Covalent Bonding Chapter 8 8 1 Molecular Compounds

Covalent Bonding Chapter 8

8 -1 Molecular Compounds Learning Target(s): 1. To learn how atoms can share electrons to form a covalent bond. 2. To learn how atoms join to form units called molecules. Success Criteria: Upon the completion of the lesson, I will be able to… 1. Distinguish between the melting points and boiling points of molecular compounds and ionic compounds. 2. Describe the information a molecular formula provides.

8 -1 Molecular Compounds Molecules and Molecular Compounds: • So far, we have learned about pure elements, monatomic noble gases, and ionic compounds (including salts). – Ionic compounds have pretty predictable properties. • They are crystalline solids with high melting points. – Other compounds have very different properties from one another. • Hydrogen chloride is a gas at room temperature, while water is a liquid. – The properties cannot be explained by ionic bonding. – The atoms of these molecules are held together with covalent bonds. » Covalent bonds form from the sharing of electrons.
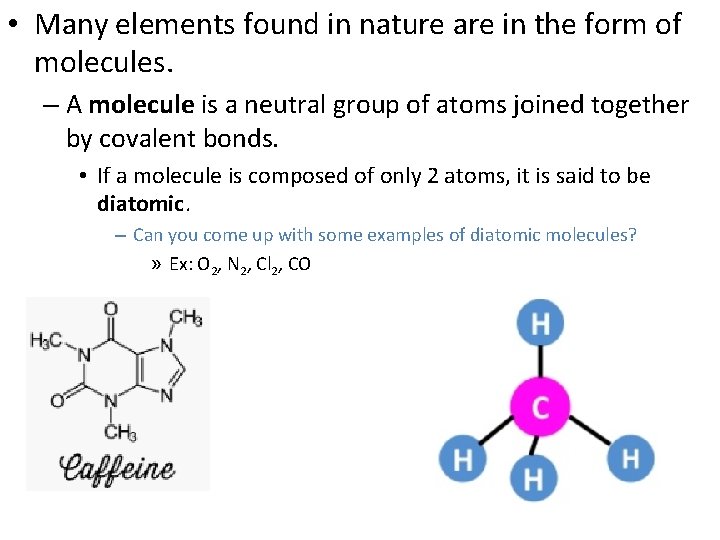
• Many elements found in nature are in the form of molecules. – A molecule is a neutral group of atoms joined together by covalent bonds. • If a molecule is composed of only 2 atoms, it is said to be diatomic. – Can you come up with some examples of diatomic molecules? » Ex: O 2, N 2, Cl 2, CO

• Molecular compounds are compounds composed of molecules. – In a molecular compound, the molecules are all the same. • Ex: Water – Molecular compounds tend to have lower melting points and boiling points than ionic compounds. – Molecular compounds also tend to be made of only nonmetals and are often liquids or gases at room temperature.

T Chart Time! – Make a t-chart that compares and contrasts between ionic and molecular compounds. • Include: Types of atoms it’s made from, boiling point, melting point, state of matter, and type of bond. Ionic Compound Molecular Compound

Molecular Formulas: • A molecular formula is the chemical formula of a molecular compound. – Molecular formulas show many atoms of each element a molecule contains. – Examples: • H 2 0 • CO 2 • C 6 H 12 O 6
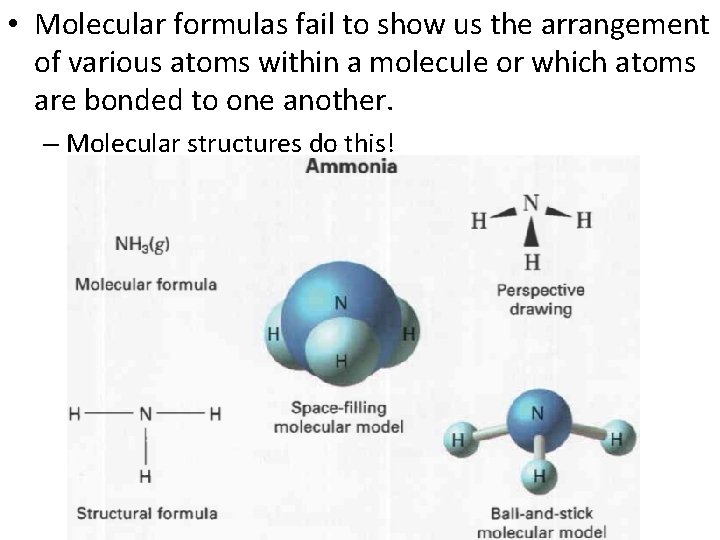
• Molecular formulas fail to show us the arrangement of various atoms within a molecule or which atoms are bonded to one another. – Molecular structures do this!

