Covalent Bonding Molecular Polarity I II III Covalent
Covalent Bonding Molecular Polarity I II III

Covalent Bonds involve sharing electrons But Just like in real life, not all sharing Is equal !!!
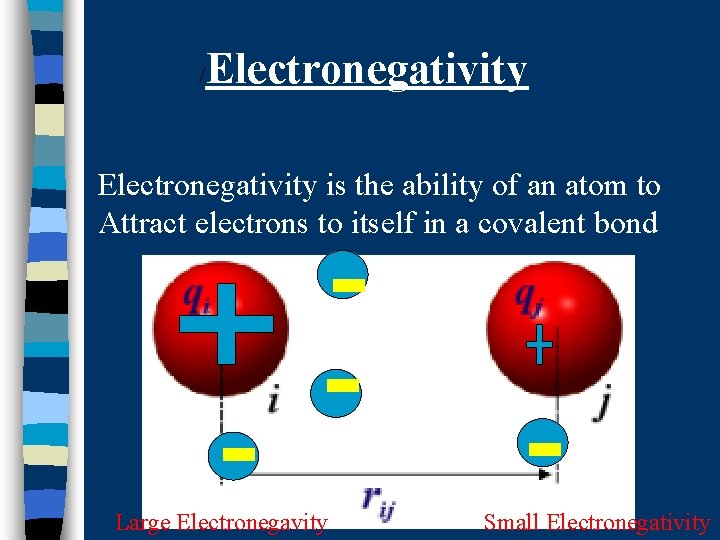
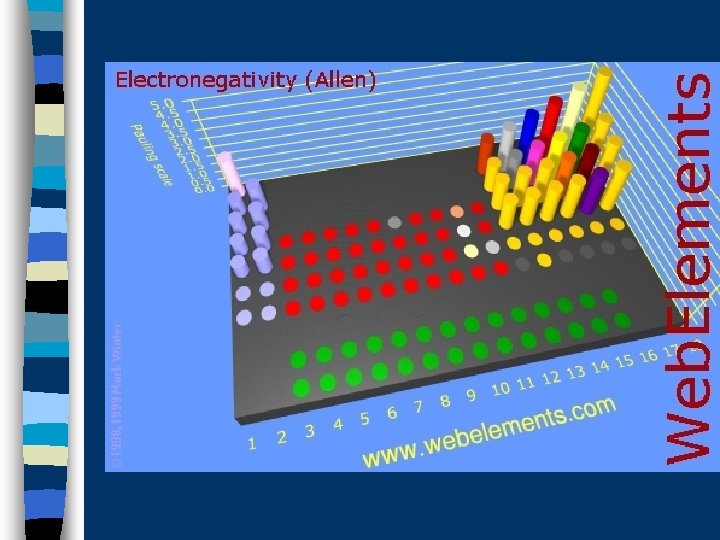
/ Electronegativity is the ability of an atom to Attract electrons to itself in a covalent bond Large Electronegavity Small Electronegativity
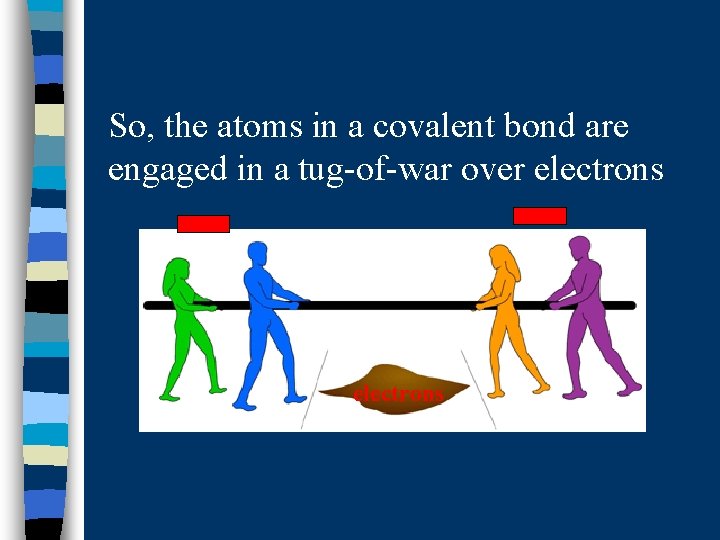
So, the atoms in a covalent bond are engaged in a tug-of-war over electrons
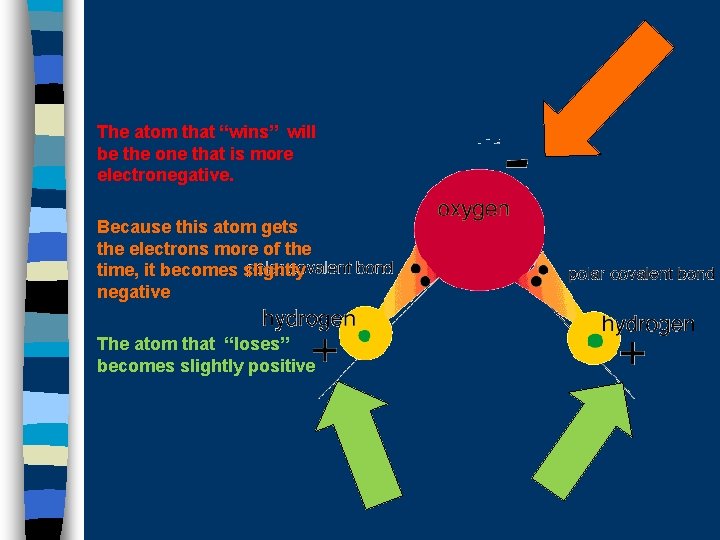
The atom that “wins” will be the one that is more electronegative. Because this atom gets the electrons more of the time, it becomes slightly negative The atom that “loses” becomes slightly positive

