Covalent bonding 1 2 3 4 5 Learning

Covalent bonding 1. 2. 3. 4. 5. Learning objectives: know that a covalent bond is the strong electrostatic attraction between two nuclei and the shared pair of electrons between them be able to draw dot-and-cross diagrams to show electrons in covalent substances, including: i molecules with single, double and triple bonds ii species exhibiting dative covalent (coordinate) bonding, including Al 2 Cl 6 and ammonium ion understand the relationship between bond lengths and bond strengths for covalent bonds


Bonding and structure What is a covalent bond ?

Bonding and structure A covalent bond is the strong electrostatic attraction between two nuclei and the shared pair of electrons between them

Bonding and structure draw a dot-and-cross diagram to show the electron arrangement in a molecule of chlorine cl 2
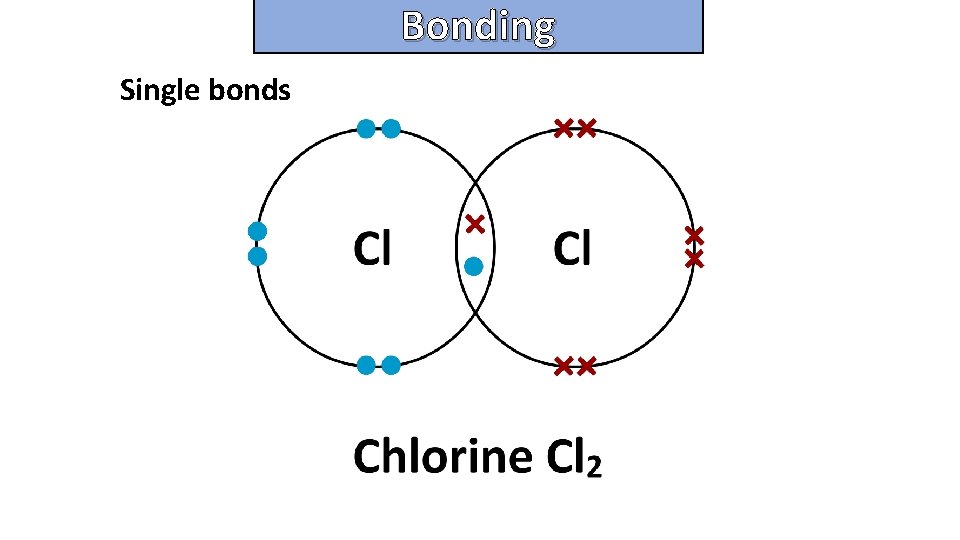
Bonding and structure Single bonds

