Covalent bond Water molecule Covalent bonds Covalent bonds

Covalent bond • Water molecule

Covalent bonds • Covalent bonds are found in covalent compounds, ie compounds that are formed by sharing electrons. • There are different kinds of covalent bonds, such as Van Der Waal’s , polar covalent bonds or hydrogen bonding.

Hydrogen bonding • It is a bond that is formed between a H and another electronegative atom such as O, F, or Cl. • It is a very strong bond so it changes the properties of the compound, example is the water molecule.
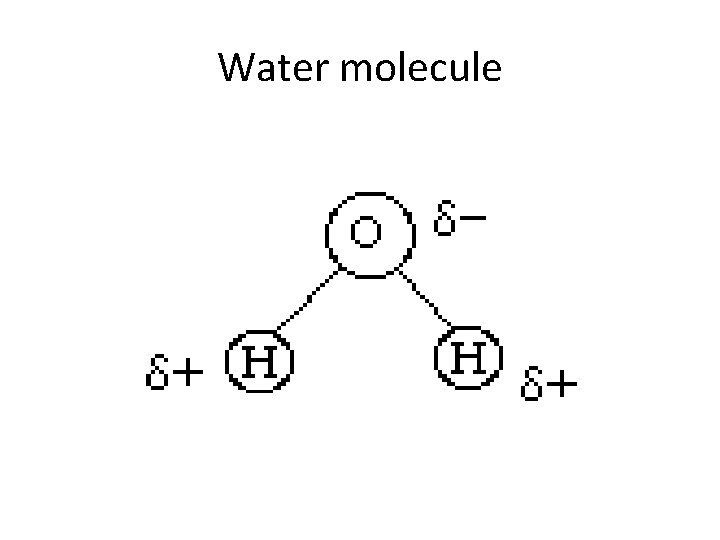
Water molecule

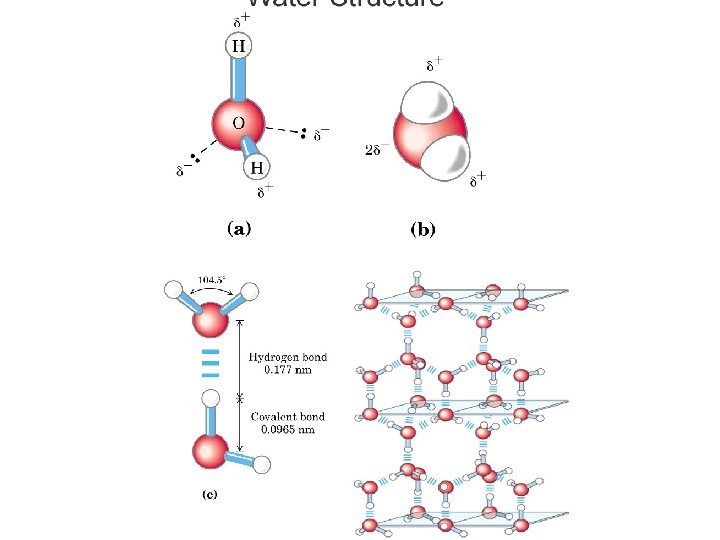
Water molecule • The symbols delta+ and delta- are used to indicate partial charges. Oxygen, because of its high electronegativity, attracts the electrons away from the hydrogen atoms, resulting in a partial negative charge on the oxygen and a partial positive charge on each of the hydrogens. The possibility of hydrogen bonds (H-bonds) is a consequence of partial charges.
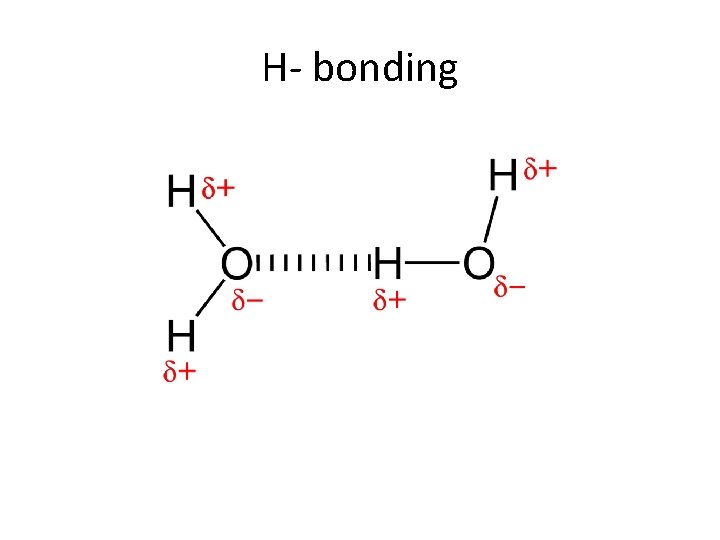
H- bonding