The Periodic Table allows us To predict differences in electronegavity
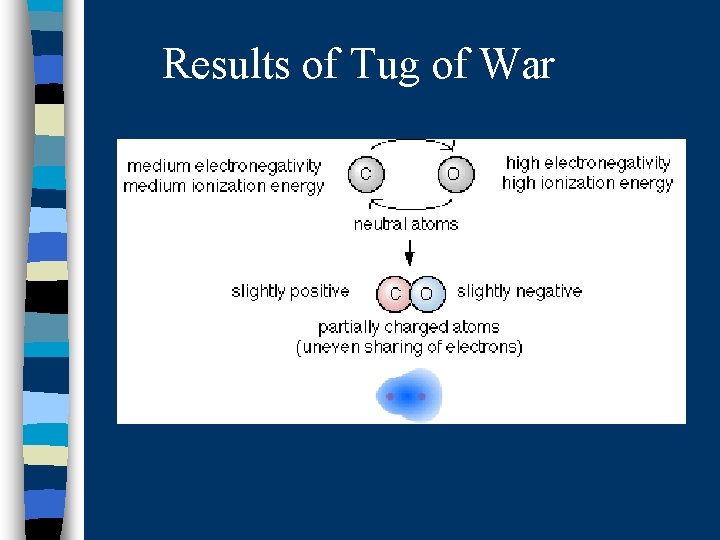
Results of Tug of War
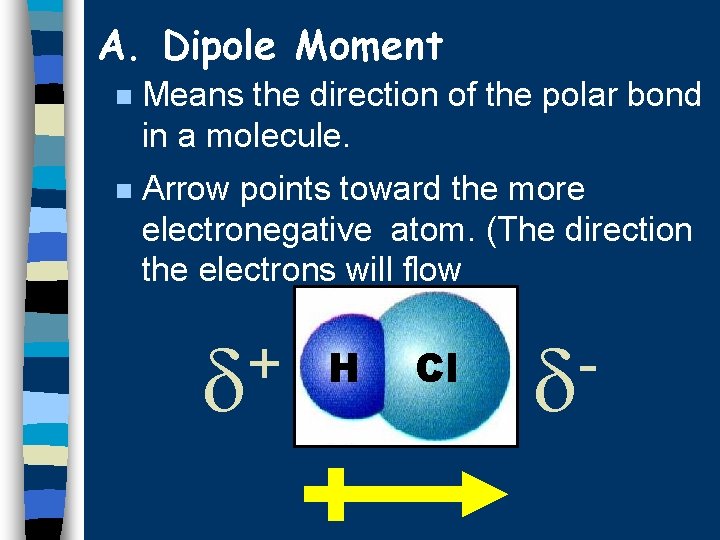
A. Dipole Moment n Means the direction of the polar bond in a molecule. n Arrow points toward the more electronegative atom. (The direction the electrons will flow + H Cl
B. Determining Molecular Polarity n Depends on: · dipole moments · molecular shape
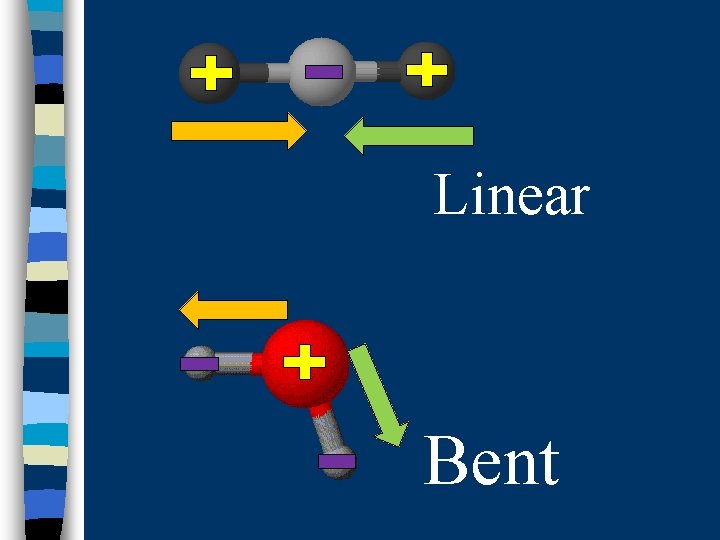
Linear Bent
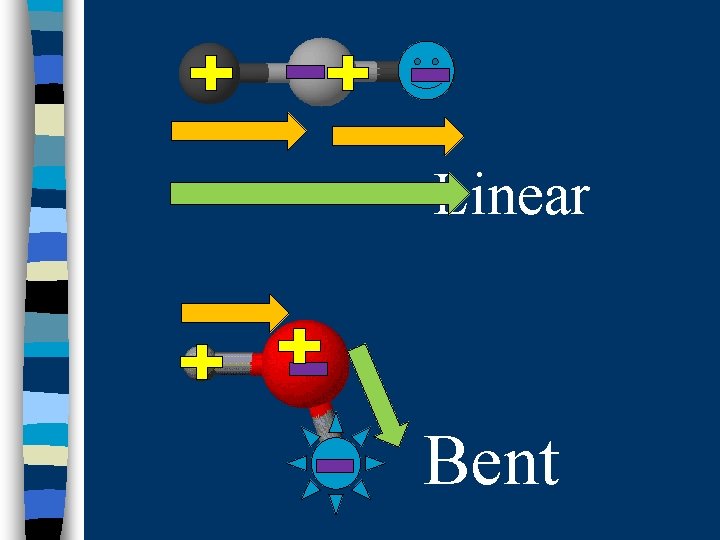
Linear Bent

Trigonal Planar
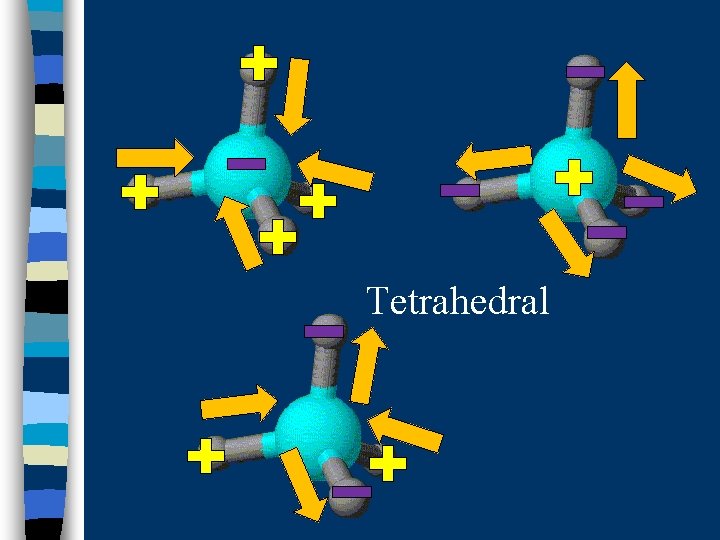
Tetrahedral
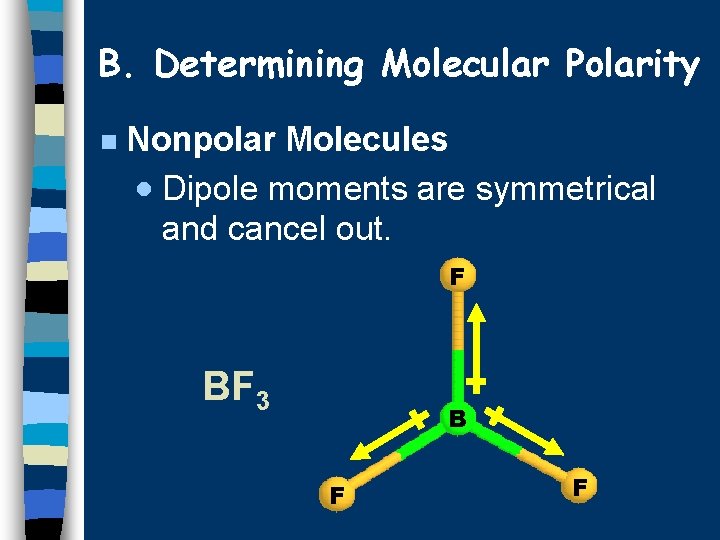
