Covalent Bonding COVALENT BONDS Formed when atoms combine

Covalent Bonding

COVALENT BONDS • Formed when atoms combine to form molecules by sharing one or more outer electrons. • Commonly identified by being two nonmetals.
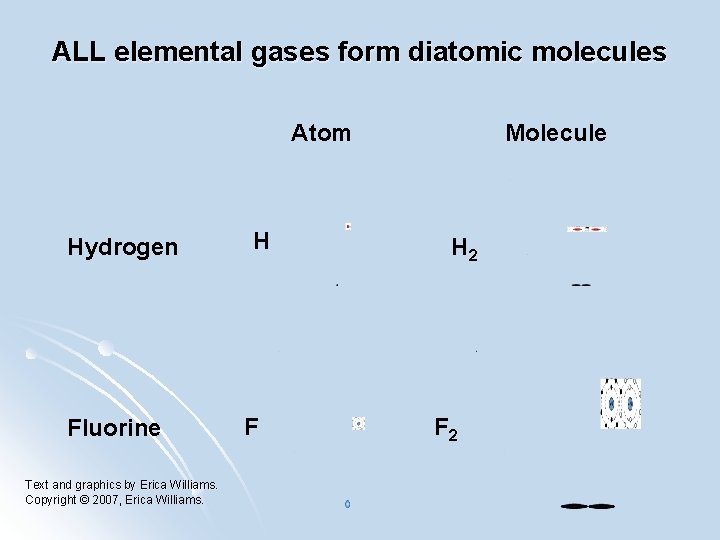
ALL elemental gases form diatomic molecules Atom Hydrogen Fluorine Text and graphics by Erica Williams. Copyright © 2007, Erica Williams. H F Molecule H 2 F 2
Other common gases Ammonia NH 3 Text and graphics by Erica Williams. Copyright © 2007, Erica Williams. Carbon dioxide CO 2 Methane CH 4
Liquids Carbon tetrafluoride CF 4 Text and graphics by Erica Williams. Copyright © 2007, Erica Williams. Methyl alcohol CH 3 OH

Naming Covalent Compounds l We use the naming system for compounds. The prefix naming system tells us how many of each atom is combined in the formula. l For example: CCl 4= Carbon tetrachloride P 2 O 5= Diphosphorus pentoxide
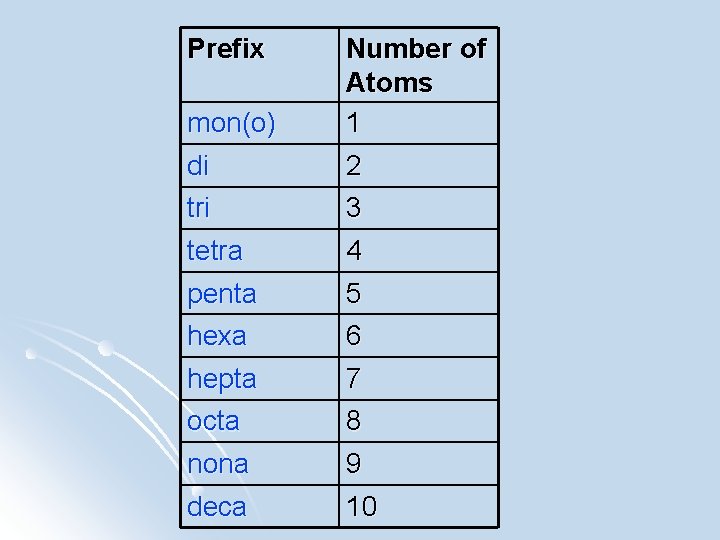
Prefix mon(o) di Number of Atoms 1 2 tri tetra 3 4 penta hexa 5 6 hepta octa 7 8 nona deca 9 10

Rules to follow: 1. Most left element is written first. 2. Prefix is placed before the element’s name. 3. If there is only 1 of the first element, we do not write the mono. Eg. CO, simply write carbon… 4. Write the name of the second element with the appropriate prefix that shows number of atoms are present in that compound. l Eg. CO becomes carbon monoxide

