Covalent Bonding atoms share e covalent molecular compounds
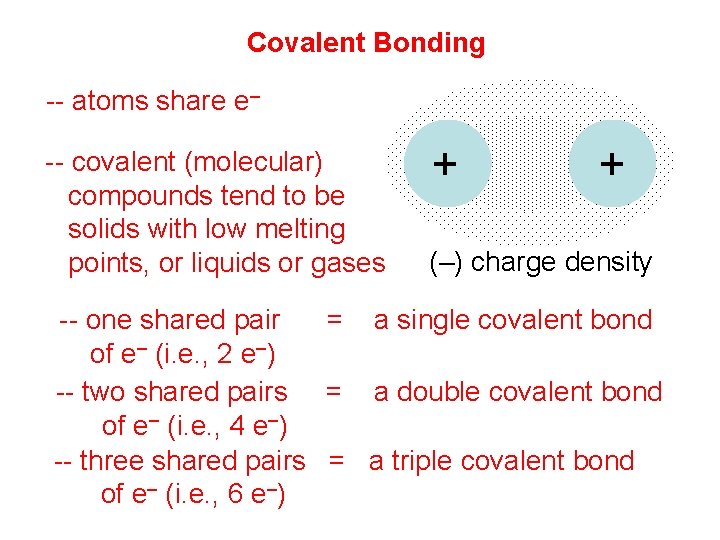
Covalent Bonding -- atoms share e– -- covalent (molecular) compounds tend to be solids with low melting points, or liquids or gases + + (–) charge density -- one shared pair = a single covalent bond of e– (i. e. , 2 e–) -- two shared pairs = a double covalent bond of e– (i. e. , 4 e–) -- three shared pairs = a triple covalent bond of e– (i. e. , 6 e–)

bond polarity: describes the sharing of e– between atoms nonpolar covalent bond: e– shared equally polar covalent bond: e– NOT shared equally electronegativity (EN): the ability of an atom in a molecule to attract e– to itself -- A bonded atom w/a large EN has a great ability to attract e–. max. EN = 4. 0 (F) -- A bonded atom w/a small EN min. EN = 0. 7 (Cs) – does not attract e very well. -- EN values have been tabulated. (see p. 308)

The DEN between bonded atoms approximates the type of bond between them. DEN < 0. 5 < DEN < 2. 0 DEN > 2. 0 nonpolar covalent ionic bond As DEN increases, bond polarity. . . increases. DEN for a C–H bond = 0. 4, so all C–H bonds (such as those in candle wax) are nonpolar.
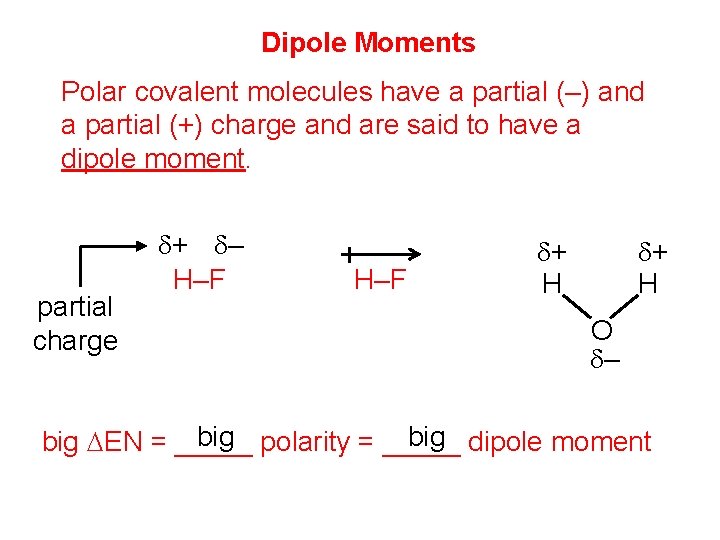
Dipole Moments Polar covalent molecules have a partial (–) and a partial (+) charge and are said to have a dipole moment. partial charge d+ d– H–F d+ H O d– big polarity = _____ big dipole moment big DEN = _____
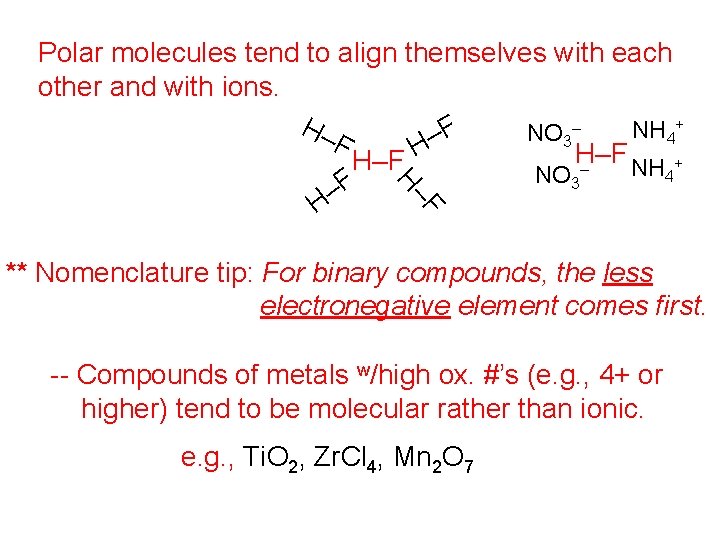
F H– Polar molecules tend to align themselves with each other and with ions. + H– – NH F NO 4 – 3 F H H–F – NH 4+ NO 3 F – H ** Nomenclature tip: For binary compounds, the less electronegative element comes first. -- Compounds of metals w/high ox. #’s (e. g. , 4+ or higher) tend to be molecular rather than ionic. e. g. , Ti. O 2, Zr. Cl 4, Mn 2 O 7