Learning Checkpoint 1. How are the melting points and boiling points of molecular compounds usually different from ionic compounds? 2. What information does a molecular formula provide? 3. What are the only elements that exist in nature as uncombined atoms? What term is used to describe such elements? 4. Describe how the molecule whose formula is NO is different from the molecule whose formula is N 2 O. 5. Give an example of a diatomic molecule found in Earth’s atmosphere. 6. What information does a molecule’s molecular structure give? 7. Look at the list of formulas below. Are there any that do not exist? Which are ionic compounds? Which formulas represent molecules? Which formulas represent compounds? How many atoms are there in SO 3? Br 2, KBr, CH 4, SO 3, N 2 H 2, Ne 2

8 -2 The Nature of Covalent Bonding Learning Target(s): 1. To learn how oxygen atoms can join in pairs to form the oxygen you breathe and can also join in groups of three oxygen atoms to form ozone in the atmosphere. Success Criteria: Upon the completion of this lesson, I will be able to… 1. Describe how electrons are shared to form covalent bonds and identify exceptions to the octet rule. 2. Demonstrate how electron dot structures represent shared electrons. 3. Describe how atoms form double or triple covalent bonds. 4. Distinguish between a covalent bond a coordinate covalent bond and describe how the strength of a covalent bond is related to its bond dissociation energy. 5. Describe how oxygen atoms are bonded in ozone.

8 -2 The Nature of Covalent Bonding The Octet Rule in Covalent Bonding: • Similar to ionic compound formation, covalent bonds also tend to follow the octet rule. – In forming covalent bonds, electron sharing usually occurs so that atoms attain the electron configurations of noble gases. • Nonmetallic atoms usually acquire a total of 8 electrons by sharing electrons.
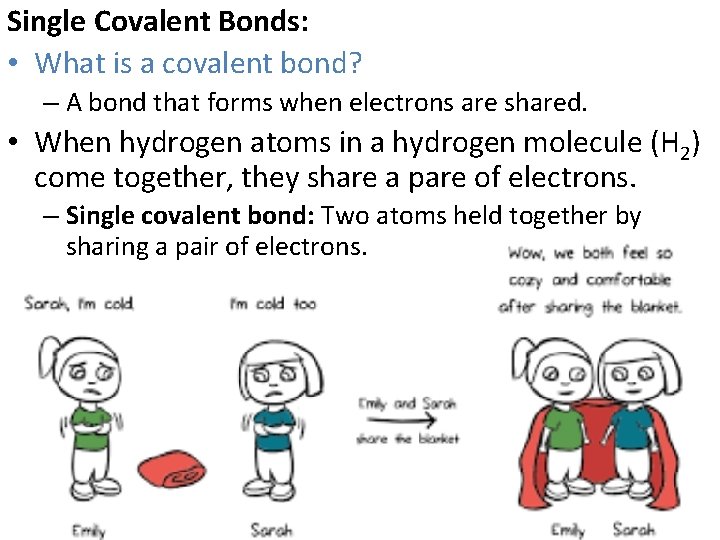
Single Covalent Bonds: • What is a covalent bond? – A bond that forms when electrons are shared. • When hydrogen atoms in a hydrogen molecule (H 2) come together, they share a pare of electrons. – Single covalent bond: Two atoms held together by sharing a pair of electrons.
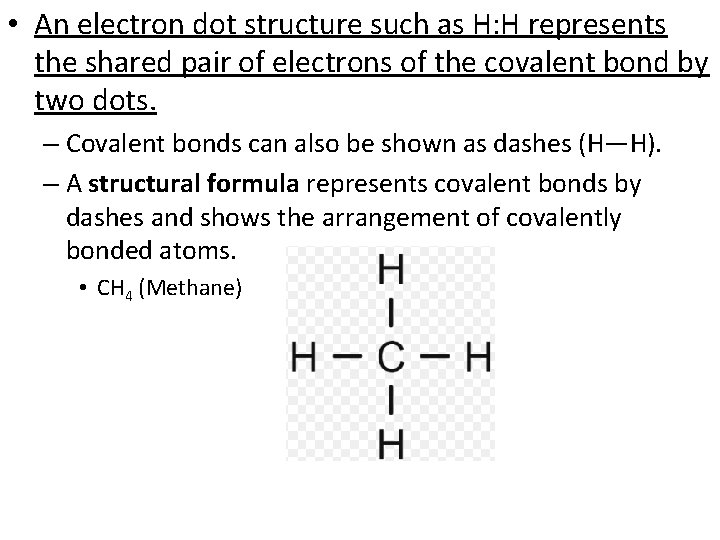
• An electron dot structure such as H: H represents the shared pair of electrons of the covalent bond by two dots. – Covalent bonds can also be shown as dashes (H—H). – A structural formula represents covalent bonds by dashes and shows the arrangement of covalently bonded atoms. • CH 4 (Methane)

• All halogens form single covalent bonds in their diatomic molecules. – Let’s look at Fluorine. • Each fluorine atom in this diatomic molecule has 7 valence electrons. – Each one will share a single electron with the other. – The other 6 electrons for each fluorine atom remain in pairs that are not shared. » These are called unshared pairs, or “lone pairs. ”
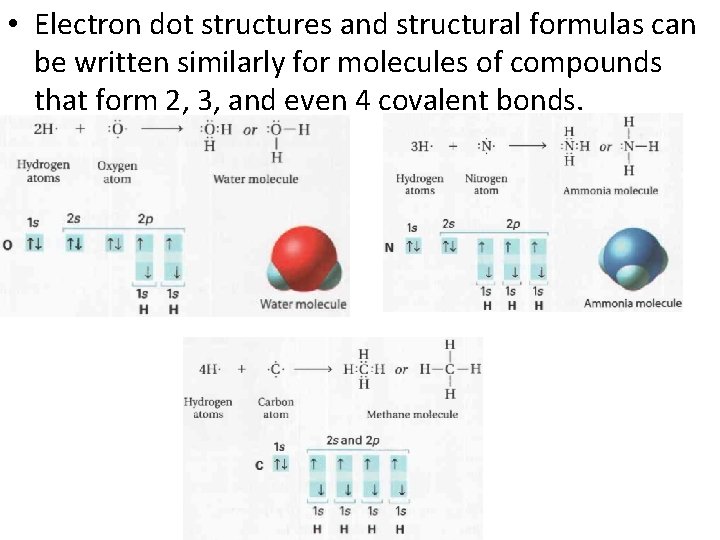
• Electron dot structures and structural formulas can be written similarly for molecules of compounds that form 2, 3, and even 4 covalent bonds.

