Covalent Bonding Learning Objectives 1 Describe how covalent

Covalent Bonding Learning Objectives: 1. Describe how covalent bonding occurs when atoms share electrons. 2. Deduce formulas for simple covalent molecules. 3. Draw dot and cross diagrams to represent simple covalent molecules. 4. Draw representations of polymers. 5. Compare different models to represent molecules.
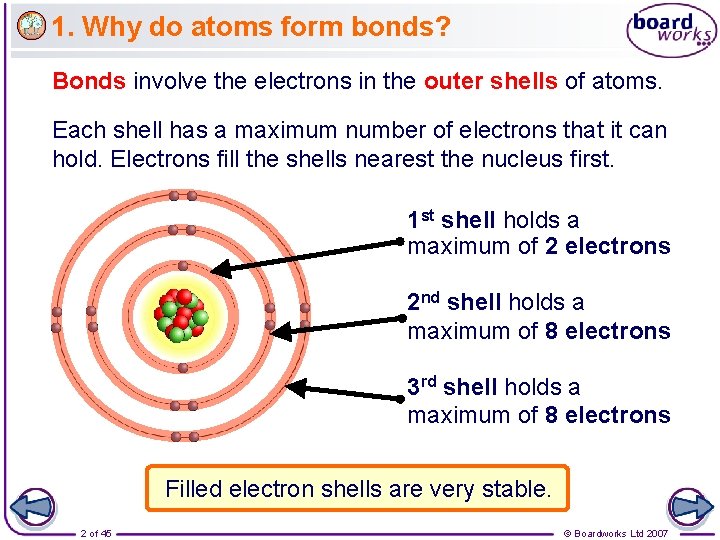
1. Why do atoms form bonds? Bonds involve the electrons in the outer shells of atoms. Each shell has a maximum number of electrons that it can hold. Electrons fill the shells nearest the nucleus first. 1 st shell holds a maximum of 2 electrons 2 nd shell holds a maximum of 8 electrons 3 rd shell holds a maximum of 8 electrons Filled electron shells are very stable. 2 of 45 © Boardworks Ltd 2007

2. Why do atoms form bonds? The atoms of noble gases have completely full outer shells and so are stable. This makes the noble gases very unreactive and so they do not usually form bonds. The atoms of other elements have incomplete outer electron shells and so are unstable. By forming bonds, the atoms of these elements are able to have filled outer shells and become stable. 3 of 45 © Boardworks Ltd 2007
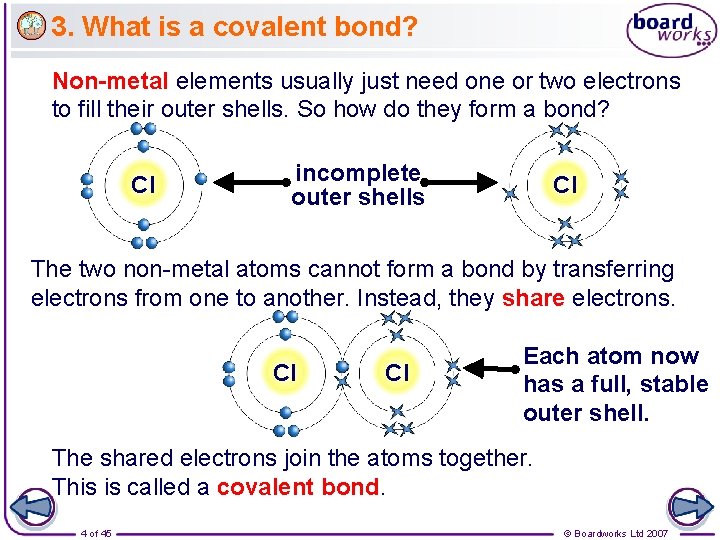
