Covalent Bonding Covalent Bonds n Covalent bonds occur

Covalent Bonding
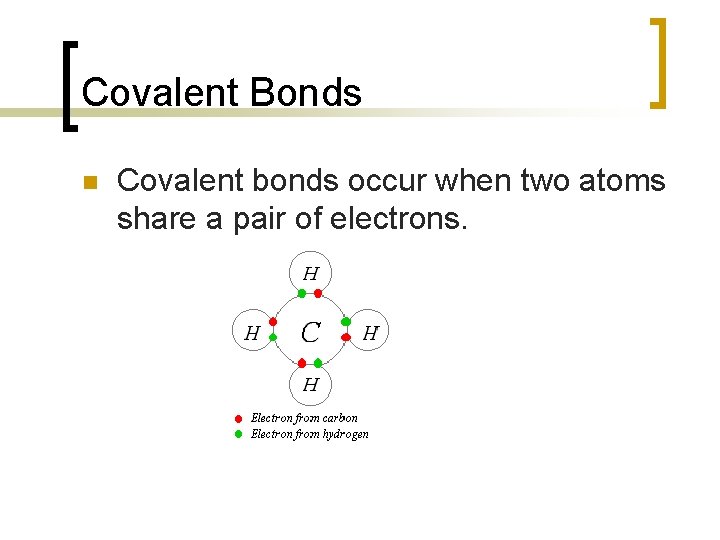
Covalent Bonds n Covalent bonds occur when two atoms share a pair of electrons.

Why does it work? n n Atoms are most stable when the outer electron shell is full – octet rule Some atoms can steal electrons from other atoms – ionic bonds ¡ Metal + nonmetal
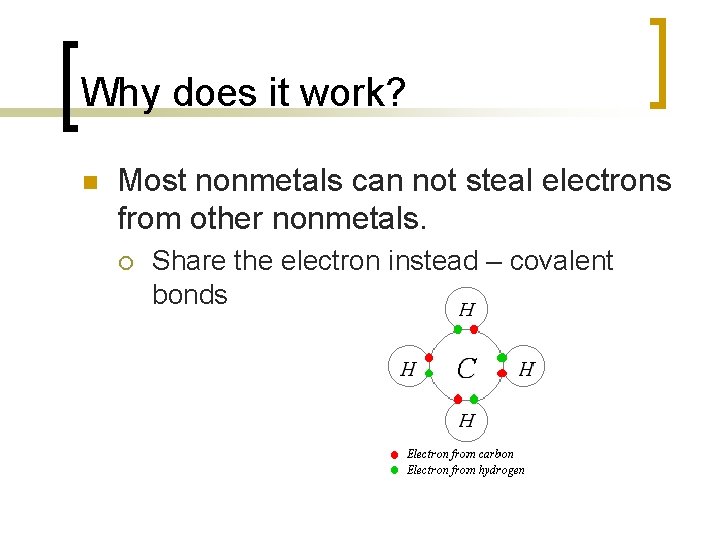
Why does it work? n Most nonmetals can not steal electrons from other nonmetals. ¡ Share the electron instead – covalent bonds

How can we predict which bonds are covalent or ionic? n n n The difference in electronegativity of elements predicts ionic or covalent bonds. If there is 1. 8 or higher difference in electronegativity, the bond will be ionic. Below 1. 8 is covalent.

Determining the number of bonds an atom can form n n The number of valance electrons determines the number of covalent bonds an atom can form. Any valance electron that does not have a pair is free to bond.

Rules of Thumb for Covalent Bonds n Zero or eight valance electrons: ¡ n No bonds can be formed. Four or less valance electrons: ¡ Can form the same number of bonds as valance electrons because all of the electrons are free.

Rules of Thumb for Covalent Bonds n Five to seven valance electrons ¡ The atom can only make bonds with the valance electrons that are not already paired.

Practice Problem 1 n How many covalent bonds can calcium form?

Practice Problem 2 n How many covalent bonds can arsenic form?
- Slides: 10