• Did you notice something odd about carbon and its 4 covalent bonds? • Let’s go back a slide and look… – Carbon only has 2 valence electrons in its 2 p sublevel. • However, CH 2 is unstable. – For a small cost in energy, carbon can promote a 2 s electron to the empty 2 p sublevel. • Now there are 4 orbitals with only 1 electron and each is able to bond. – CH 4 is far more stable!

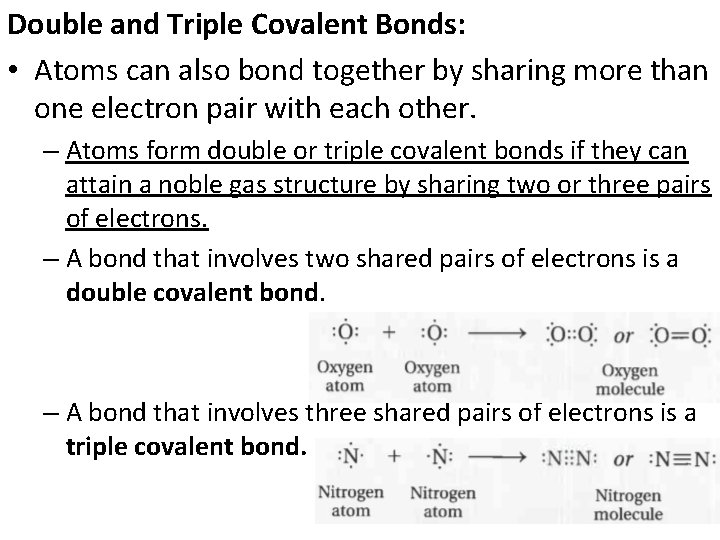
Double and Triple Covalent Bonds: • Atoms can also bond together by sharing more than one electron pair with each other. – Atoms form double or triple covalent bonds if they can attain a noble gas structure by sharing two or three pairs of electrons. – A bond that involves two shared pairs of electrons is a double covalent bond. – A bond that involves three shared pairs of electrons is a triple covalent bond.

• Up to this point, all of our single and triple covalent bonds have been described with diatomic molecules. – It is important to note that they also exist between unlike atoms. – Try drawing structural formulas for these: • Acetylene (C 2 H 2) • Carbon Dioxide (CO 2)

Coordinate Covalent Bonds: • Let’s take a look at an odd form of covalent bond… • In carbon monoxide, carbon and oxygen bond together. – Oxygen only needs two electrons to achieve noble gas status, but carbon needs four… • So that both atoms can be happy, oxygen donates one of its unshared pairs! – A coordinate covalent bond is a covalent bond in which one atom contributes both bonding electrons. » In a structural formula, this is represented as an arrow pointing from the atom that donated the unshared pair of electrons. » In a coordinate covalent bond, the shared electron pair comes from one of the bonding atoms.
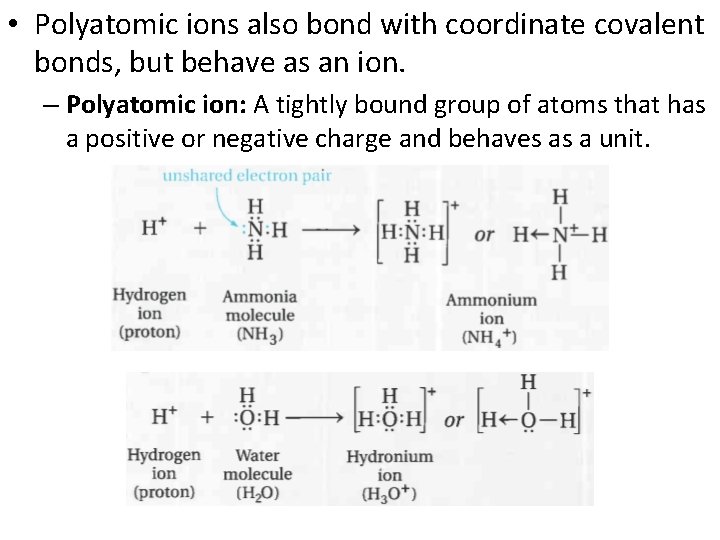
• Polyatomic ions also bond with coordinate covalent bonds, but behave as an ion. – Polyatomic ion: A tightly bound group of atoms that has a positive or negative charge and behaves as a unit.

