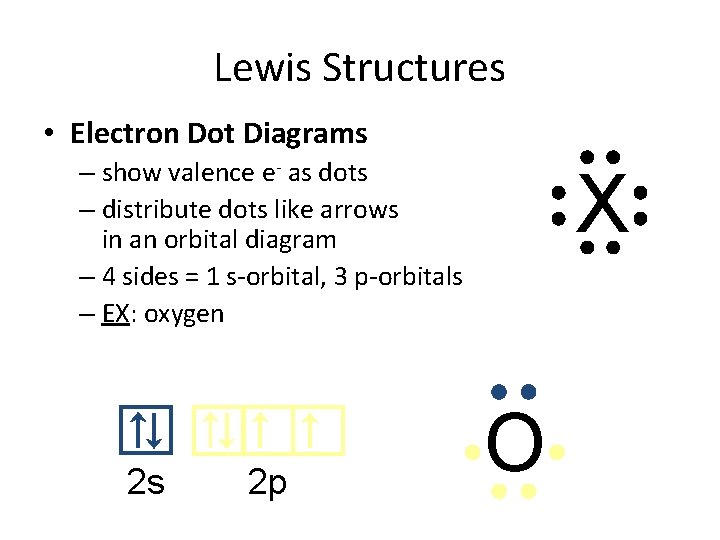
Lewis Structures • Electron Dot Diagrams – show valence e- as dots – distribute dots like arrows in an orbital diagram – 4 sides = 1 s-orbital, 3 p-orbitals – EX: oxygen 2 s 2 p X O

Lewis Structures Octet Rule Most atoms form bonds in order to obtain 8 valence e Full energy level stability ~ Noble Gases Remember the exceptions: H, B, N, P, S and others Ne

Single, Double or Triple Covalent Bonds • Single Bonds – Chlorine & Hydrogen • Double Bonds – Oxygen • Triple Bonds – Nitrogen – Carbon forms up to four bonds, can form single, double or triple bonds.
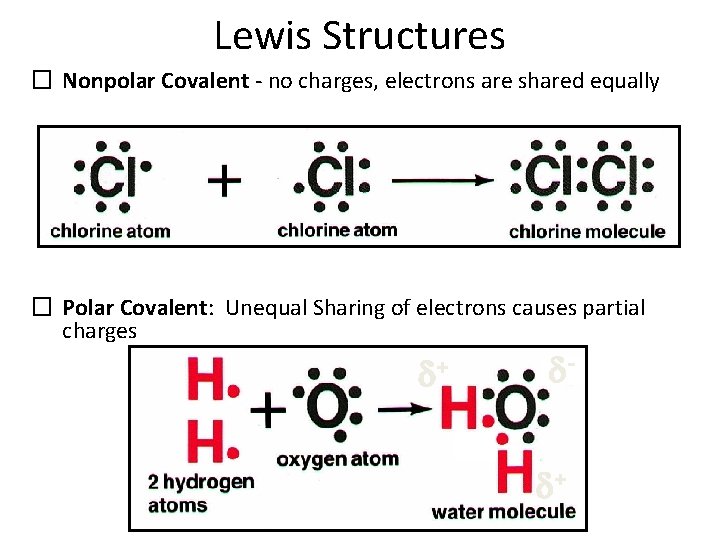
Lewis Structures � Nonpolar Covalent - no charges, electrons are shared equally � Polar Covalent: Unequal Sharing of electrons causes partial charges + - + C. Johannesson

Polarity • POLARITY: Covalent molecules will usually display the characteristic of Polarity. This is because of the chemical property of electronegativity. Some atoms attract electrons more strongly, and thus electrons are not shared equally. The slight separation of charge is Polarity. Water is a very polar covalent molecule.
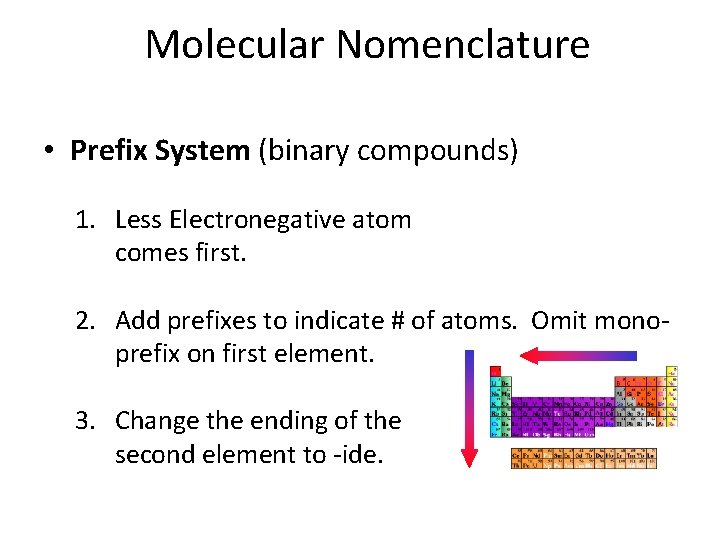
Molecular Nomenclature • Prefix System (binary compounds) 1. Less Electronegative atom comes first. 2. Add prefixes to indicate # of atoms. Omit monoprefix on first element. 3. Change the ending of the second element to -ide.

Molecular Nomenclature PRPEFIX monoditritetrapentahexaheptaoctanonadeca- NUMBER 1 2 3 4 5 6 7 8 9 10

Molecular Nomenclature z. CCl 4 ycarbon tetrachloride z. N 2 O ydinitrogen monoxide z. SF 6 ysulfur hexafluoride

Molecular Nomenclature zarsenic trichloride y. As. Cl 3 zdinitrogen pentoxide y. N 2 O 5 ztetraphosphorus decoxide y. P 4 O 10
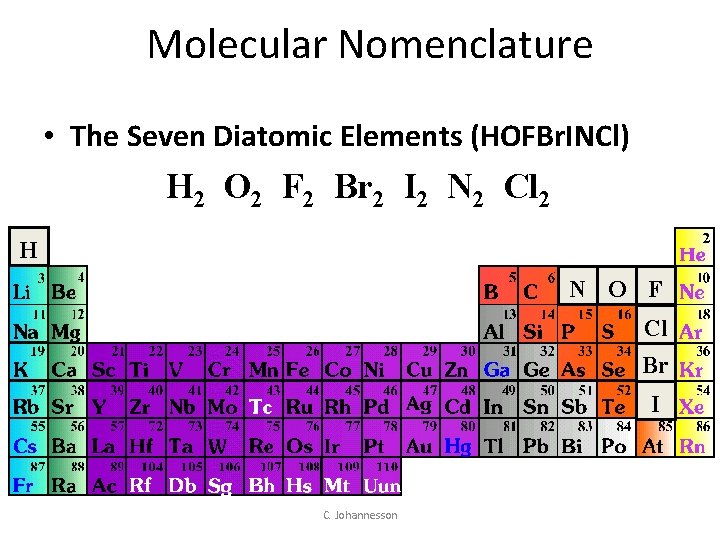
Molecular Nomenclature • The Seven Diatomic Elements (HOFBr. INCl) H 2 O 2 F 2 Br 2 I 2 N 2 Cl 2 H N O F Cl Br I C. Johannesson
- Slides: 14