Hydrogen-Oxygen Bonding • Covalent bonds can also have partial charges when the atoms involved have different electronegativities. Water is perhaps the most obvious example of a molecule with partial charges.
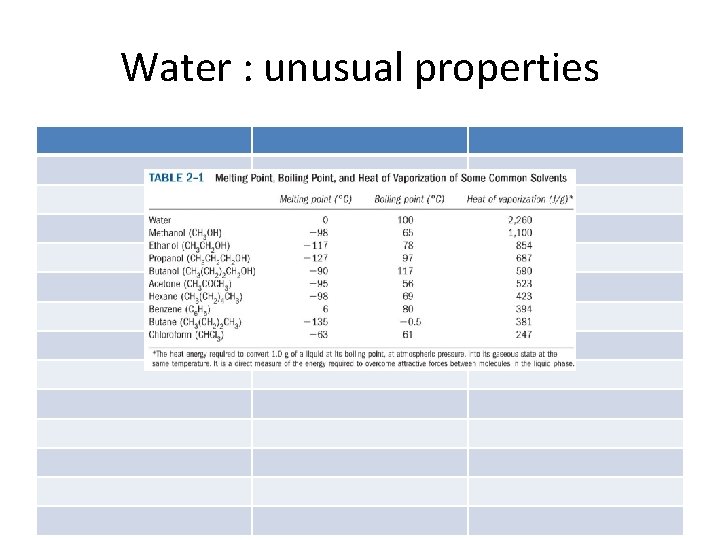
Water : unusual properties

Ice lighter than water

Water : special properties • Ice lighter than water: [reserves animal and plant life in frozen lakes, seas and rivers. • Water is used as the main solvent since it can dilute many different substances. • Water has a high boiling point so provides a stable environment that is essential for life.

Chemical reactions
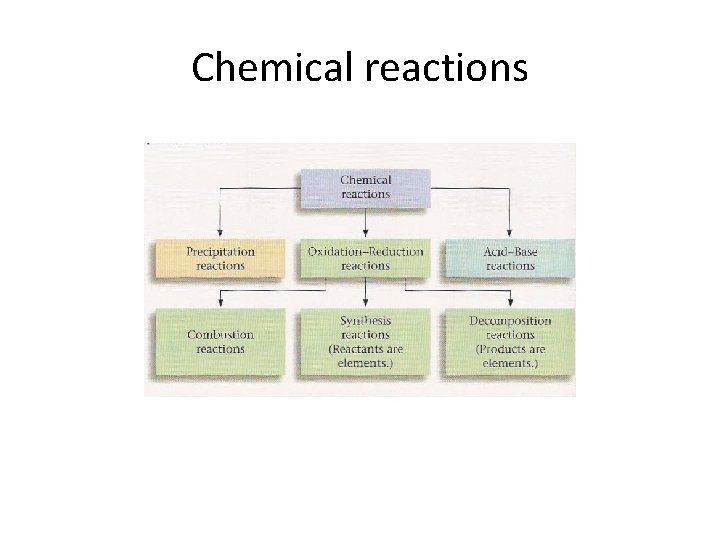
Chemical reactions

Types of chemical reactions • • Six basic types Combustion Synthesis Decompositions Displacement Double displacement Acid base Redox

Combustion • 1) Combustion: A combustion reaction is when oxygen combines with another compound to form water and carbon dioxide. These reactions are exothermic, meaning they produce heat. An example of this kind of reaction is the burning of napthalene: • C 10 H 8 + 12 O 2 ---> 10 CO 2 + 4 H 2 O

Synthesis • 2) Synthesis: A synthesis reaction is when two or more simple compounds combine to form a more complicated one. These reactions come in the general form of: • A + B ---> AB • One example of a synthesis reaction is the combination of iron and sulfur to form iron (II) sulfide: • 8 Fe + S 8 ---> 8 Fe. S

Decomposition • 3) Decomposition: A decomposition reaction is the opposite of a synthesis reaction - a complex molecule breaks down to make simpler ones. These reactions come in the general form: • AB ---> A + B • One example of a decomposition reaction is the electrolysis of water to make oxygen and hydrogen gas: • 2 H 2 O ---> 2 H 2 + O 2