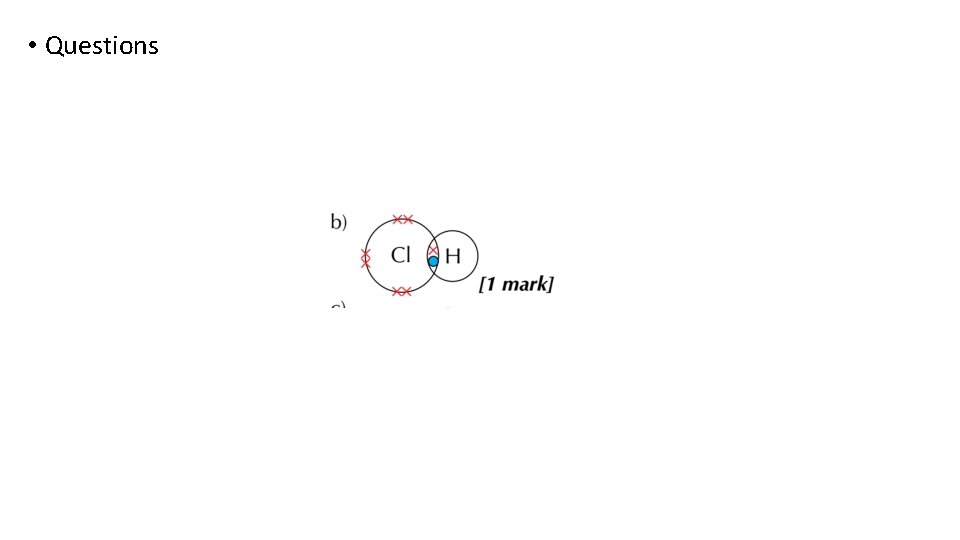
Bonding and structure draw a dot-and-cross diagram to show the electron arrangement in a molecule of hydrogen chloride HCl
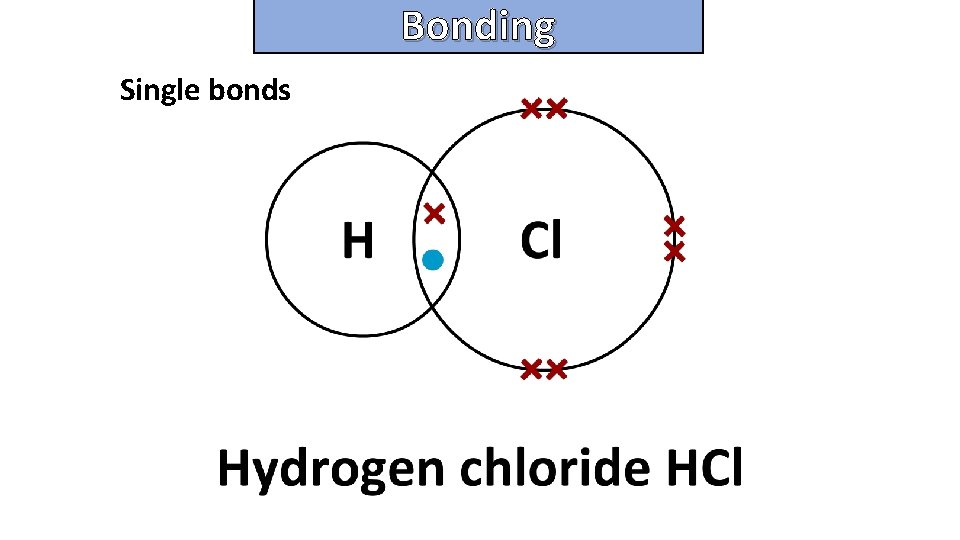
Bonding and structure Single bonds

Bonding and structure draw a dot-and-cross diagram to show the electron arrangement in a molecule of methane ch 4
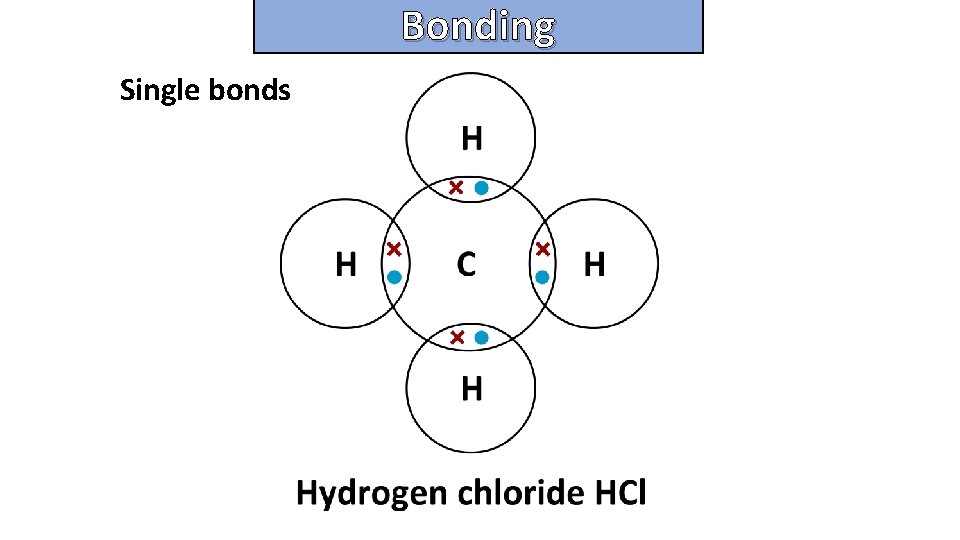
Bonding and structure Single bonds