3. What is a covalent bond? Non-metal elements usually just need one or two electrons to fill their outer shells. So how do they form a bond? Cl incomplete outer shells Cl The two non-metal atoms cannot form a bond by transferring electrons from one to another. Instead, they share electrons. Cl Cl Each atom now has a full, stable outer shell. The shared electrons join the atoms together. This is called a covalent bond. 4 of 45 © Boardworks Ltd 2007
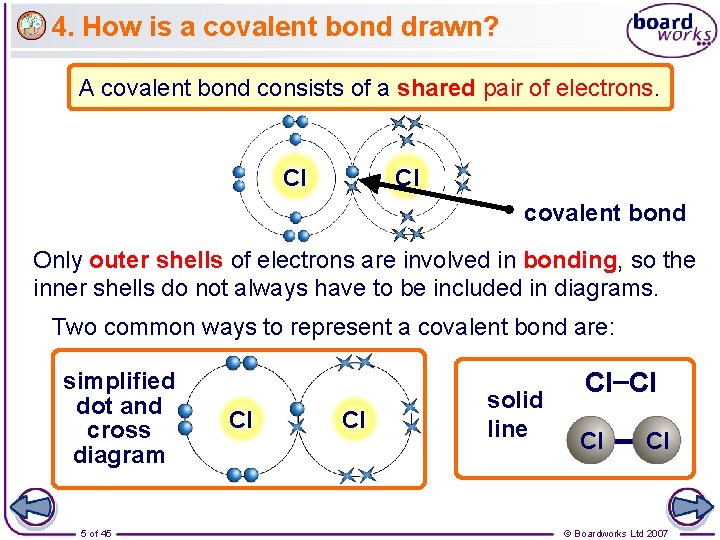
4. How is a covalent bond drawn? A covalent bond consists of a shared pair of electrons. Cl Cl covalent bond Only outer shells of electrons are involved in bonding, so the inner shells do not always have to be included in diagrams. Two common ways to represent a covalent bond are: simplified dot and cross diagram 5 of 45 Cl Cl solid line Cl–Cl Cl – Cl © Boardworks Ltd 2007
5. Comparing covalent and ionic bonding 6 of 45 © Boardworks Ltd 2007

6. Covalent bonding in hydrogen Many non-metal elements, such as hydrogen, exist as simple diatomic molecules (molecule made of two atoms) that contain covalent bonds. How is a covalent bond formed in hydrogen? H H Each hydrogen atom needs one more electron in its outer shell and so each atom shares its single unpaired electron. This shared pair of electrons forms a covalent bond and so creates a diatomic molecule of hydrogen. Some molecules contain double or triple covalent bonds. How are these are formed? 