Bond Dissociation Energies: • When hydrogen atoms combine to form a hydrogen molecule, a large quantity of heat is released. – This means the product is far more stable than the reactants. • In fact, it takes 435 k. J of energy to break apart all of the bonds in 1 mole (6. 02 x 1023 atoms-more on this later). – Bond dissociation energy is the energy required to break the bond between two covalently bonded atoms. • A large bond dissociation energy corresponds to a strong covalent bond. – Triple covalent bonds are stronger than double covalent bonds, which are still stronger than single covalent bonds.

Resonance: • Sometimes, structural formulas can be written in more than one way. – A resonance structure is a structure that occurs when it is possible to write two or more valid electron dot formulas that have the same number of electron pairs for a molecule or ion. • Ozone is a pretty good example of this. *Notice the two-way arrow, which represents resonance.

Exceptions to the Octet Rule: • For some molecules or ions, it is impossible to draw electron dot structures that satisfy the octet rule. – The octet rule cannot be satisfied in molecules whose total number of valence electrons is an odd number. There also molecules in which an atom has fewer, or more, than a complete octet of valence electrons.

1. 2. 3. 4. 5. 6. 7. 8. 9. Learning Checkpoint: What electron configurations do atoms usually achieve by sharing electrons to form covalent bonds? How is an electron dot structure used to represent a covalent bond? When are two atoms likely to form a double covalent bond between them? Triple bond? How is a coordinate covalent bond different from other covalent bonds? How is the strength of a covalent bond related to its bond dissociation energy? Draw the electron dot resonance structures for ozone and explain how they describe its bonding. List three ways in which the octet rule can sometimes fail to be obeyed. What kinds of information does a structural formula reveal about the compound it represents? Draw electron dot structures for the following molecules, which have only single covalent bonds. 1. H 2 S 2. PH 3 3. Cl. F 10. Use the bond dissociation energies of H 2 (435 k. J/mol) and of a typical carbon-carbon bond (347 k. J/mol) to decide which bond is stronger. Explain your reasoning.

8 -3 Bonding Theories Learning Target(s): 1. To learn how attractive and repulsive forces influence the shapes of molecules. Success Criteria: Upon the completion of this lesson, I will be able to… 1. Describe the relationship between atomic and molecular orbitals. 2. Describe how VSEPR theory helps predict the shapes of molecules. 3. Identify ways in which orbital hybridization is useful in describing molecules.

8 -3 Bonding Theories Molecular Orbitals: • So far, we have been simplifying the way we portray covalent bonds in molecules. – We’ve been assuming that the orbitals are those of the individual atoms. • There is a quantum mechanical model of bonding that shows the electrons in molecules using orbitals that only exist in groupings of atoms. – Molecular orbitals are orbitals that apply to the entire molecule. • Just as an atomic orbital belongs to a particular atom, a molecular orbital belongs to a molecule as a whole. – Similarly, two electrons are required to fill a molecular orbital. » A molecular orbital that can be occupied by two electrons of a covalent bond is called a bonding orbital.
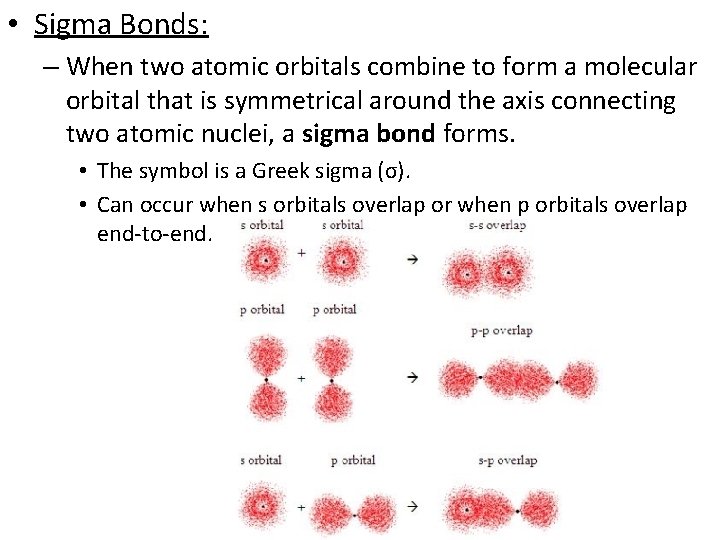
• Sigma Bonds: – When two atomic orbitals combine to form a molecular orbital that is symmetrical around the axis connecting two atomic nuclei, a sigma bond forms. • The symbol is a Greek sigma (σ). • Can occur when s orbitals overlap or when p orbitals overlap end-to-end.
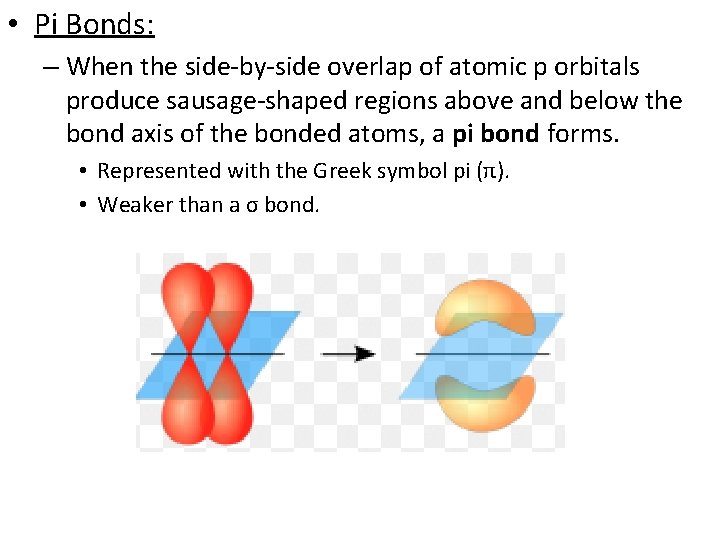
• Pi Bonds: – When the side-by-side overlap of atomic p orbitals produce sausage-shaped regions above and below the bond axis of the bonded atoms, a pi bond forms. • Represented with the Greek symbol pi (π). • Weaker than a σ bond.