Bonding and structure draw a dot-and-cross diagram to show the electron arrangement in a molecule of water H 2 O
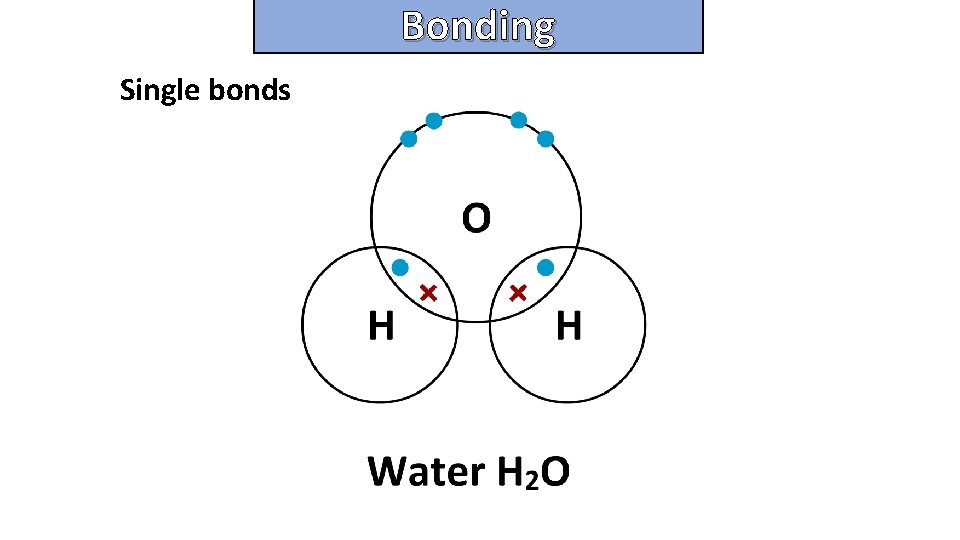
Bonding and structure Single bonds

Bonding and structure draw a dot-and-cross diagram to show the electron arrangement in a molecule of oxygen o 2
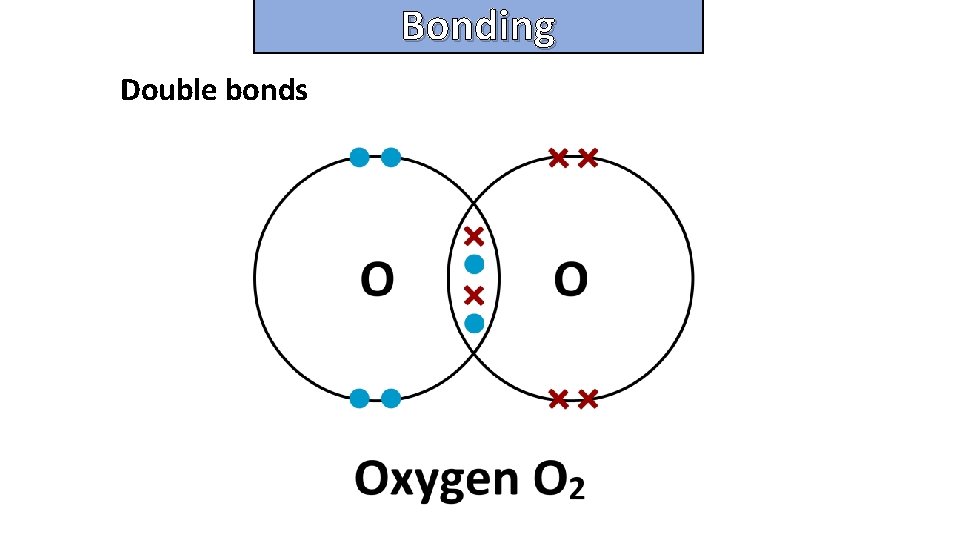
Bonding and structure Double bonds