Single displacement • 4) Single displacement: This is when one element trades places with another element in a compound. These reactions come in the general form of: • A + BC ---> AC + B • One example of a single displacement reaction is when magnesium replaces hydrogen in water to make magnesium hydroxide and hydrogen gas: • Mg + 2 H 2 O ---> Mg(OH)2 + H 2

Double displacement • 5) Double displacement: This is when the anions and cations of two different molecules switch places, forming two entirely different compounds. These reactions are in the general form: • AB + CD ---> AD + CB • One example of a double displacement reaction is the reaction of lead (II) nitrate with potassium iodide to form lead (II) iodide and potassium nitrate: • Pb(NO 3)2 + 2 KI ---> Pb. I 2 + 2 KNO 3

Acid-base • 6) Acid-base: This is a special kind of double displacement reaction that takes place when an acid and base react with each other. • The H+ ion in the acid reacts with the OH- ion in the base, causing the formation of water. Generally, the product of this reaction is salt and water: • HA + BOH ---> H 2 O + BA • One example of an acid-base reaction is the reaction of hydrochloric acid (HCl) with sodium hydroxide (Na. OH): • HCl + Na. OH ---> Na. Cl + H 2 O • Also called neutralization reaction

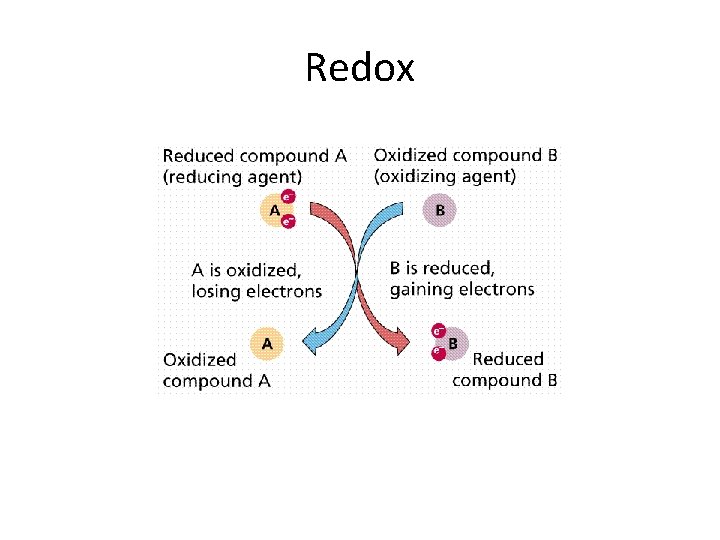
Redox Reactions (Oxidation - Reduction Reactions) • Redox reactions involve the loss or gain of electrons. • Oxidation = loss of electrons. All metals loose electrons to become cations. • Reduction = gain of electrons. All non-metals loose electrons to become anions. • Example Ca + O 2 --- 2 Ca. O • In these reactions, the valency or the oxidation number of the reactants change.

Redox reactions Ca + O 2 --- 2 Ca. O Can split the reaction into two half equations First half equation: Ca ---- Ca 2+ + 2 e- Loss of electrons is the oxidation part. • Second half equation: • O + 2 e- --- 2 O 2 - Gain of electrons is the reduction part. • •

Balancing redox reactions • Ca ---- Ca 2+ + 2 e • O + 2 e- --- 2 O 2 - this is not correct because oxygen is diatomic so we should write: • O 2 + 4 e- ---- 2 O 2 -. THIS IS CORRECT. • So now we need to have 4 e- not two, therefore we go back to the first half equation and adjust: • 2 Ca ---- 2 Ca 2+ + 4 eadd • O 2 + 4 e- --- 2 O 2 • 2 Ca + O 2 --- cancelled. 2 Ca. O note that electrons are

Redox = transfer of electrons • Redox reactions then, involve the transfer of electrons from one reactant to another. . . When there is oxidation, there is also reduction. • This is like the ionic bonding reactions that we learned earlier. • These are reactions between metals and nonmetals

Redox and oxidation number • • • For Example: Zn + 2 HCl -> Zn 2+ + H 2 +2 Cl. In this reaction we are interested in Zn + 2 H+ -> Zn 2+ + H 2 Zn is oxidised to Zn 2+ (loses 2 electrons) So for Zn 2+ the O. N. increases from 0 to +2 Therefore oxidation = loss of electrons OR increase in oxidation number.