VSEPR Theory: • Just like a picture or drawing of you may fail to do justice to the way you actually look (they never show me as flawlessly as I actually am), electron dot structures and structural formulas fail to represent how a molecule looks in 3 -D. • The Valence-Shell Electron-Pair Repulsion Theory (VSEPR Theory) explains the 3 -D shape of molecules. – VSEPR Theory tells us that the repulsion between electron pairs causes molecular shapes to adjust so that the valenceelectron pairs stay as far apart as possible. – Why do they do that?
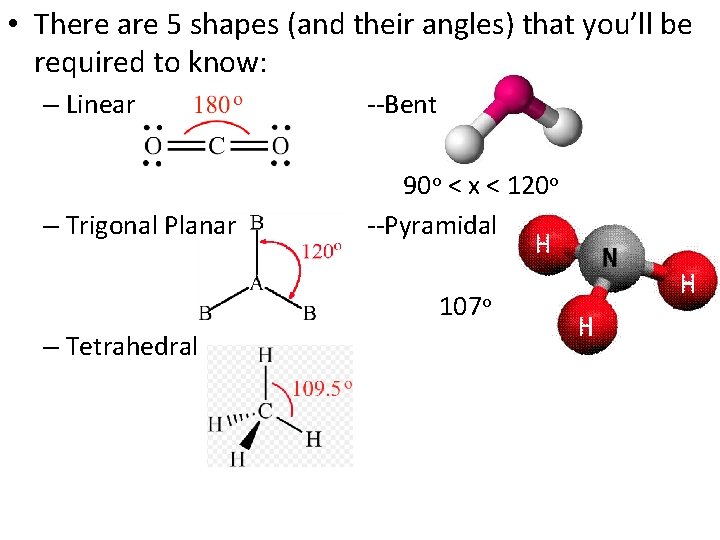
• There are 5 shapes (and their angles) that you’ll be required to know: – Linear --Bent – Trigonal Planar 90 o < x < 120 o --Pyramidal 107 o – Tetrahedral

• Additional VSEPR Theory Support: – Watch the first 3! – Intro. to VSEPR – VSEPR Practice Problems – VSEPR Theory: Common Mistakes – Extra Stuff…(Not required) – Trigonal Bipyramidal Family – Octahedral Family – VSEPR Practice Problems (Advanced) • Quiz E. C. may be based off of this one!