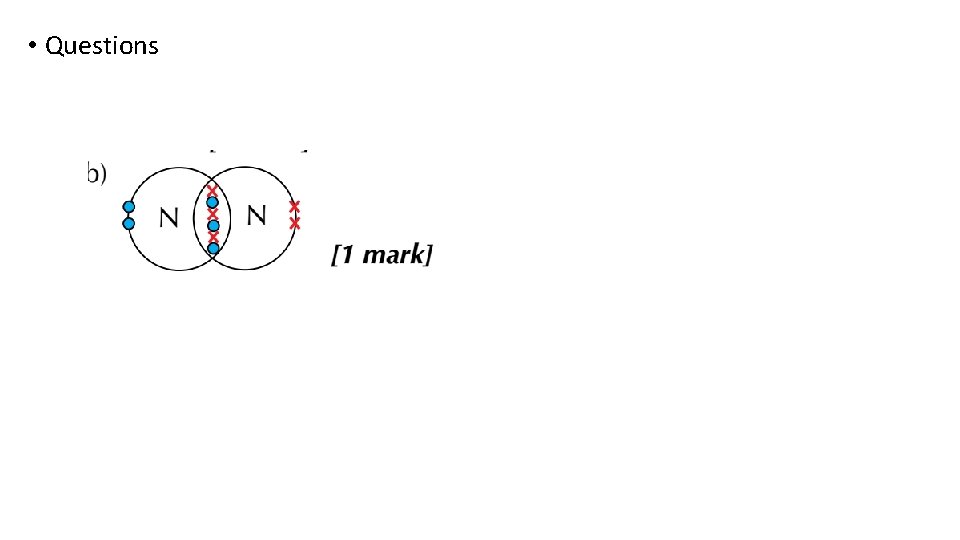
Bonding and structure draw a dot-and-cross diagram to show the electron arrangement in a molecule of nitrogen N 2
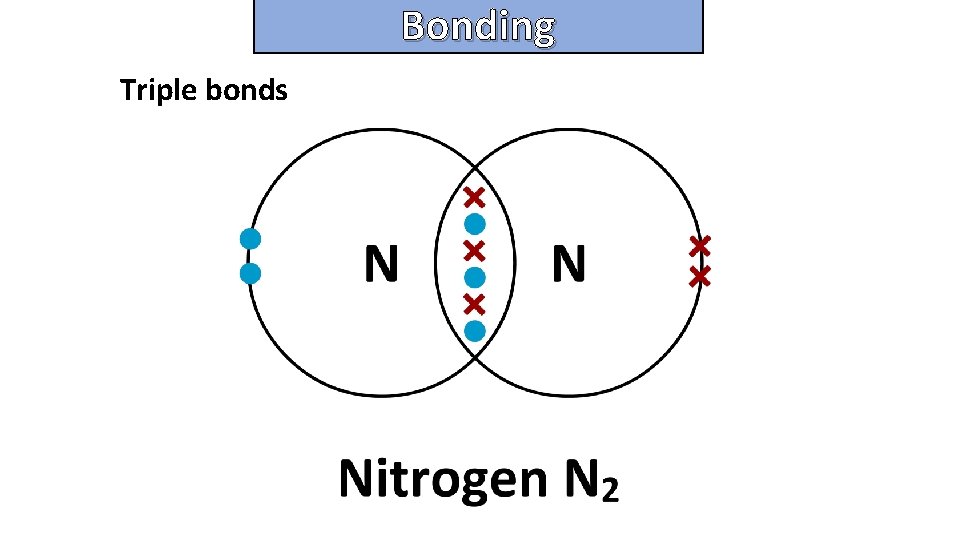
Bonding and structure Triple bonds

Bonding and structure draw a dot-and-cross diagram to show the electron arrangement in a carbon monoxide molecule CO
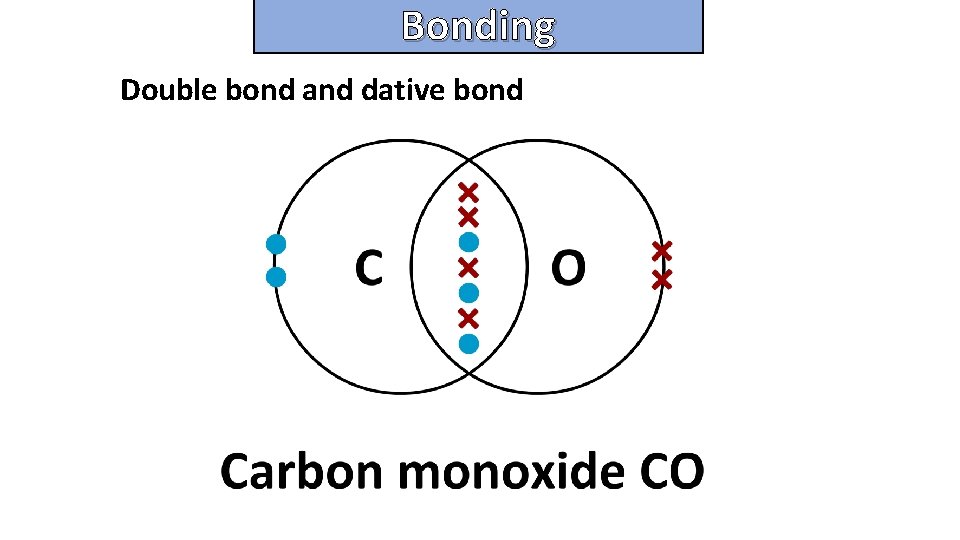
Bonding and structure Double bond and dative bond