7 of 45 © Boardworks Ltd 2007
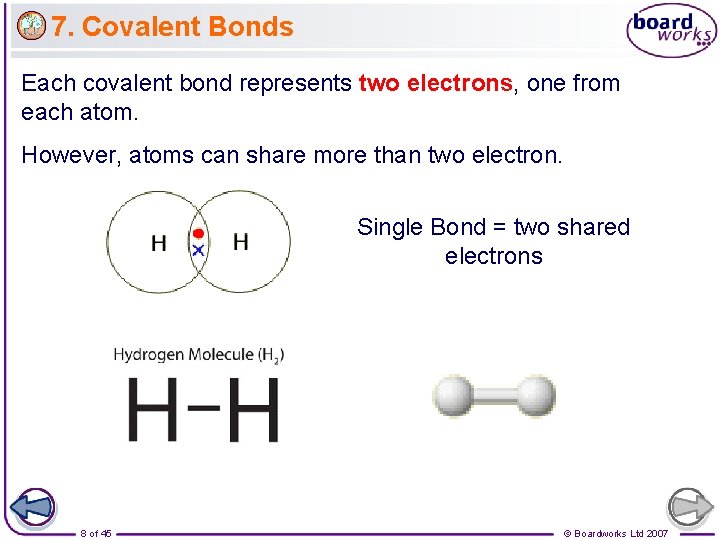
7. Covalent Bonds Each covalent bond represents two electrons, one from each atom. However, atoms can share more than two electron. Single Bond = two shared electrons 8 of 45 © Boardworks Ltd 2007

7. Covalent Bonds Each covalent bond represents two electrons, one from each atom. However, atoms can share more than two electron. Double Bond = four shared electrons 9 of 45 © Boardworks Ltd 2007
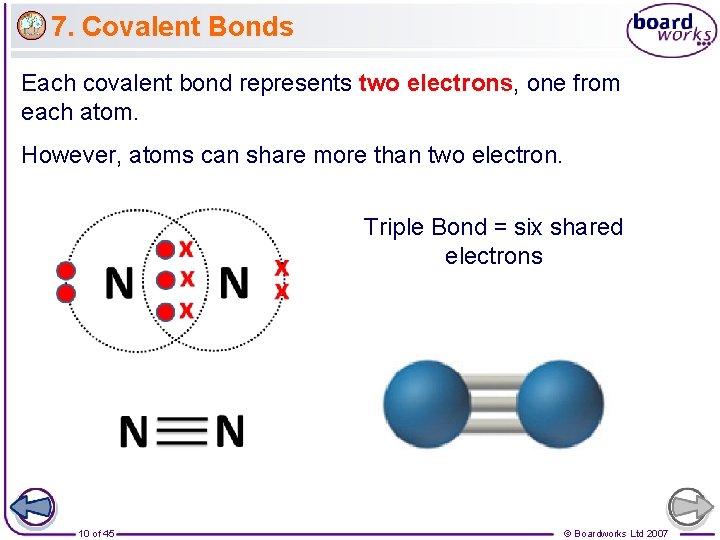
7. Covalent Bonds Each covalent bond represents two electrons, one from each atom. However, atoms can share more than two electron. Triple Bond = six shared electrons 10 of 45 © Boardworks Ltd 2007
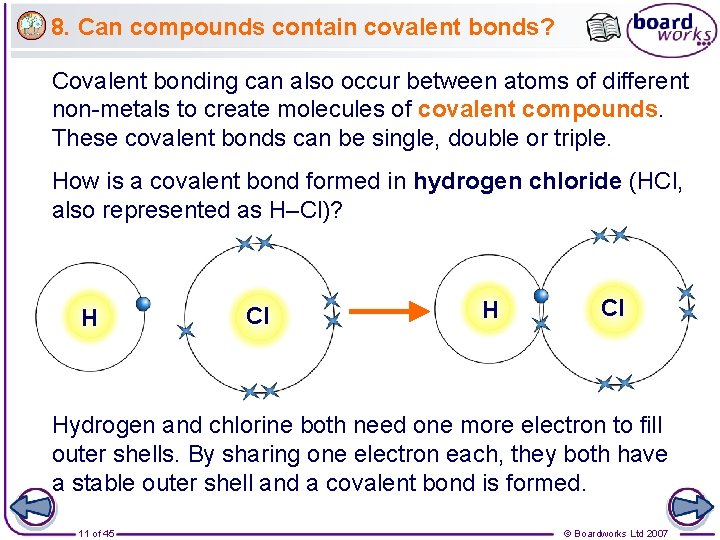
8. Can compounds contain covalent bonds? Covalent bonding can also occur between atoms of different non-metals to create molecules of covalent compounds. These covalent bonds can be single, double or triple. How is a covalent bond formed in hydrogen chloride (HCl, also represented as H–Cl)? H Cl Hydrogen and chlorine both need one more electron to fill outer shells. By sharing one electron each, they both have a stable outer shell and a covalent bond is formed. 11 of 45 © Boardworks Ltd 2007
