Covalent Bonding Covalent Bonding Electrons are shared between

Covalent Bonding
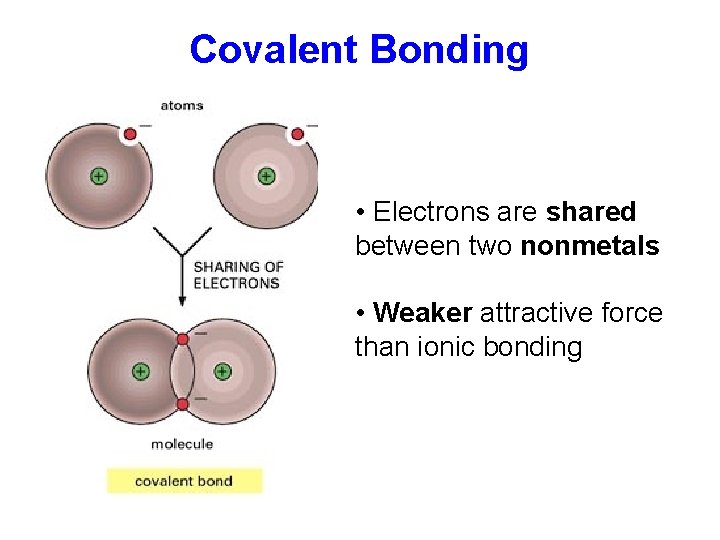
Covalent Bonding • Electrons are shared between two nonmetals • Weaker attractive force than ionic bonding