Bonding and structure draw a dot-and-cross diagram to show the electron arrangement in an aluminum chloride molecule
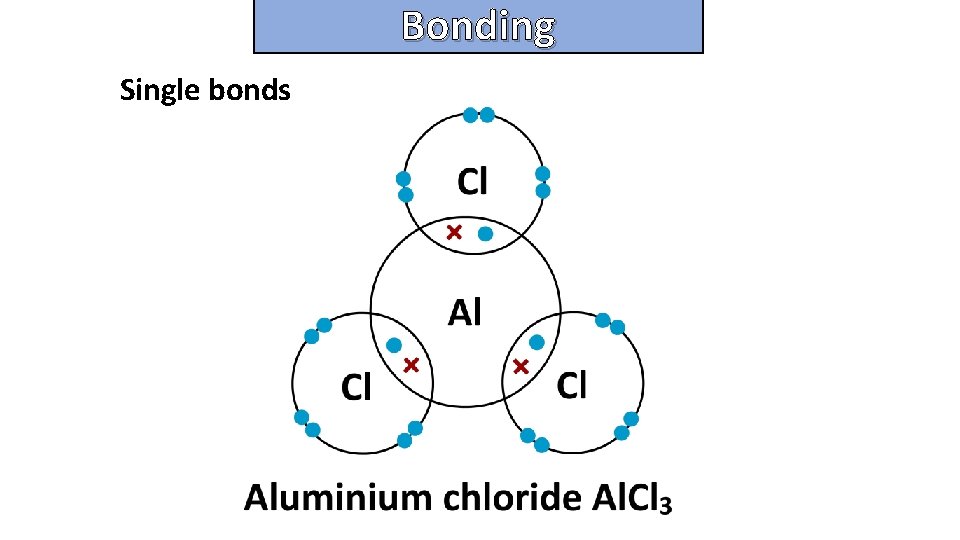
Bonding and structure Single bonds

