Covalent Bonding Bonding between a nonmetal and nonmetal
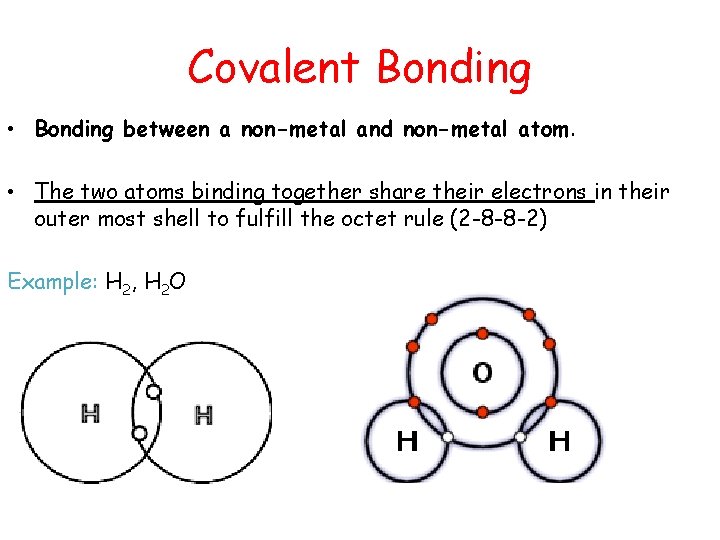
Covalent Bonding • Bonding between a non-metal and non-metal atom. • The two atoms binding together share their electrons in their outer most shell to fulfill the octet rule (2 -8 -8 -2) Example: H 2, H 2 O

Chemical Reactions • Occur when bonds are formed or broken causing substances to recombine into different substances. à All of the chemical reactions inside an organism = metabolism. Writing chemical equations: Reactants 2 molecules of H 2 1 molecule of O 2 Products 2 molecules of H 2 O Question: How many atoms of hydrogen do I have as reactants? As products? Conservation of mass: Atoms are neither created nor destroyed.

Mixtures and Solutions When elements combine chemically to form a compound, the elements no longer have their original properties. Mixture: Combination of substances in which individual components retain their own properties. Ex: mixing sand sugar together Solution: One or more substances (solute) are evenly distributed in another substance (solvent). Ex: mixing sugar and water together

Acids and Bases Chemical reactions in organisms depend on very specific environmental conditions: • Available energy • Temperature • Concentrations of substances • p. H: measure of how acidic or basic a solution is We classify the strength of acids and bases using the p. H scale.

Acids • Have a p. H below 7 • Molecular formulas for acids usually start with a hydrogen “H” or end with a carboxyl group “COOH”. Ex: HF, CH 3 COOH • An acid, when placed in water, will dissociate into H+ ions. Ex: HCl when placed in water will break down into H+ and Cl-
Bases • Have a p. H above 7 • The molecular formula of bases usually end with a hydroxyl group “OH” (exception: if the molecule starts with carbon “C” and ends with “OH” it is alcohol and not a base). Ex: Na. OH • Dissociate into OH- ions when put in water. Ex: Na. OH breaks up into Na+ and OH-

Homework p. 151 #1 -5
- Slides: 7