Redox and O. N. • Zn + 2 HCl -> Zn 2+ + H 2 +2 Cl • H+ is reduced to H 2 (gains 2 electrons) • H+ has an oxidation number (valency) of +1, and is reduced to an oxidation number (valency) of 0. • So for H+ the Oxidation number is reduced. • Therefore reduction = gain of electrons or decrease in O. N.

Redox and O. N. • Zn + 2 HCl -> Zn 2+ + H 2 +2 Cl • In this reaction we are not interested in Cl because on the left the O. N, for Cl is -1 and on the right is also -1. • Therefore the chlorine is NOT changed in its ion state, so is not oxidised or reduced

Redox and O. N. • So oxidation increases the oxidation number (valency). • So, reduction decreases the oxidation number (valency).
Redox

Redox reactions • Reaction between Al and O 2 , Na and O 2 , and Al and Cl 2.

Magic words for Redox • OIL RIG • OIL = oxidation is loss of electrons • RIG = reduction is gain of electrons

Importance of Redox • Six key elements make up ~95% of all living organisms: carbon (C), hydrogen (H), oxygen (O), nitrogen (N), phosphorus (P) and sulfur (S). There also a number of other elements important to living organisms such as potassium (K), calcium (Ca), and magnesium (Mg). Chemical oxidation and reduction reactions involving these key elements They are crucial for life because they link the chemical, biotic, and geologic systems together. These reduction-oxidation (redox) reactions are usually mediated by organisms and especially bacteria, who gain energy from the exchanges of electrons.

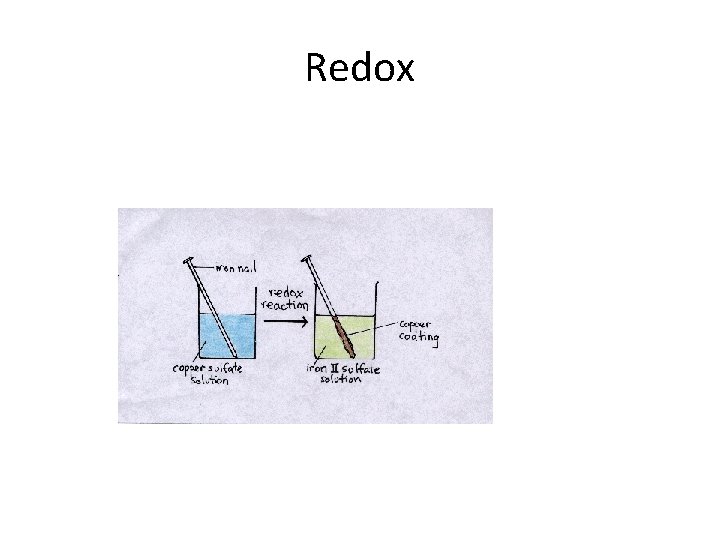
Redox reaction • A simple demonstration of a redox reaction involves placing a solid piece of copper wire in a silver nitrate solution. Within minutes the wire begins to look fuzzy or furry, as small silver crystals begin to form on the wire. Meanwhile, the originally clear silver nitrate solution begins to take on a pale bluish tint. Furthermore, if the crystals are shaken off of the wire we see that the wire partially disintegrated. • The overall equation for our demonstration describes the events: • Cu(s) + 2 Ag. NO 3 (aq) → Cu(NO 3)2 (aq) + 2 Ag(s)
Redox

Oxidation; old definition • The term oxidation originally referred to substances combining with oxygen, as happens when an iron bar rusts or a campfire log burns. We often refer to these two examples as corrosion and combustion.

Redox : remember • Oxidation the loss of electrons = OIL • OIL = Oxidation Is Loss • Reduction the gaining of electrons = RIG • RIG = Reduction Is Gain

Battery • A battery is a device that converts chemical energy directly to electrical energy. It consists of one or more voltaic cells. Each voltaic cell consists of two half cells connected in series by a conductive electrolyte. One half-cell is the negative electrode (the anode) and the other is the positive electrode (the cathode). In the redox reaction that powers the battery, reduction occurs in the cathode, while oxidation occurs in the anode. The electrodes do not touch each other but are electrically connected by the electrolyte, which can be either solid or liquid. In many cells, the materials are enclosed in a container, and a separator, which is porous to the electrolyte, which prevents the electrodes from coming into contact.