Hybrid Orbitals: • VSEPR theory is great for describing molecular shape, but does a poor job of identifying the types of bonds that formed. – Orbital hybridization provides information about both molecular bonding and molecular shape. – In hybridization, several atomic orbitals mix to form the same total number of equivalent hybrid orbitals.
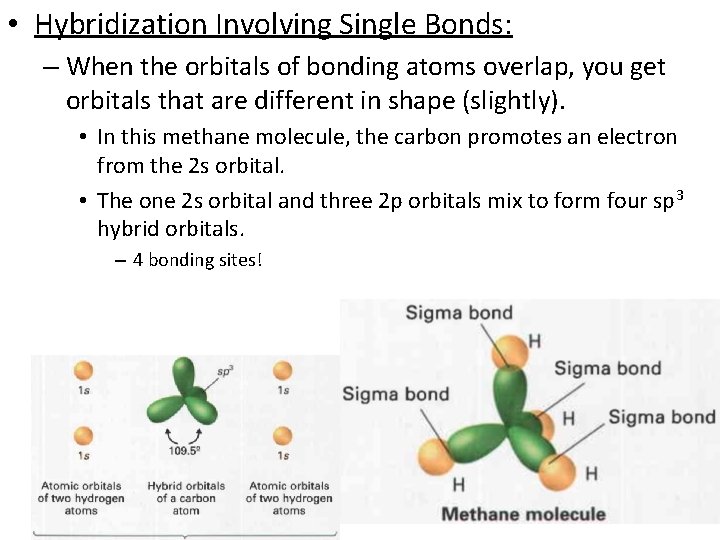
• Hybridization Involving Single Bonds: – When the orbitals of bonding atoms overlap, you get orbitals that are different in shape (slightly). • In this methane molecule, the carbon promotes an electron from the 2 s orbital. • The one 2 s orbital and three 2 p orbitals mix to form four sp 3 hybrid orbitals. – 4 bonding sites!
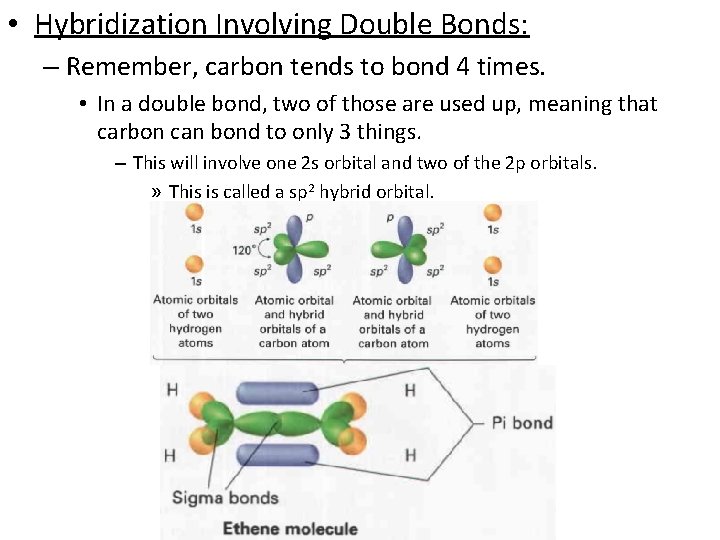
• Hybridization Involving Double Bonds: – Remember, carbon tends to bond 4 times. • In a double bond, two of those are used up, meaning that carbon can bond to only 3 things. – This will involve one 2 s orbital and two of the 2 p orbitals. » This is called a sp 2 hybrid orbital.
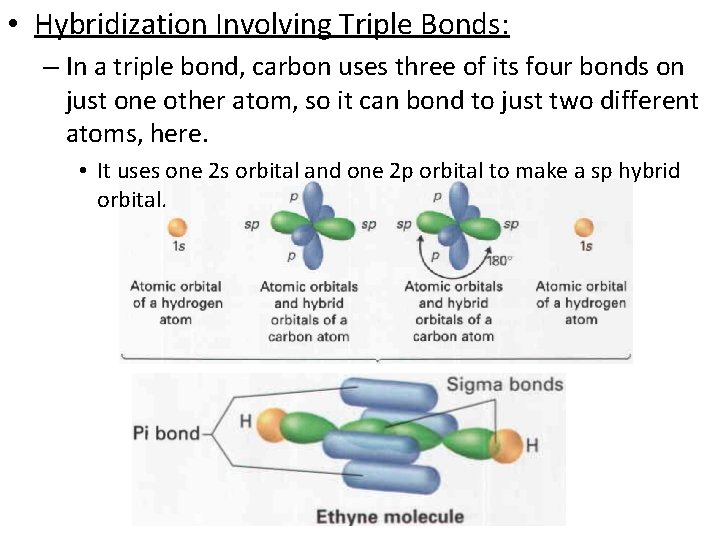
• Hybridization Involving Triple Bonds: – In a triple bond, carbon uses three of its four bonds on just one other atom, so it can bond to just two different atoms, here. • It uses one 2 s orbital and one 2 p orbital to make a sp hybrid orbital.
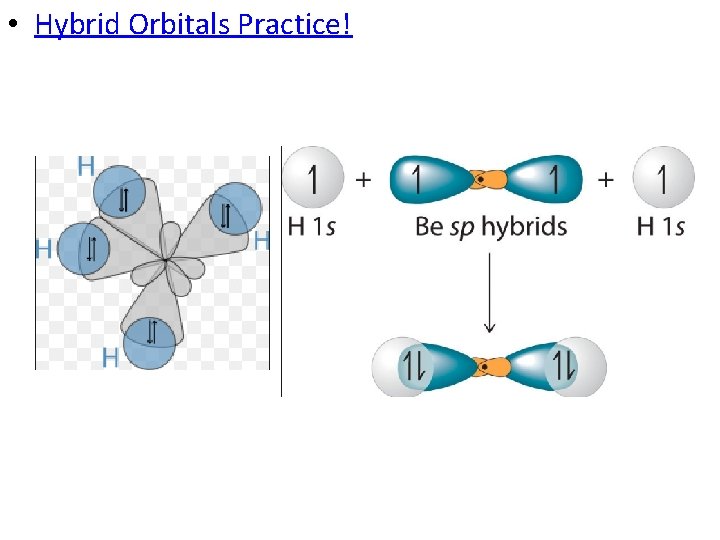
• Hybrid Orbitals Practice!

Learning Checkpoint: 1. How are atomic and molecular orbitals related? 2. Explain how the VSEPR theory can be used to predict the shapes of molecules. 3. How is orbital hybridization useful in describing molecules? 4. What shape would you expect a simple carbon-containing compound to have if the carbon atom has the following hybridizations? a. sp 2 b. sp 3 c. sp 5. What is a sigma bond? Describe, with the aid of a diagram, how the overlap of two half-filled 1 s orbitals produces a sigma bond. 6. How many sigma bonds and how many pi bonds are in an ethyne molecule (C 2 H 2)? 7. The BF 3 molecule is trigonal planar. The attachment of a fluoride ion to the boron in BF 3, through a coordinate covalent bond, creates the BF 4 - ion. What is the geometric shape of this ion?