Properties of Molecular Compounds State at SATP Structure Typically gases & liquids, but some solids Variable (waxy to brittle) Melting & Boiling Points Low Electrical Conductivity Nonelectrolyte Solubility in Water Variable (low to high) Confused? ? Don’t worry…more answers to come
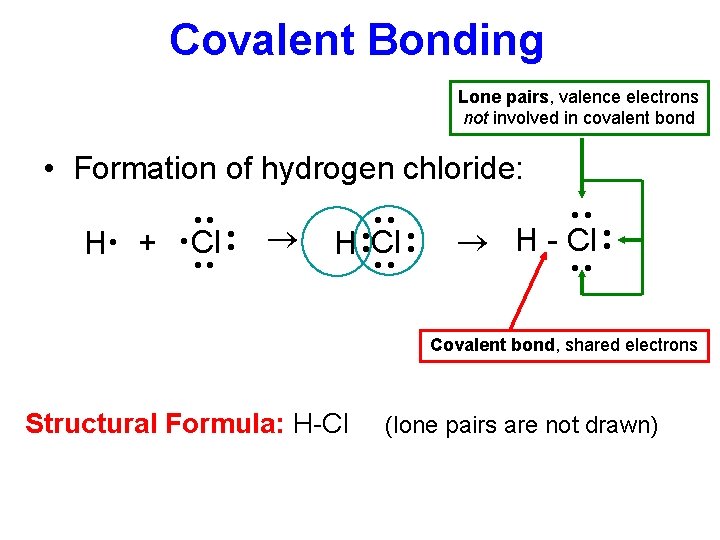
Covalent Bonding Lone pairs, valence electrons not involved in covalent bond • Formation of hydrogen chloride: H Cl ·· ·· ·· ® ·· · ·· Cl ·· ·· ® H - Cl ·· H· + ·· ·· Covalent bond, shared electrons Structural Formula: H-Cl (lone pairs are not drawn)
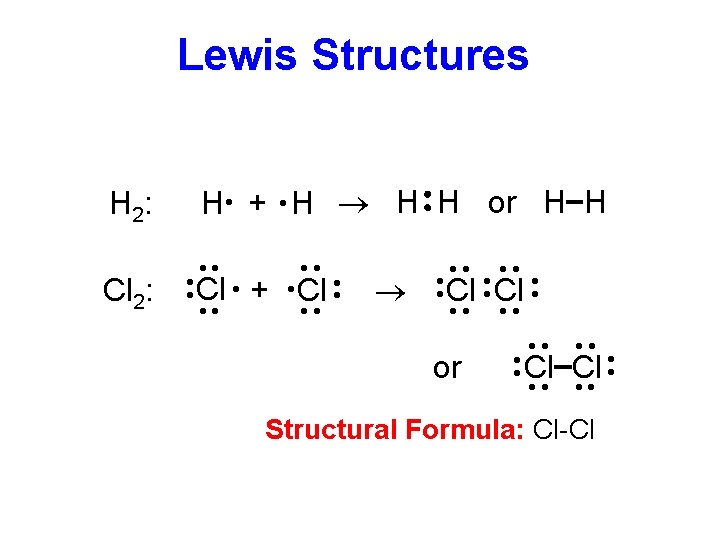
Lewis Structures H · + · H ® H H or H H ·· ·· ·· Cl Cl ·· ·· Structural Formula: Cl-Cl ·· or ·· Cl Cl ·· ·· · · ® ·· ·· ·· Cl + Cl ·· ·· ·· Cl 2: ·· ·· H 2:
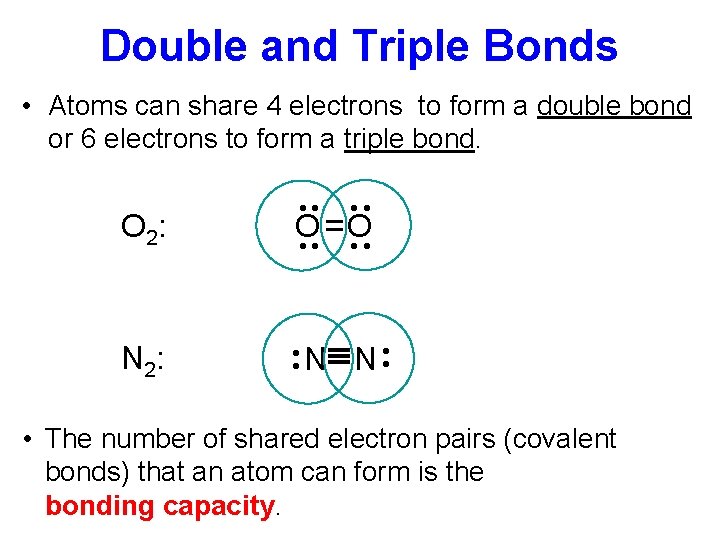
Double and Triple Bonds • Atoms can share 4 electrons to form a double bond or 6 electrons to form a triple bond. ·· ·· N 2: N N ·· O =O ·· ·· ·· O 2: • The number of shared electron pairs (covalent bonds) that an atom can form is the bonding capacity.
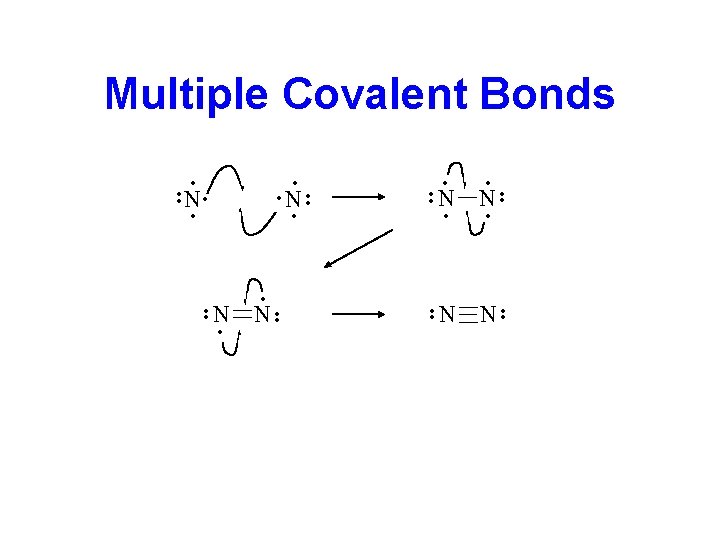
Multiple Covalent Bonds • • • • N N • • • N • • • •


Drawing Lewis Structures 1. Arrange the element symbols. • Central atoms are generally those with the highest bonding capacity. • Carbon atoms are always central atoms • Hydrogen atoms are always peripheral atoms 2. Add up the number of valence electrons from all atoms. • For polyatomic ions, add one electron for each negative charge and subtract one for each positive charge. 3. Draw a skeleton structure with atoms attached by single bonds. 4. Complete the octets of peripheral atoms. 5. Place extra electrons on the central atom. 6. If the central atom doesn’t have an octet, try forming multiple bonds by moving lone pairs.

Practice Problems • Examples – Methanal CH 2 O – Chlorate (polyatomic ion) • p. 77 -81 #1 -15