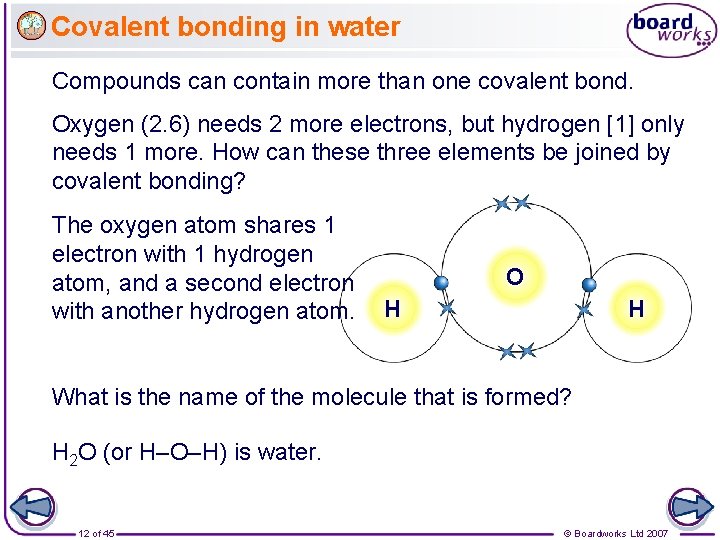
Covalent bonding in water Compounds can contain more than one covalent bond. Oxygen (2. 6) needs 2 more electrons, but hydrogen [1] only needs 1 more. How can these three elements be joined by covalent bonding? The oxygen atom shares 1 electron with 1 hydrogen atom, and a second electron with another hydrogen atom. O H H What is the name of the molecule that is formed? H 2 O (or H–O–H) is water. 12 of 45 © Boardworks Ltd 2007
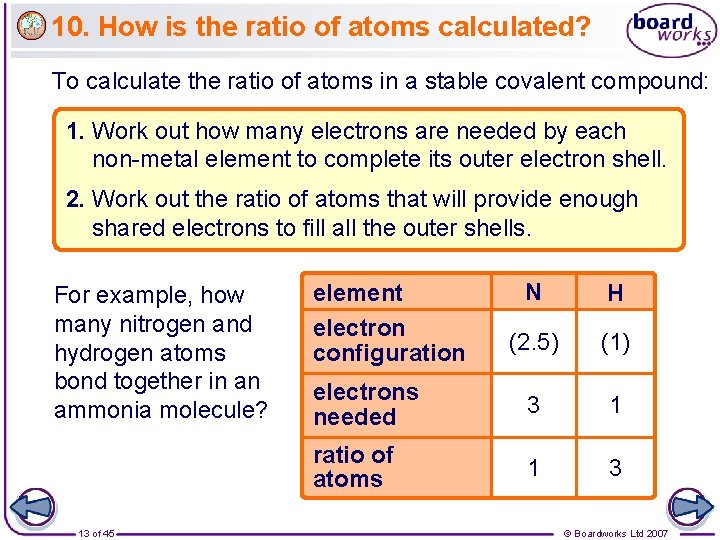
10. How is the ratio of atoms calculated? To calculate the ratio of atoms in a stable covalent compound: 1. Work out how many electrons are needed by each non-metal element to complete its outer electron shell. 2. Work out the ratio of atoms that will provide enough shared electrons to fill all the outer shells. For example, how many nitrogen and hydrogen atoms bond together in an ammonia molecule? 13 of 45 N H (2. 5) (1) electrons needed 3 1 ratio of atoms 1 3 element electron configuration © Boardworks Ltd 2007
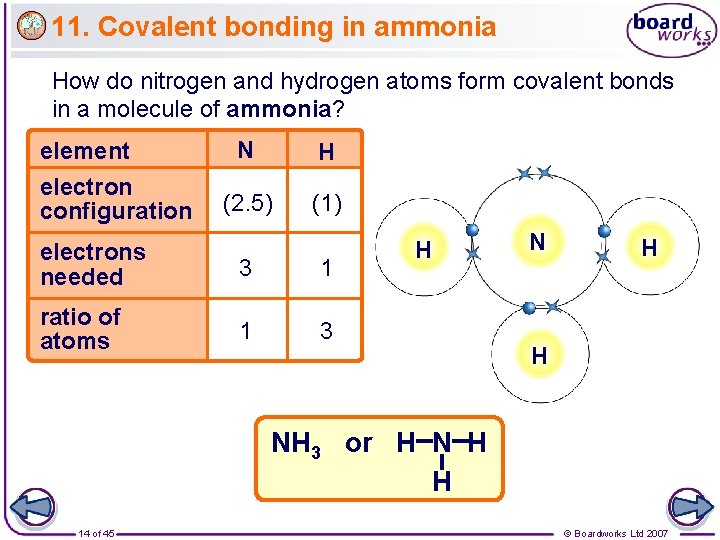