8 -4 Polar Bonds and Molecules Learning Target(s): 1. To learn that the polar bonds in water molecules influence their behavior, including the distinctive geometry of snowflakes. Success Criteria: Upon the completion of this lesson, I will be able to… 1. Describe how electronegativity values determine the distribution of charge in a polar molecule. 2. Describe what happens to polar molecules when they are placed between oppositely charged metal plates. 3. Evaluate the strength of intermolecular attractions compared with the strength of ionic and covalent bonds. 4. Identify the reason why network solids have high melting points.

8 -4 Polar Bonds and Molecules Bond Polarity: • As if covalent bonds weren’t already bad enough, how would you feel if I told you there was more? • Covalent bonds differe in terms of how the bonded atoms share the lectrons. – Bond character depends on the kind and number of atoms joined together…

• When atoms are the same size as one another, electrons are shared equally. – Both atoms are stable and happy! – Nonpolar covalent bonds form when electrons are shared equally. • This is most often the case in diatomic molecules (H 2, O 2, etc. ).

• A polar covalent bond forms when atoms do not equally share electrons. – The more electronegative atom attracts electrons more strongly and gains a slight negative (δ-) charge. The less electronegative atom has a slightly positive (δ+) charge.

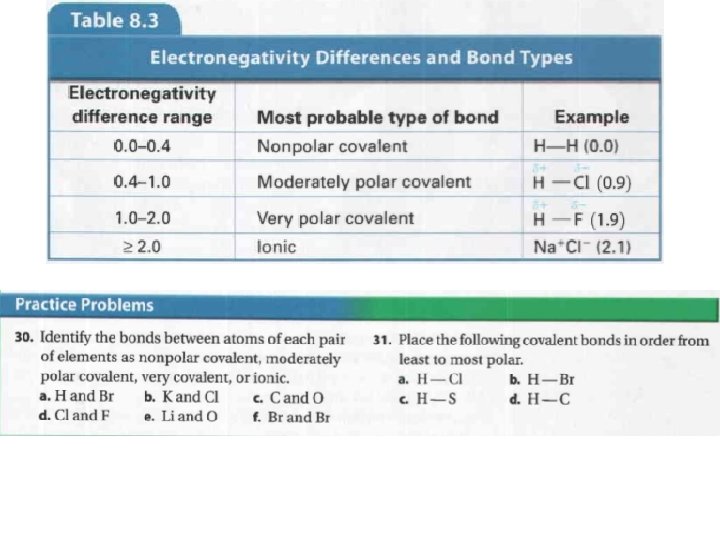
• Let’s look at a hydrogen chloride (HCl) molecule. – It is composed of hydrogen (electronegativity = 2. 1) and chlorine (electronegativity = 3. 0) • Make a prediction… Which partial charge will each atom have? • Chlorine has a higher electronegativity, so it will be partially negative. • If the electronegativity difference reaches 2. 0, it is very likely an electron will be transferred, rather than shared. – An ionic bond will form, rather than a covalent bond.

Polar Molecules: • The presence of a polar covalent bond in a molecule often makes the entire molecule polar. – In a polar molecule, one end of the molecule is slightly negative and the other end is slightly positive. • A molecule with two charged poles is known as a dipole. – Hydrogen chloride is a dipole. – When polar molecules are placed between oppositely charged plates, they tend to become oriented with respect to the positive and negative plates.

• The effect of polar bonds on the polarity of an entire molecule depends on the shape of the molecule and the orientation of the bonds. – Although there are polar bonds in CO 2, the molecule is nonpolar because it is linear and the polar bonds cancel out because they are in opposing direction. – Although water also only has 3 atoms, it is bent, so the polar bonds do not cancel. This leaves water as polar.
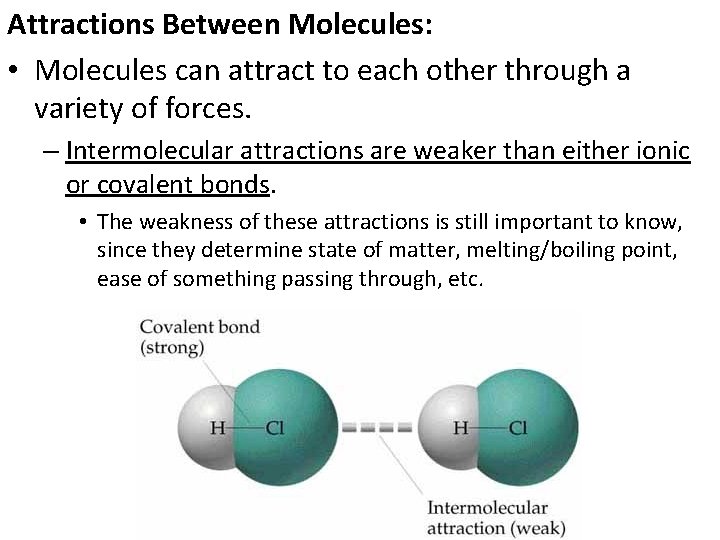
Attractions Between Molecules: • Molecules can attract to each other through a variety of forces. – Intermolecular attractions are weaker than either ionic or covalent bonds. • The weakness of these attractions is still important to know, since they determine state of matter, melting/boiling point, ease of something passing through, etc.
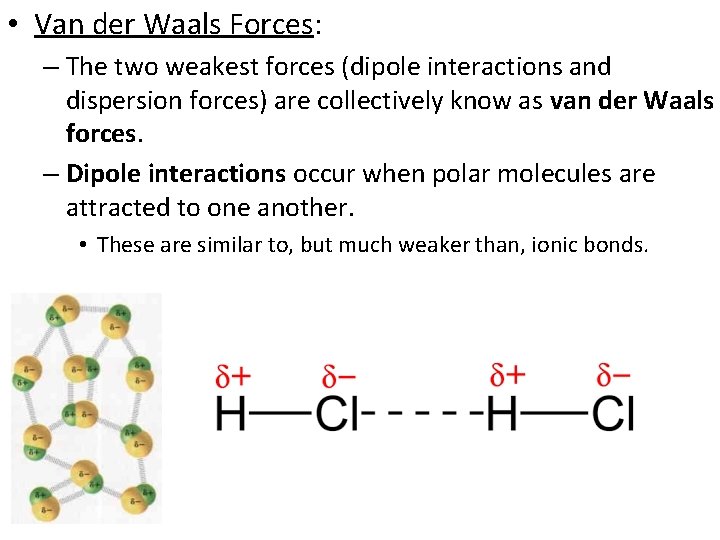
• Van der Waals Forces: – The two weakest forces (dipole interactions and dispersion forces) are collectively know as van der Waals forces. – Dipole interactions occur when polar molecules are attracted to one another. • These are similar to, but much weaker than, ionic bonds.