Zn battery

Zn battery • In a dry cell, the outer zinc container is the anode (-). The zinc is oxidised according to the following half-equation. • Zn(s) → Zn 2+(aq) + 2 e- A graphite rod surrounded by a powder containing manganese(IV) oxide is the cathode(+). The manganese dioxide is mixed with carbon powder to increase the conductivity of the cathode mixture. The cathode reaction is as follows: • 2 Mn. O 2(s) + 2 H+(aq) + 2 e- → Mn 2 O 3(s) + H 2 O(l) The H+ comes from the NH 4+(aq): • NH 4+(aq) → H+(aq) + NH 3(aq) and the NH 3 combines with the Zn 2+.

Oxidation number (ON) • The oxidation number is a number identical with the valency but with a sign, expressing the charge on the ion in question when formed from the neutral atom. • Thus, the oxidation number of chlorine in hydrochloric acid HCl is -1, while it is +1 in hypochlorous acid (HCl. O) • Similarly we can say that the oxidation number of chlorine in chloric acid (HCl. O 3) is +5, and in perchloric acid (HCl. O 4) +7.

Calculation of O. N. • • What are the O. N. In the following? ? Mg. O 2 Fe. Cl 3, Fe. Cl 2, KMn. O 4, Mn. O 4 - , Mn. O 2 NO, NO 2, HNO 3, NO 3 - , NH 3 Note that some elements have a constant O. N. Other elements , have a variable O. N. In molecules sum of O. N. Is zero

Radioactivity

Radioactivity : definition • It is the spontaneous breakdown of nuclei to form smaller and more stable atoms, release particles and energy. • Radioactivity is a natural phenomenon and it occurs because some nuclei are very large. • Usually atoms with atomic number bigger than 80 are radioactive. • They contain many protons and neutrons
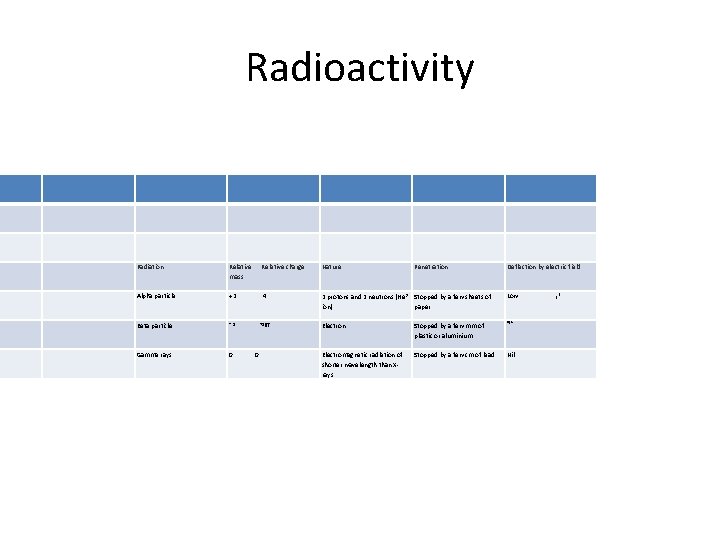
Radioactivity Radiation Relative mass Relative charge Nature Alpha particle +2 4 2 protons and 2 neutrons (He 2+ Stopped by a few sheets of ion) paper Low Beta particle ~1 TSfff Electron Stopped by a few mm of plastic or aluminium High Gamma rays 0 Electromagnetic radiation of shorter wavelength than Xrays Stopped by a few cm of lead Nil 0 Penetration Deflection by electric field f* ; ,
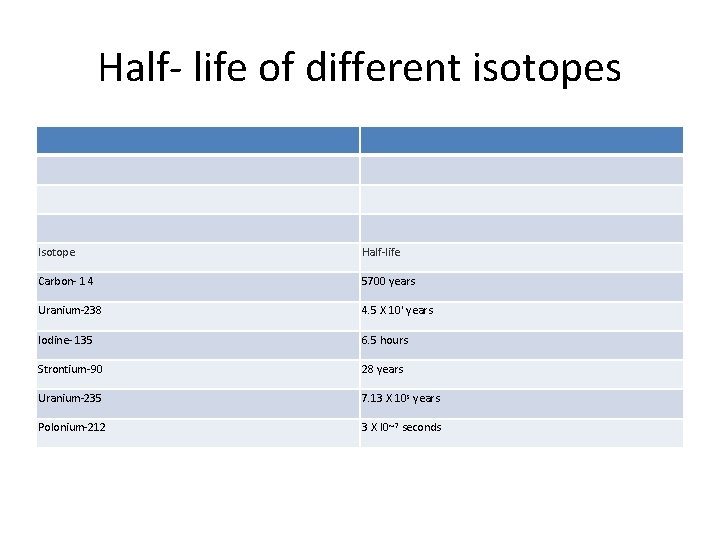
Half- life of different isotopes Isotope Half-life Carbon- 1 4 5700 years Uranium-238 4. 5 X 10' years Iodine- 135 6. 5 hours Strontium-90 28 years Uranium-235 7. 13 X 10 s years Polonium-212 3 X I 0~7 seconds
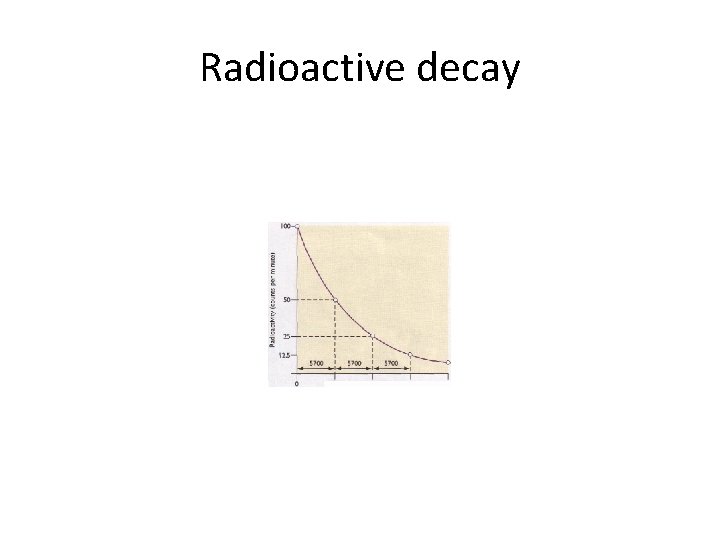
Radioactive decay