11. Covalent bonding in ammonia How do nitrogen and hydrogen atoms form covalent bonds in a molecule of ammonia? element electron configuration N H (2. 5) (1) electrons needed 3 1 ratio of atoms 1 3 H N H H NH 3 or H N H H 14 of 45 © Boardworks Ltd 2007

12. Write the Formula and Draw Diagrams for the Following a) b) c) d) e) f) g) h) Hydrogen gas Chlorine gas Oxygen gas Nitrogen gas Hydrogen chloride Water (hydrogen and oxygen) Ammonia (nitrogen and hydrogen) Methane (carbon and hydrogen)
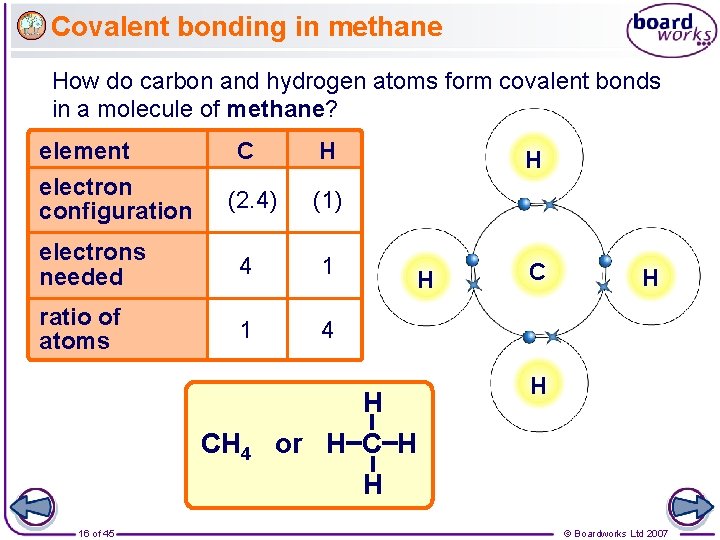
Covalent bonding in methane How do carbon and hydrogen atoms form covalent bonds in a molecule of methane? element C H (2. 4) (1) electrons needed 4 1 ratio of atoms 1 4 electron configuration CH 4 16 of 45 H H H or H C H H © Boardworks Ltd 2007
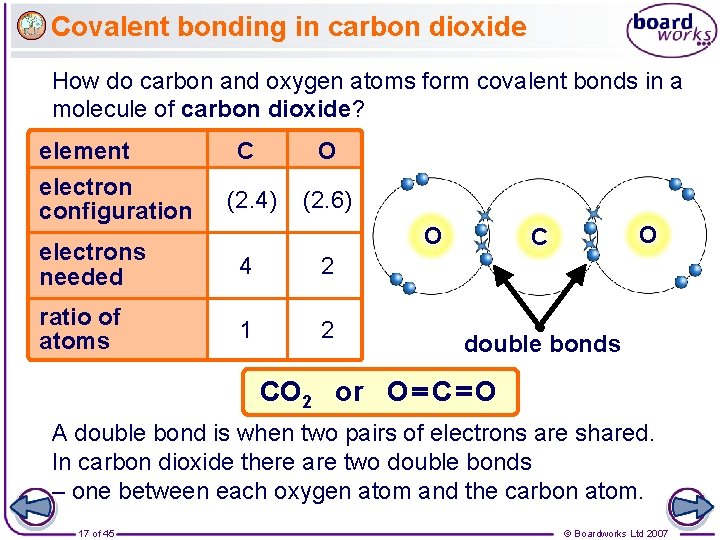
Covalent bonding in carbon dioxide How do carbon and oxygen atoms form covalent bonds in a molecule of carbon dioxide? element electron configuration C O (2. 4) (2. 6) O electrons needed 4 2 ratio of atoms 1 2 O C double bonds CO 2 or O C O A double bond is when two pairs of electrons are shared. In carbon dioxide there are two double bonds – one between each oxygen atom and the carbon atom. 17 of 45 © Boardworks Ltd 2007
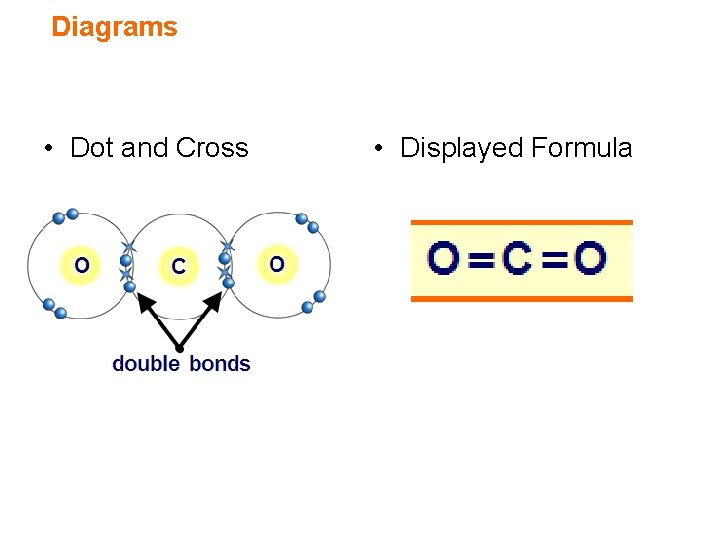
Diagrams • Dot and Cross • Displayed Formula
- Slides: 18