Lewis Structures (or “electron-dot structures”) 1. Sum the valence e– for all atoms. If the species is ion, add one e– for every (–); subtract one e– for e 2. Write the element symbols and connect the symbols with single bonds. 3. Complete octets for the atoms on the exterior of the structure, but NOT for H. 4. Count up the valence e– on your L. S. and compa that to the # from Step 1. -- If your LS doesn’t have enough e–, place as ma as needed on central atom. -- If LS has too many e– OR if central atom doesn an octet, use multiple bonds.
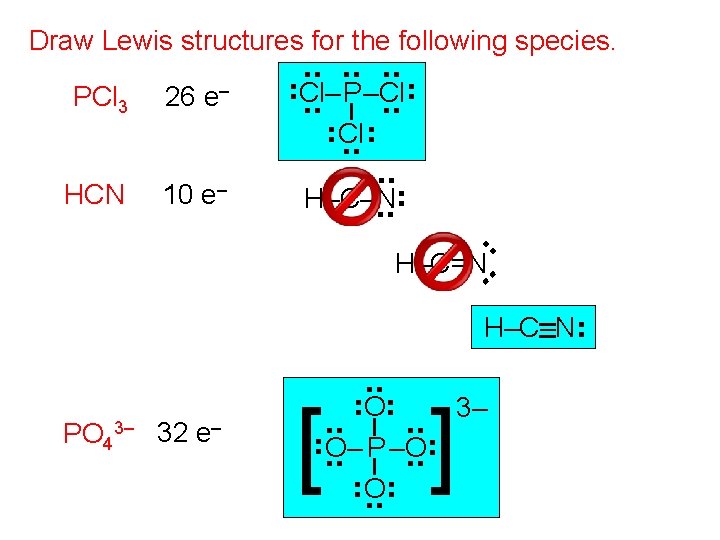
Draw Lewis structures for the following species. 10 e– . . HCN . . . 26 e– . . PCl 3 . . . Cl–. . P–Cl. . . . H–C–N. . H–C=N. . H–C–N . . ] . . . [ O. . O– P –O. . . . PO 43– 32 e– . . 3–
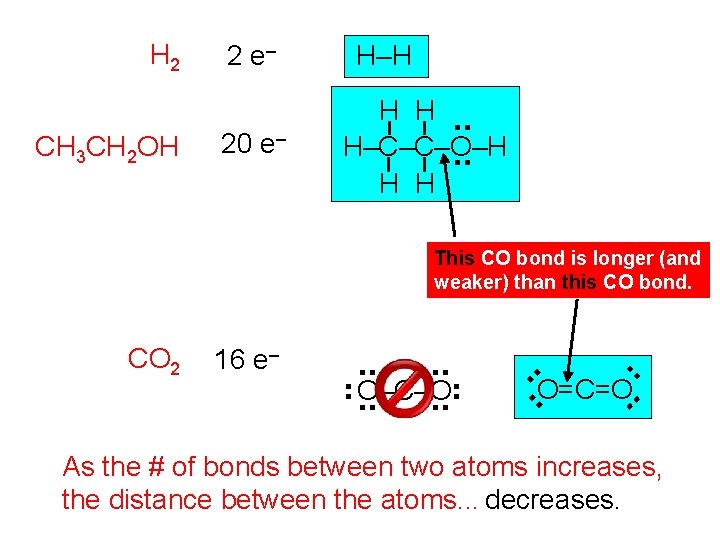
H 2 CH 3 CH 2 OH 2 e– 20 e– H–H H H. . H–C–C–O–H. . H H This CO bond is longer (and weaker) than this CO bond. . . O=C=O. . O–C–O. . . 16 e– . . CO 2 As the # of bonds between two atoms increases, the distance between the atoms. . . decreases.
- Slides: 8