Half - life • Definition : It is the time taken for the radioactivity or the mass of a radioactive isotope to be reduced by half. • For example C-14 is radioactive and has a halflife of 5700 years. If have 100 Kgs of Carbon today in 11400 years how much carbon will I have left? ? • Answer = 25 Kgs.

Radioactive isotopes • Stable : have a very long half - life, e. g. uranium • Unstable : Have a very short half - life , e. g. pollonium

Nuclear equations

Alpha particle • This is a helium atom = • When an atom breaks down by alpha decay: – Its atomic number is reduce by 2 • • Its RAM is reduced by 4. Uranium Polonium radon

Alpha particle. . . • Alpha particles are made of 2 protons and 2 neutrons. • This means that they have a charge of +2, and a mass of 4 We can write them as , or, because they're the same as a helium nucleus, = • Alpha particles are relatively slow and heavy. • They have a low penetrating power - you can stop them with just a sheet of paper. • Because they have a large charge, alpha particles ionise other atoms strongly.

Alpha decay

1. Beta decay Beta particles have a charge of minus 1, and a mass of about 1/2000 th of a proton. This means that beta particles are the same as an electron. We can write them as or as . They are fast, and light. Beta particles have a medium penetrating power - they are stopped by a sheet of aluminium or plastics such as perspex. Beta particles ionise atoms that they pass, but not as strongly as alpha particles do

Beta decay

Exercise • Complete and balance the following nuclear equations:

Gamma rays • Gamma rays are waves, not particles. This means that they have no mass and no charge. So we sometimes write. • Gamma rays have a high penetrating power it takes a thick sheet of metal such as lead, or concrete to reduce them significantly. • Gamma rays do not directly ionise other atoms, although they may cause atoms to emit other particles which will then cause ionisation.
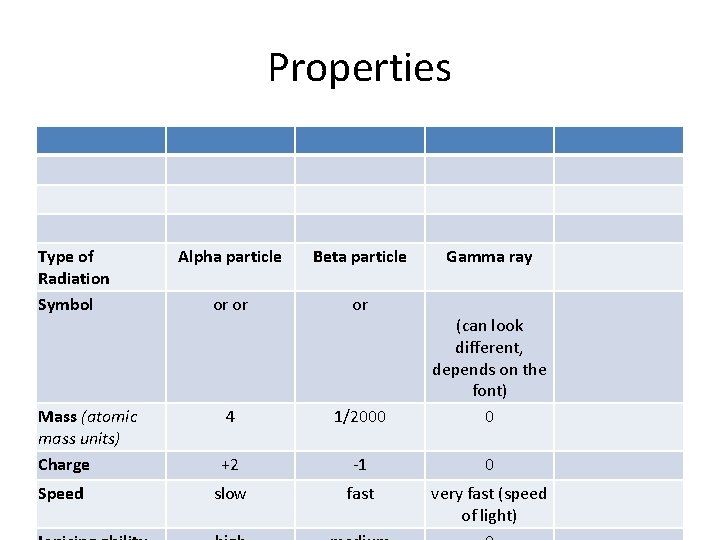
Properties Type of Radiation Symbol Mass (atomic mass units) Charge Speed Alpha particle Beta particle Gamma ray or or or 4 1/2000 (can look different, depends on the font) 0 +2 -1 0 slow fast very fast (speed of light)

Penetration
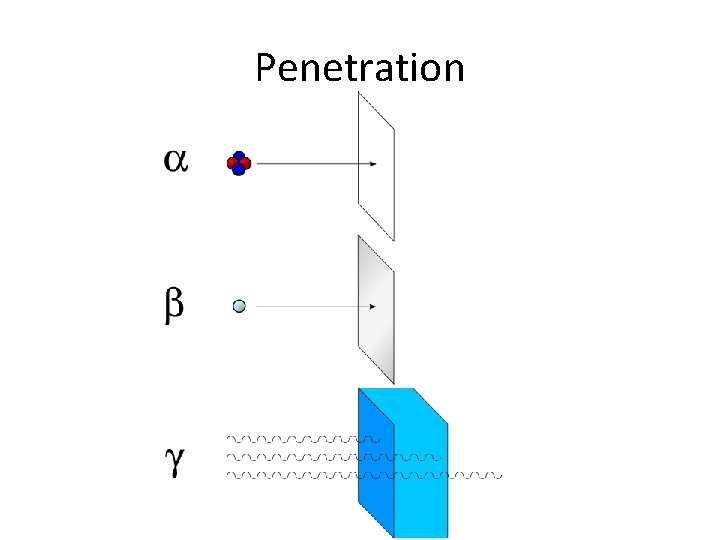
Penetration

Uses of radioactivity • • • Nuclear energy Carbon dating in archaelogy Medical applications Cancer treatment Industry Sterilization of food products- stop growth of bacteria

Carbon dating • Animals and plants have a known proportion of Carbon-14 (a radioisotope of Carbon) in their tissues. • When they die they stop taking Carbon in, then the amount of Carbon-14 goes down at a known rate (Carbon-14 has a half-life of 5700 years). • The age of the ancient organic materials can be found by measuring the amount of Carbon 14 that is left.

Medical applications • The most common tracer is called Technetium -99 and is very safe because it only emits gamma rays and doesn't cause much ionisation. • Radioisotopes can be used for medical purposes, such as checking for a blocked kidney. To do this a small amount of Iodine-123 is injected into the patient, after 5 minutes 2 Geiger counters are placed over the kidneys.

Cancer treatment • Because Gamma rays can kill living cells, they are used to kill cancer cells without having to resort to difficult surgery. This is called "Radiotherapy", and works because cancer cells can't repair themselves when damaged by gamma rays, as healthy cells can. • It's vital to get the dose correct - too much and you'll damage too many healthy cells, too little and you won't stop the cancer from spreading in time.

Detect leaking pipes • Also radioisotopes are used in industry, to detect leaking pipes. To do this, a small amount is injected into the pipe. It is then detected with a GM counter above ground

Sterilising • Even after it has been packaged, gamma rays can be used to kill bacteria, mould and insects in food. • This process prolongs the shelf-life of the food, but sometimes changes the taste. • Gamma rays are also used to sterilise hospital equipment, especially plastic syringes that would be damaged if heated