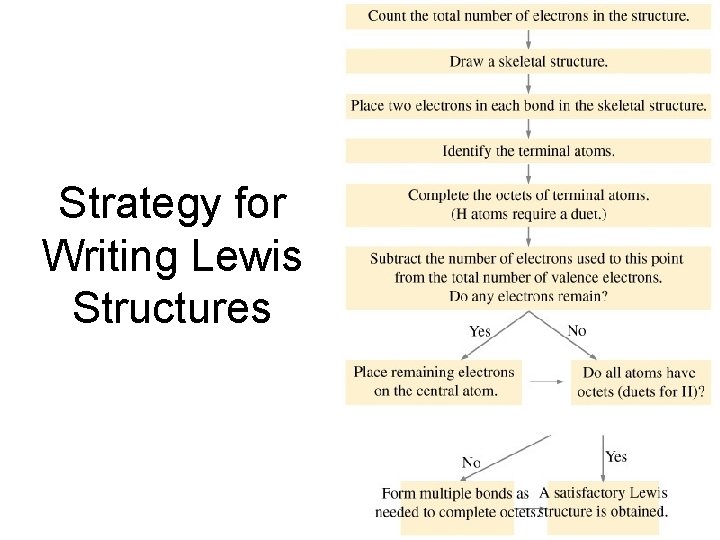
Strategy for Writing Lewis Structures
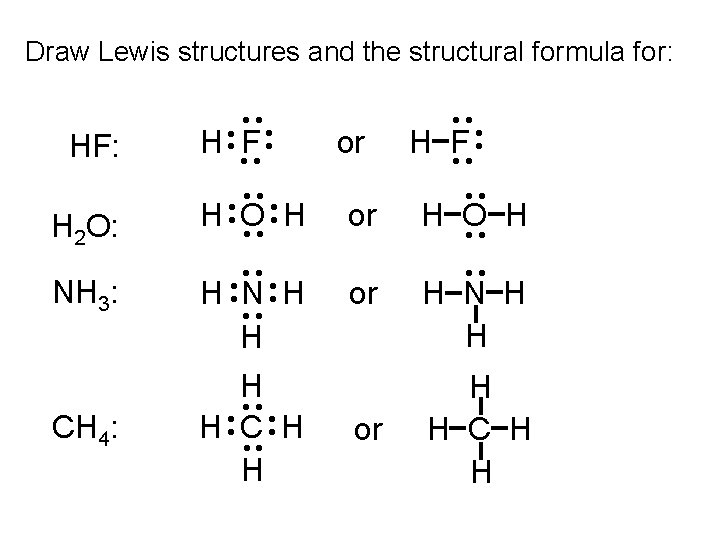
Draw Lewis structures and the structural formula for: CH 4: ·· ·· ·· H N H ·· H C H ·· H or or ·· H F ·· ·· ·· H O H ·· ·· NH 3: ·· ·· H 2 O: H ·F· ·· HF: ·· ·· H O H ·· ·· H N H H C H H
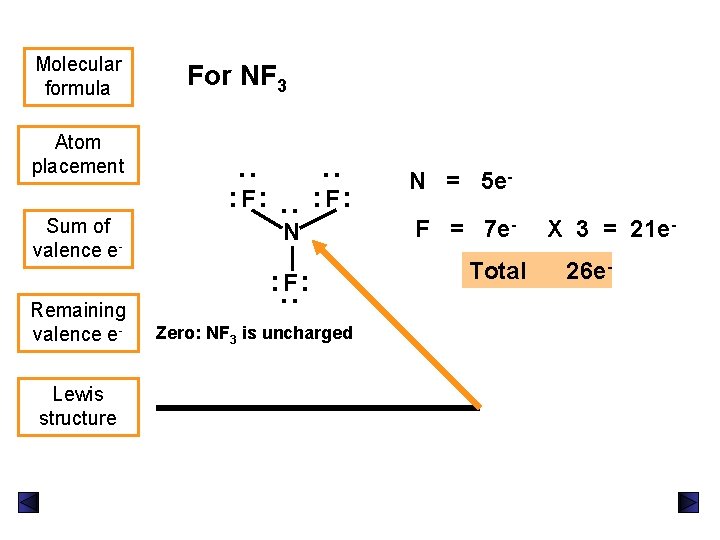
Sum of valence e- : : F: : Atom placement For NF 3 : Molecular formula N Remaining valence e. Lewis structure : : F: Zero: NF 3 is uncharged N = 5 e. F = 7 e. Total X 3 = 21 e 26 e-

SAMPLE PROBLEM 10. 1 Write a Lewis structure for CCl 2 F 2, one of the compounds responsible for the depletion of stratospheric ozone. SOLUTION: : Cl : : Cl C F: : F: : Make bonds and fill in remaining valence electrons placing 8 e- around each atom. F F : Steps 2 -4: C has 4 valence e-, Cl and F each have 7. The sum is 4 + 4(7) = 32 valence e-. Cl C : Step 1: Carbon has the highest bonding capacity and is the central atom. The other atoms are placed around it. Cl : PROBLEM: Writing Lewis Structures for Molecules with One Central Atom

