Covalent Bonding Ionic Bonding Remember that in an

Covalent Bonding

Ionic Bonding Remember that in an ionic bond, transferred between electrons are _____ atoms. This transfer creates _____; ions both +___ _ and ___; that are _____ attracted to each other. This attraction holds the compound ____ together.

K F

K F

K F

K F

K F

v. The ionic bond is the attraction _____ between the positive K+ ion and the negative F- ion K + F _

Covalent Bonding Instead of transferring electrons, what if we ______ share them? Look at hydrogen… H It has ____ one valence electron – needsone ___ more to complete its valence shell.

Covalent Bonding Let’s add another hydrogen… H H Each hydrogen needs one more electron – what would happen if they ____ shared the one they already have?

Covalent Bonding Now -- _____ both hydrogens have full ____ valence shells! HH one By sharing ____ pair of electrons, both atoms are full _____!

The other side…? What about the other side of the table? Cl Cl has ___ 7 valence electrons – only needs ___ more to fill its one valence shell. Could we do the same thing with 2 Cl’s?
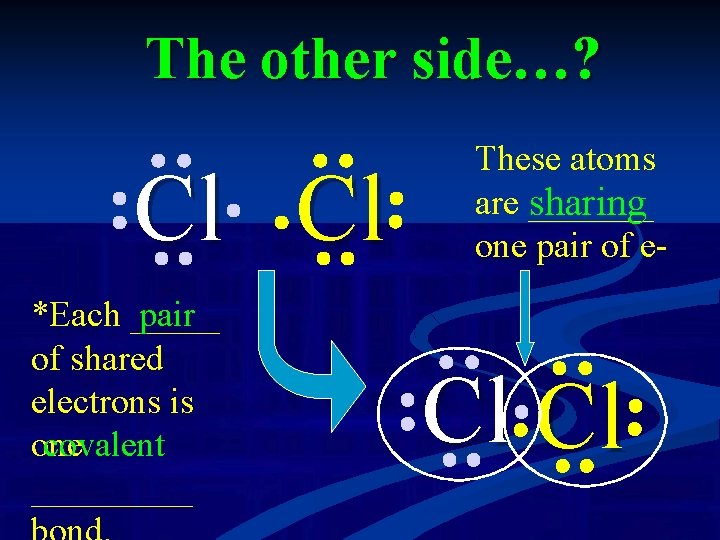
The other side…? Cl Cl *Each _____ pair of shared electrons is covalent one _____ These atoms are _______ sharing one pair of e- Cl Cl
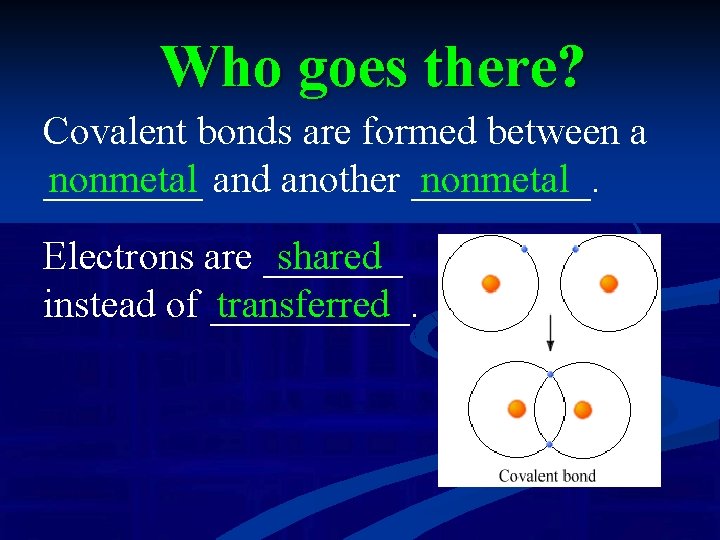
Who goes there? Covalent bonds are formed between a ____ nonmetal and another _____. nonmetal Electrons are _______ shared instead of _____. transferred

You drew it wrong! If you noticed – I drew the dot diagram differently than I taught you… I didn’t make a mistake – it’s just easier to think of these like ‘puzzle pieces’. If you match up the unpaired electrons, it’s easier to draw circles around the octets…

Predictions… Is there a way to predict ______ how many covalent bonds an element will form? Look at the number of ____ valence electrons you start with. Then figure out how many ______ more are needed to ____ complete your valence shell. That’s the number of _______ bonds that element will form!

Predictions… All elements in group 17 ___ needone ____ more valence electron, so they will one ____ covalent bond. form Group ___ 16 elements have ___, 6 need ___, 2 so they form ___ 2 covalent bonds. Group ___ 15 elements have ___, 5 need ___, 3 so they form ___ 3 covalent bonds.

What’s the difference? How do you know if a formula is a covalent or ionic compound? Look at the ______ types of atoms that make up the compound! Ionic = _______ metal + _____ nonmetal Covalent = _____ nonmetal + _____ nonmetal

What am I? Ca. Cl 2 NO 2 NH 3 ionic covalent Mg. Br 2 ionic

What’s My Name? Remember that for ionic compounds, the ______ metal comes first, then the nonmetal -- which ends in ______. “-ide” _____ Since covalent compounds are made of two ______, nonmetals how do you come up with the name?

What’s My Name? All covalent compounds havetwo ____ names – one for the ______ first element second and one for the _______. What we have to do is add a _______ prefix to tell us how many ______ atoms of each element are ____ present in the compound.
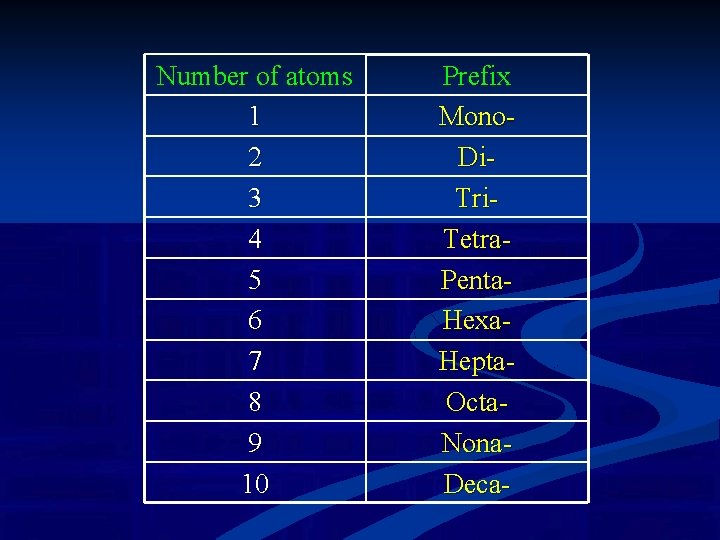
Number of atoms 1 2 3 4 5 6 7 8 9 10 Prefix Mono. Di. Tri. Tetra. Penta. Hexa. Hepta. Octa. Nona. Deca-

Rules for Naming 1. Write the _____ name of the first element. than 1 of the first 2. If there is more _____ element – add the appropriate prefix _______. 3. The second element ALWAYS _____ gets a prefix! 4. Change the end of the second ____ name to ______. “-ide”

Examples CO 2 Carbon dioxide N 2 O 3 H 2 O Dinitrogen trioxide Dihydrogen monoxide
- Slides: 24