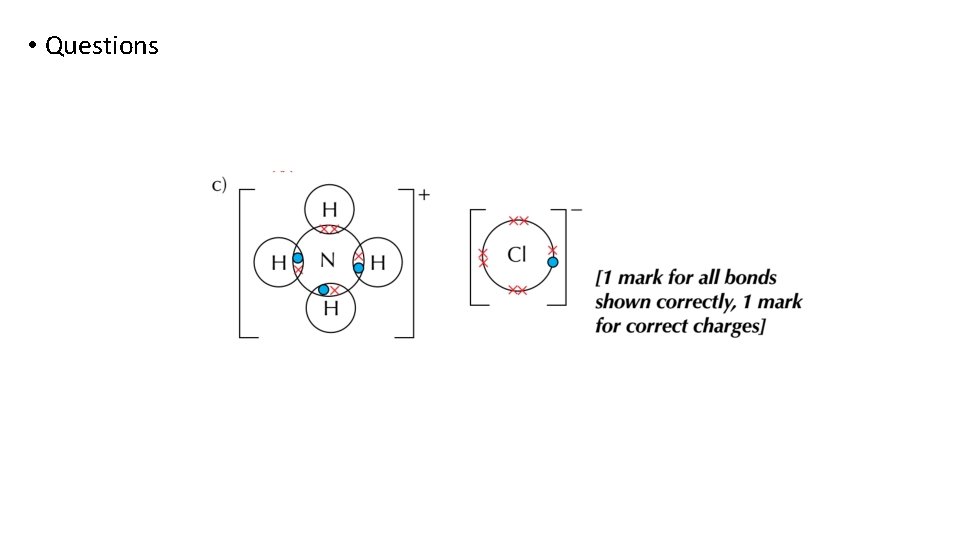
Bonding and structure draw a dot-and-cross diagram to show the electron arrangement in a molecule of Aluminum chloride Al 2 Cl 6
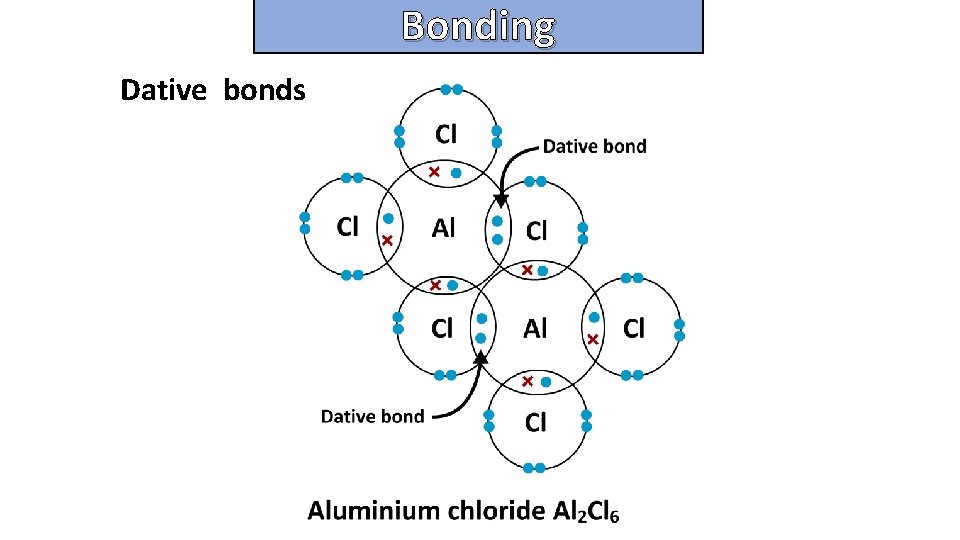
Bonding and structure Dative bonds

Bonding and structure what is dative (coordinate) bonding ?
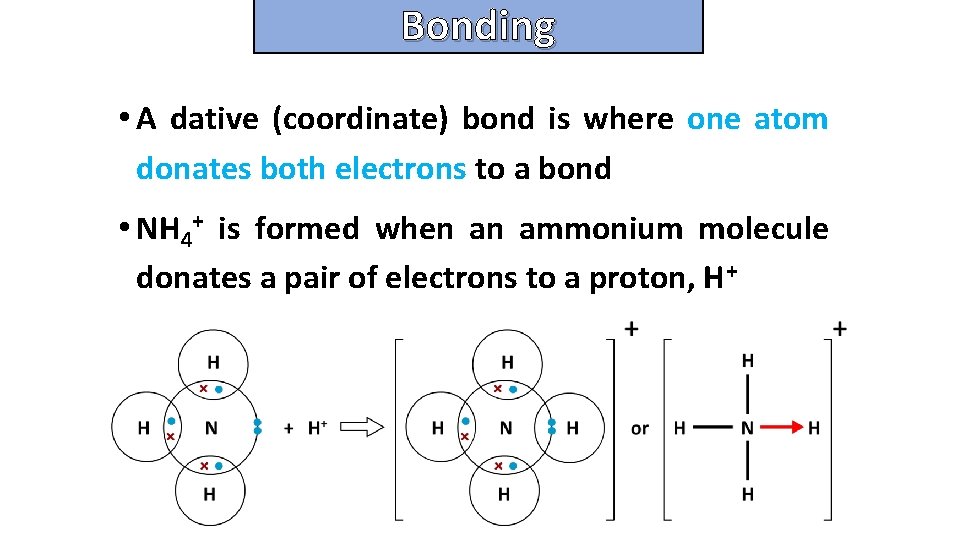
Bonding and structure • A dative (coordinate) bond is where one atom donates both electrons to a bond • NH 4+ is formed when an ammonium molecule donates a pair of electrons to a proton, H+