– Dispersion forces, the weakest of all molecular interactions, are caused by the motion of electrons, even between nonpolar molecules. • As a electrons move around molecules, they can momentarily closer to another molecule. – This causes the other molecule’s electrons to push to the opposite side (momentarily), creating an instantaneous charge, similar to a polar charge. • Examples: – Fluorine has relatively few electrons and are gases at room temperature because of their weak dispersion forces. – Bromine has a larger number of electrons, generating a larger dispersion force, which causes bromine to be a liquid at room temperature. – Iodine has even more electrons and even stronger dispersion forces, causing it to be a solid at room temperature.

• Hydrogen Bonds: – In water, we see a unique dipole interaction. • Water is exceptionally polar (Remember the biology stuff? ). – There is a strong attraction between the partial positive charge of hydrogen atom of one molecule and the partial negative charge of oxygen on another. • This occurs in many hydrogen containing molecules. • Hydrogen bonds are attractive forces in which a hydrogen covalently bonded to a very electronegative atom is also weakly bonded to an unshared electron pair of another electronegative atom. – This other atom may be a part of the same molecule (this is how proteins fold and DNA stays together) or from a different one.

Intermolecular Attractions and Molecular Properties: • The physical properties of a compound depend on the type of bonding it displays (ionic or covalent). – A great range of physical properties occurs among covalent compounds, due in part to the intermolecular interactions that take place.

– Compared to ionic compounds, covalent (molecular) compounds tend to have lower melting and boiling points. • Only weak attractions between molecules need to be broken to change the state of matter. – There a few molecular solids that are very stable and don’t melt until 1000 o. C is reached. • Network solids are solids in which all of the atoms are covalently bonded to one another. – Melting a network solid would require breaking covalent bonds throughout the solid. – Diamond is an example of a network solid.
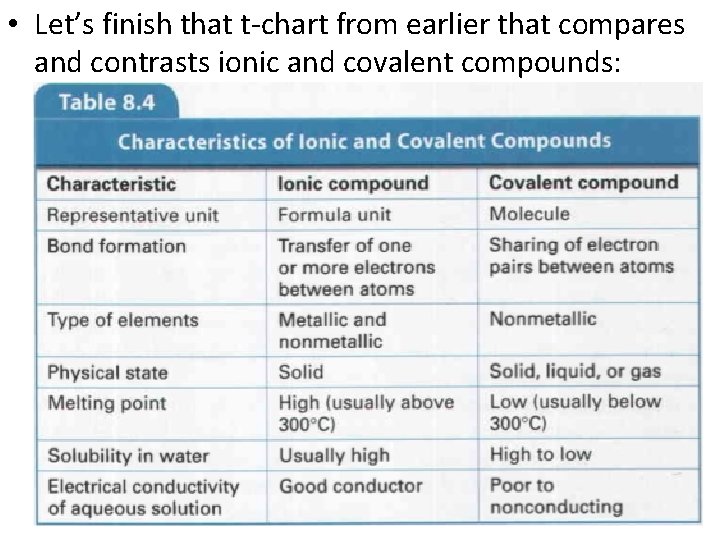
• Let’s finish that t-chart from earlier that compares and contrasts ionic and covalent compounds:

Learning Checkpoint: 1. How do electronegativity values determine the charge distribution in a polar covalent bond? 2. What happens when polar molecules are between metal plates with oppositely charged metal plates? 3. Compare the strengths of intermolecular forces to the strengths of ionic bonds and covalent bonds. 4. Explain why network solids have higher melting points. 5. Not every molecule with polar bonds is polar. Explain this statement. Use CCl 4 as an example. 6. Draw the structural formula for each molecule. Identify polar covalent bonds by assigning slightly positive and slightly negative symbols to the appropriate atoms. 1. HOOH 2. Br. Cl 3. HBr 4. H 2 O 7. How does a network solid differ from most other covalent compounds?
- Slides: 54