Diatoms All elemental gases form diatomic molecules. Diatomic molecules are composed of only atom. Hydrogen (H 2), Nitrogen (N 2), Oxygen (O 2), Fluorine (F 2), Chlorine (Cl 2), Bromine (Br 2) and Iodine (I 2) These molecules are single atom, all by itself. found as a
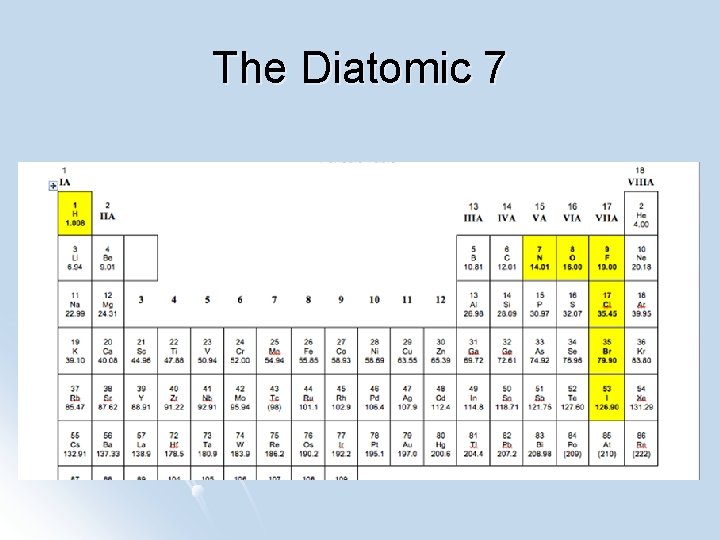
The Diatomic 7

Other examples: CS 2 = carbon disulphide l SO 3= Sulphur trioxide l CCl 4= Carbon tetrachloride l P 2 O 5= Diphosphorus pentoxide l
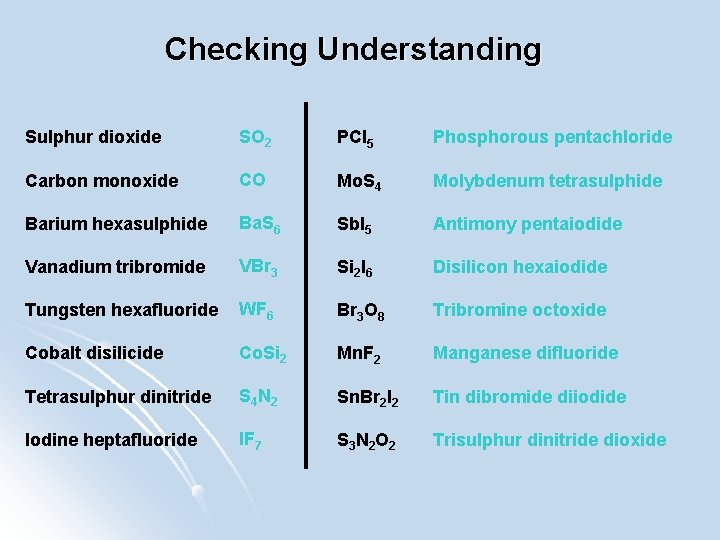
Checking Understanding Sulphur dioxide SO 2 PCl 5 Phosphorous pentachloride Carbon monoxide CO Mo. S 4 Molybdenum tetrasulphide Barium hexasulphide Ba. S 6 Sb. I 5 Antimony pentaiodide Vanadium tribromide VBr 3 Si 2 I 6 Disilicon hexaiodide Tungsten hexafluoride WF 6 Br 3 O 8 Tribromine octoxide Cobalt disilicide Co. Si 2 Mn. F 2 Manganese difluoride Tetrasulphur dinitride S 4 N 2 Sn. Br 2 I 2 Tin dibromide diiodide Iodine heptafluoride IF 7 S 3 N 2 O 2 Trisulphur dinitride dioxide

The End
- Slides: 13