Bonding and structure Explain the relationship between bond lengths and bond strengths for covalent bond
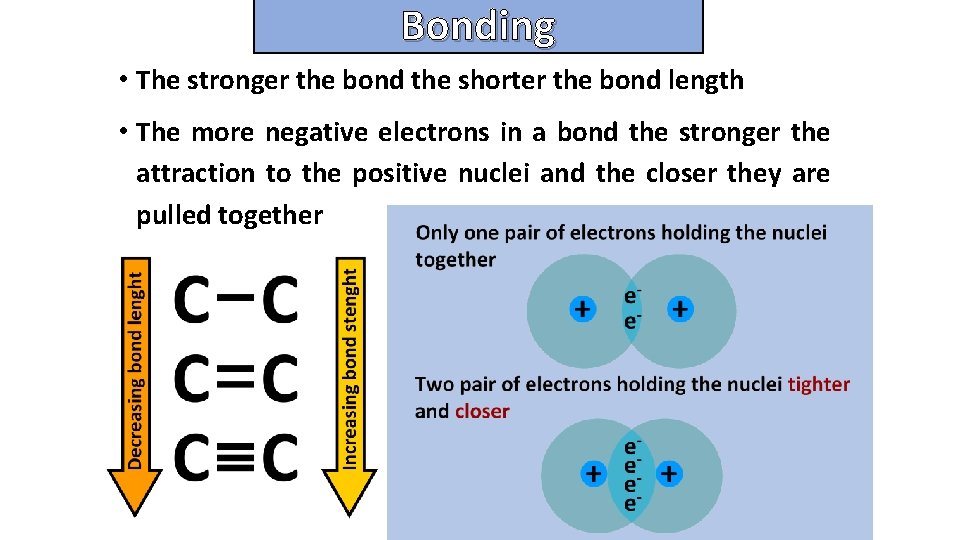
Bonding and structure • The stronger the bond the shorter the bond length • The more negative electrons in a bond the stronger the attraction to the positive nuclei and the closer they are pulled together

• Questions
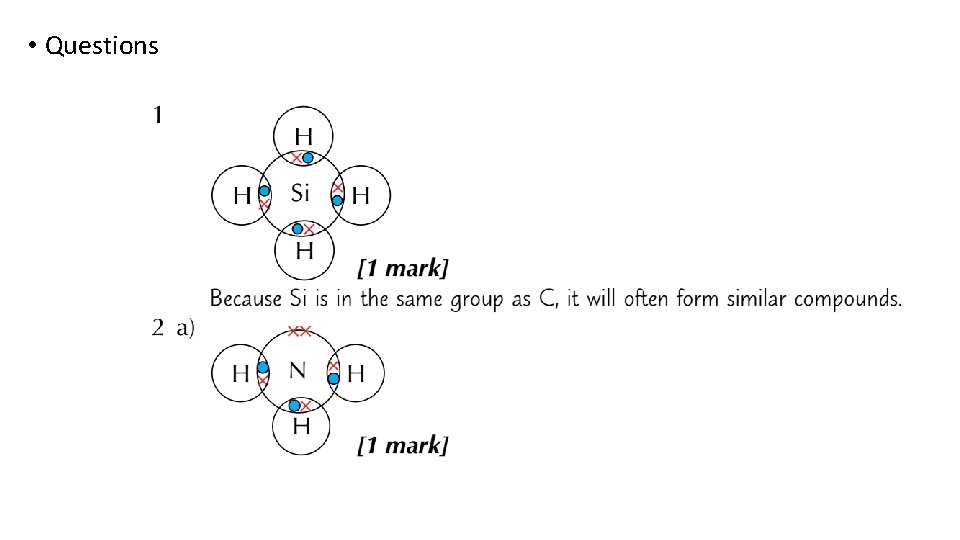
• Questions
• Questions
• Questions

• Questions
• Questions

• Questions

• Questions

• Questions

• Questions

• Questions

• Questions
- Slides: 38