B. Determining Molecular Polarity n Nonpolar Molecules · Dipole moments are symmetrical and cancel out. F BF 3 B F F
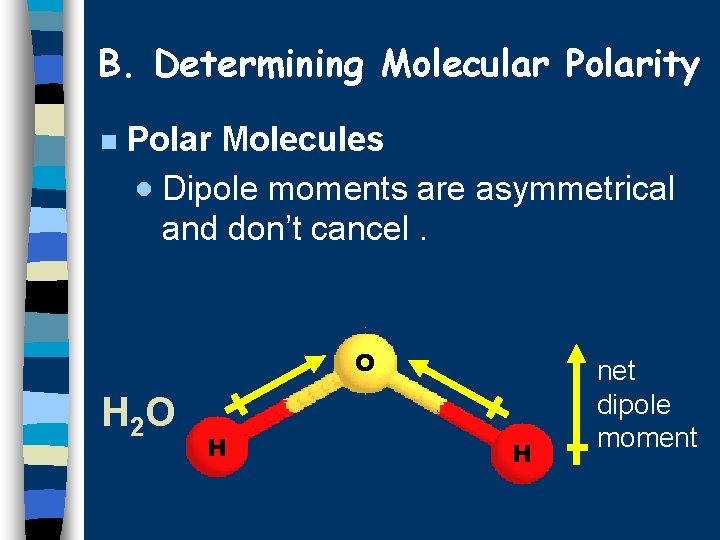
B. Determining Molecular Polarity n Polar Molecules · Dipole moments are asymmetrical and don’t cancel. O H 2 O H H net dipole moment
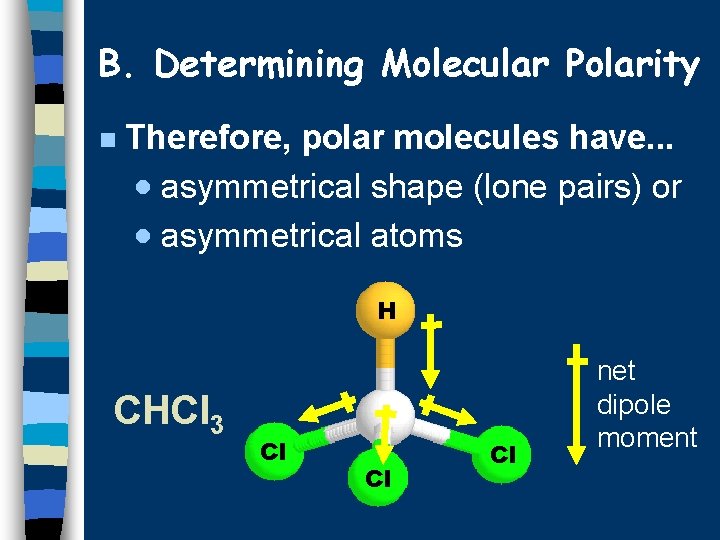
B. Determining Molecular Polarity n Therefore, polar molecules have. . . · asymmetrical shape (lone pairs) or · asymmetrical atoms H CHCl 3 Cl Cl Cl net dipole moment
So, polarity is more of a spectrum than a black and white determination Polar---------------------------nonpolar

More Nonpolar Molecules • Share electrons the “best” • Have the closest electronegativities (or smallest differences) • Have lower boiling and melting points • . . . so More likely to be gases at room temperature • Like dissolves like. . . • Use a nonpolar solvent to dissolve a nonpolar solute

More Polar Molecules • Do not share well at all! • Their electronegativity differences are great • Are more like ionic compounds • Have higher melting and boiling points • Most likely to be liquids at room temperature • Like dissolves like. . . • Use a polar/ionic solvent to dissolve a polar/ionic solute

Ionic, Polar, Nonpolar: A Continuum Ionic------------Polar--------Nonpolar Electrons transferred E- shared unequally High mp/bp Solids at STP E- shared Low mp/bp Liquids at STP Gas at STP
- Slides: 21