COVALENT BONDING Covalent Bonding Many compounds do not

COVALENT BONDING

Covalent Bonding � Many compounds do not form ionic bonds. These compounds contain two or more nonmetallic atoms. � For example, CO 2 is made of two different non-metals, carbon and oxygen. These compounds are formed through the sharing of valence electrons.
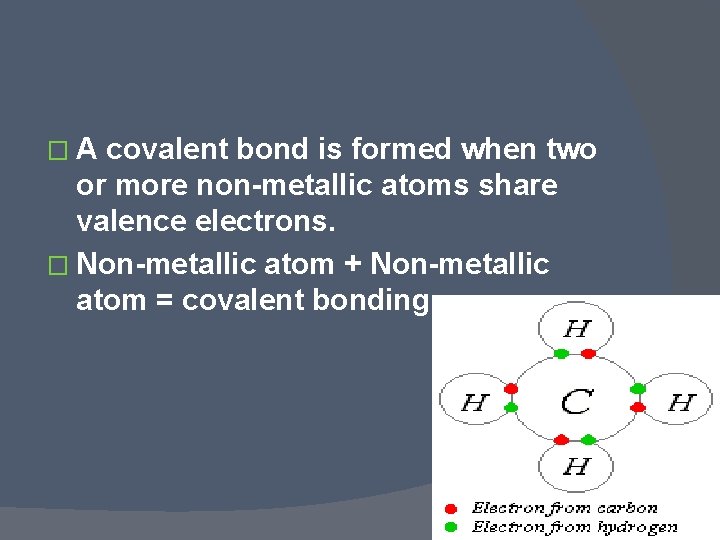
� A covalent bond is formed when two or more non-metallic atoms share valence electrons. � Non-metallic atom + Non-metallic atom = covalent bonding

Compounds vs. Molecules � Two non-metallic elements combine to form covalent compounds (or bonds). Compounds contain at least two different types of atoms. � A molecule is the smallest unit of a covalent compound. � A molecule of a compound has different characteristic properties than the atoms which form it.

Example � Two hydrogen atoms form a covalent bond by sharing electrons to produce a hydrogen molecule. A hydrogen molecule still possesses the properties of hydrogen. No new substance is formed since only one type of atom, H, is present.
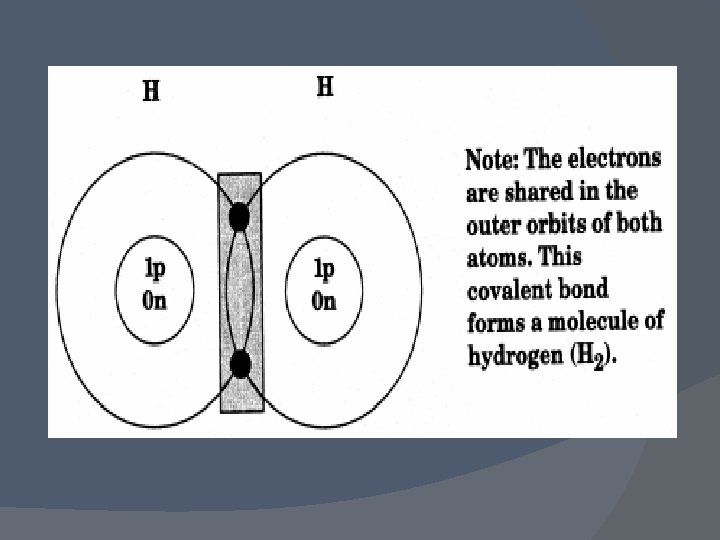

� By sharing their electrons, both atoms are satisfied they have filled outer orbits (the outer orbits are the same as for helium) and are stable. � The two hydrogen atoms form a diatomic molecule (i. e. , two atoms of hydrogen share electrons to make a single molecule of hydrogen gas).

The elements forming diatomic gases are unstable as single atoms and combine almost instantaneously to form stable molecules. � Note that diatomic molecules are still classified as elements even though they are molecules. While it may seem strange that a molecule is called an element rather than a compound, remember that diatomic molecules are made of only one kind of atom. �

Name of Element Formula or Symbol for one molecule one atom of of the element Hydrogen H H 2(gas) Nitrogen N N 2(gas) Oxygen O O 2(gas) Fluorine F F 2(gas) Chlorine Cl Cl 2(gas) Bromine Br Br 2(liquid) Iodine I I 2(solid)

HINT!!!! � If the diatomic elements are placed in a different order the symbols spell out a word that can help you remember these diatomic elements. � Remember “Count HOFBr. INCl”, pronounced as “Count Hoffbrinkle”. If you remember “Count HOFBr. INCl” you will never forget which elements are diatomic

Organic Compounds � Covalent bonds can also be classified as organic compounds. This is when 2 non-metallic elements combine together. � Example: e. g. CO , C H O

Diatomic or Organic Compound? Example – If the element was on it’s own. . . Fluorine Carbon Sulphur Hydrogen Helium Chlorine Oxygen Forms a Diatomic or Organic Compound?

Octet Rule The only way that two non-metallic atoms can satisfy their need to gain electrons when they bond is through sharing valence electrons. The sharing of electrons between two non-metallic atoms allows each atom to have a complete octet of electrons at least part of the time. � The shared electrons are attracted at the same time to each nucleus in each atom. This is the “glue” that holds the atoms together. � A covalent bond is formed when two atoms share one or more pairs of electrons in order to obtain a complete octet of electrons. �
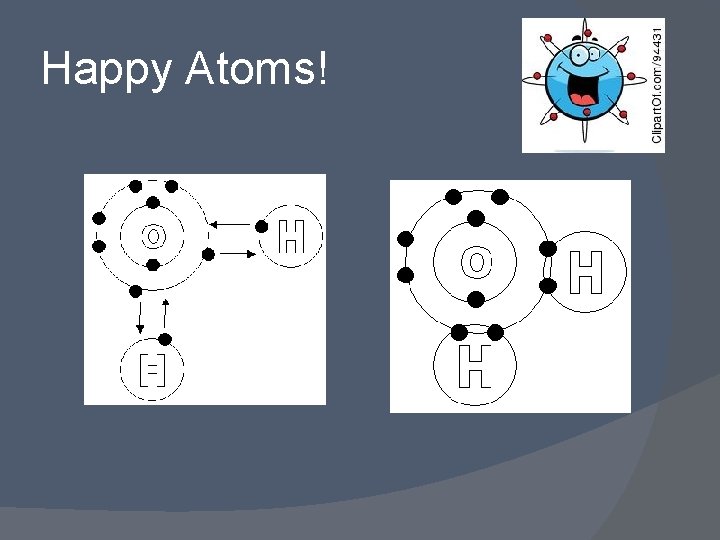
Happy Atoms!
- Slides: 14