Control of thickness • In paper mills, the thickness of the paper can be controlled by measuring how much beta radiation passes through the paper to a Geiger counter. • The counter controls the pressure of the rollers to give the correct thickness. With paper, or plastic, or aluminium foil, b rays are used, because a will not go through the paper. • We choose a source with a long half-life so that it does not need to be replaced often.
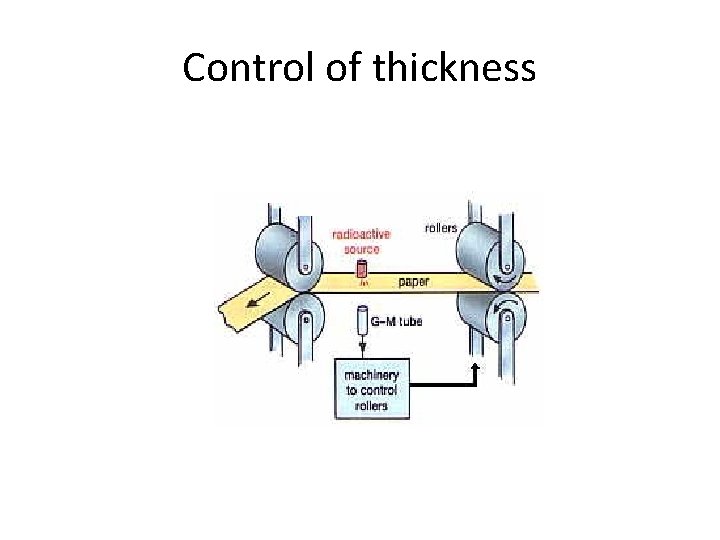
Control of thickness

Nuclear reactor • All nuclear reactors now in operation use nuclear fission (link to advantages of nuclear energy). Nuclear fission is the process where the nucleus (hence ‘nuclear’ energy) of a ‘heavy’, fissionable atom is split. Enormous amounts of energy are released in this process. • An atom’s nucleus can only split if it is ‘fissionable’. Only the nuclear isotopes Uranium 235 (U 235), Plutonium 239 and Uranium 238 are of this type. Only U 235 occurs naturally.

Nuclear reactor
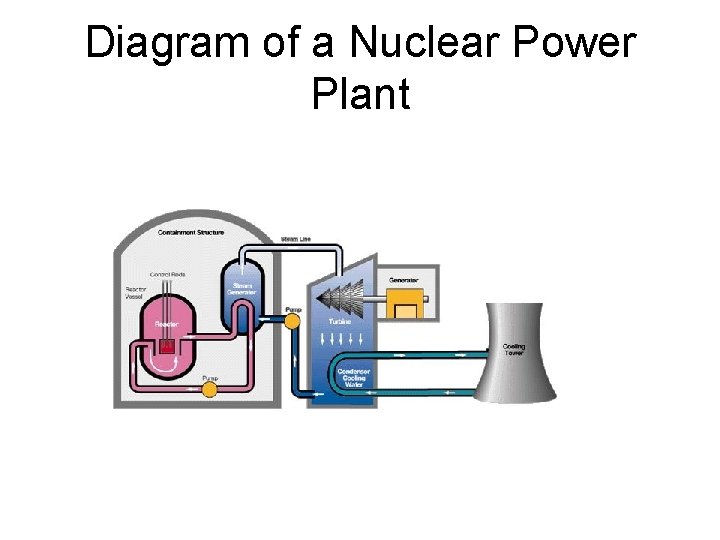
Diagram of a Nuclear Power Plant

Geiger Muller counter

Geiger-Müller • Geiger counters are used to detect radiation usually gamma and beta radiation, but certain models can also detect alpha radiation. • The sensor is a Geiger-Müller tube, an inert gas-filled tube (usually helium, with halogens added) that briefly conducts electricity when a particle or photon of radiation temporarily makes the gas conductive.

Units • The activity of a source is measured in Becquerels (Bq), One Becquerel is one decay per second. • The amount of radiation that your cells absorb is measured in grays (Gy), One gray is one Joule of energy absorbed by 1 kg of your body. This is the dose you receive. • Counts per minute (cpm).

Carbon dating : problem • In an old tomb anarchaelogist found a piece of wood which measured 20 cpm of radioactivity. • Estimate the age of the tomb provided that new wood gives 80 cpm. The half life for C-14 is 5700 years.

Uses of radioactivity

Atomic bomb • Nuclear fission - You can split the nucleus of an atom into two smaller fragments with a neutron. This method usually involves isotopes of uranium (uranium-235, uranium-233) or plutonium-239. • Nuclear fusion -You can bring two smaller atoms, usually hydrogen or hydrogen isotopes (deuterium, tritium), together to form a larger one (helium or helium isotopes); this is how the sun produces energy • In either process, fission or fusion, large amounts of heat energy and radiation are given off.

Atomic bomb
- Slides: 77