SAMPLE PROBLEM 10. 2 Hydrogen can have only one bond so C and O must be next to each other with H filling in the bonds. There are 4(1) + 4 + 6 = 14 valence e-. C has 4 bonds and O has 2 pair of nonbonding e-. H : SOLUTION: Write the Lewis structure for methanol (molecular formula CH 4 O), an important industrial alcohol that is being used as a gasoline alternative in car engines. H C O : PROBLEM: Writing Lewis Structure for Molecules with More than One Central Atom H H
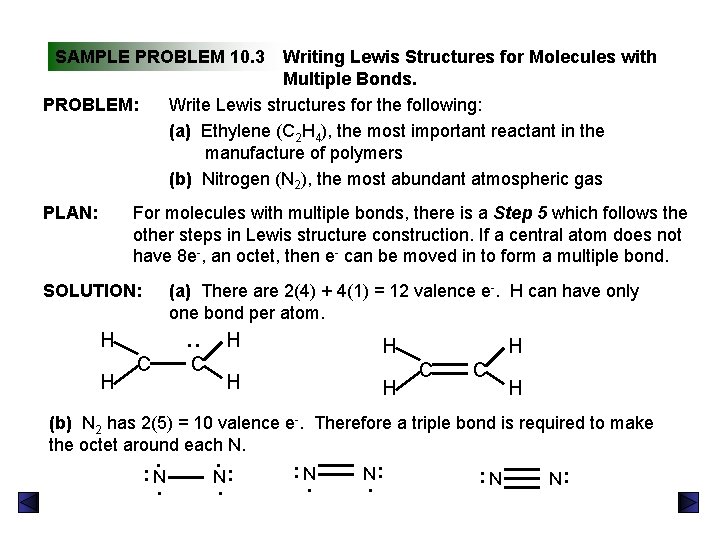
SAMPLE PROBLEM 10. 3 Writing Lewis Structures for Molecules with Multiple Bonds. Write Lewis structures for the following: (a) Ethylene (C 2 H 4), the most important reactant in the manufacture of polymers (b) Nitrogen (N 2), the most abundant atmospheric gas PROBLEM: PLAN: For molecules with multiple bonds, there is a Step 5 which follows the other steps in Lewis structure construction. If a central atom does not have 8 e-, an octet, then e- can be moved in to form a multiple bond. SOLUTION: (a) There are 2(4) + 4(1) = 12 valence e-. H can have only one bond per atom. H H : H C C H (b) N 2 has 2(5) = 10 valence e-. Therefore a triple bond is required to make the octet around each N. N. : : : . N N : . : N.

Polyatomic Ions • Many compounds contain a combination of covalent and ionic bonds. • E. g. Na. OH • OH- is a polyatomic ion with a covalent bond between O and H • Ionic bond between Na+ and OH-

Coordinate Covalent Bonds A covalent bond in which both of the shared electrons come from the same atom. E. g. NH 3 (ammonia) and H+ (hydrogen ion) to form NH 4 (ammonium)
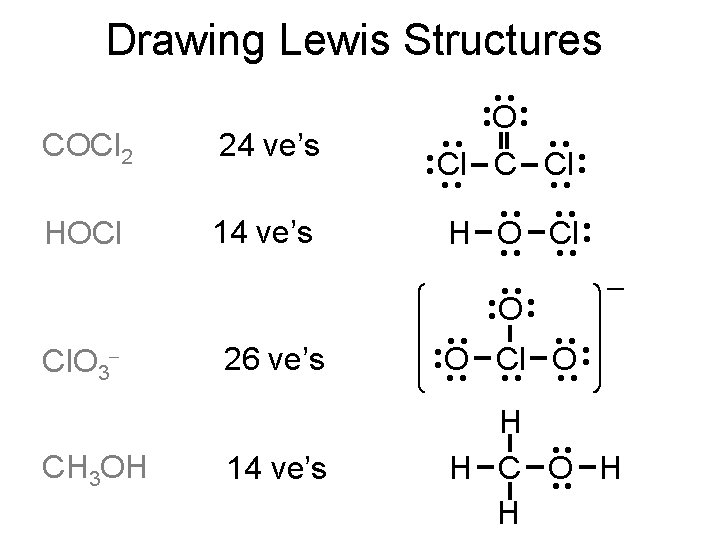
Drawing Lewis Structures ·· ·· ·· 14 ve’s ·· H O Cl ·· ·· ·· HOCl 24 ve’s ·· COCl 2 ·· ·· O ·· ·· Cl C Cl ·· ·· ·· CH 3 OH 14 ve’s ·· O Cl O ·· ·· 26 ve’s ·· Cl. O 3 ·· ·· ·· O H ·· H C O H ·· H
- Slides: 20