Covalent Bond Takes place between two nonmetals electrons

Covalent Bond * Takes place between two nonmetals * electrons are shared by both nuclei in order for each to attain a full valence shell * a molecule is formed when two or more elements bond covalently * makes a covalent/molecular compound
Molecular/Covalent Compounds

Properties • Most have low melting points and low boiling points. • Many are liquids and gases at room temperature. • Nonconductors of electricity. AKA Nonelectrolytes
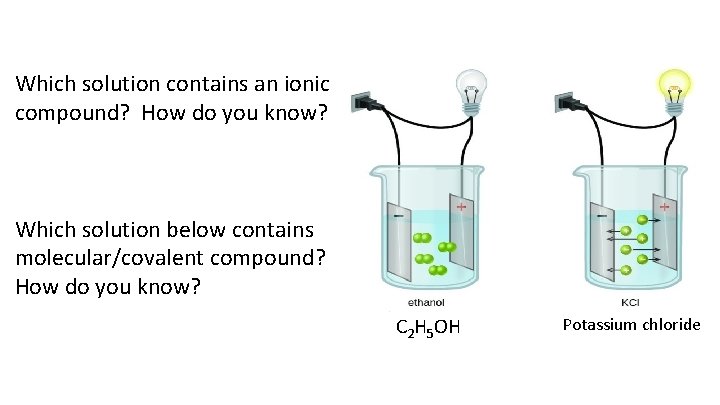
Which solution contains an ionic compound? How do you know? Which solution below contains molecular/covalent compound? How do you know? C 2 H 5 OH Potassium chloride

Naming & Writing Formulas

* to name and write formulas for covalent/molecular compounds, you HAVE to KNOW these prefixes

Naming 1. Name the first element Add the prefix that matches the subscript of that element in the formula. NEVER use mono- on the first element! 2. Name the second element & change the ending to -ide Add the prefix that matches the subscript of that element in the formula.

1. Name the first element Add the prefix that matches the subscript of that element in the formula. NEVER use mono- on the first element! 2. Name the second element & change the ending to -ide Add the prefix that matches the subscript of that element in the formula. N 2 O 5 dinitrogen pentaoxide

1. Name the first element Add the prefix that matches the subscript of that element in the formula. NEVER use mono- on the first element! 2. Name the second element & change the ending to -ide Add the prefix that matches the subscript of that element in the formula. CO carbon monoxide

1. Name the first element Add the prefix that matches the subscript of that element in the formula. NEVER use mono- on the first element! 2. Name the second element & change the ending to -ide Add the prefix that matches the subscript of that element in the formula. SH 2 sulfur dihydride

1. Name the first element Add the prefix that matches the subscript of that element in the formula. NEVER use mono- on the first element! 2. Name the second element & change the ending to -ide Add the prefix that matches the subscript of that element in the formula. P 4 O 6 tetraphosphorus hexaoxide

some rule breakers….
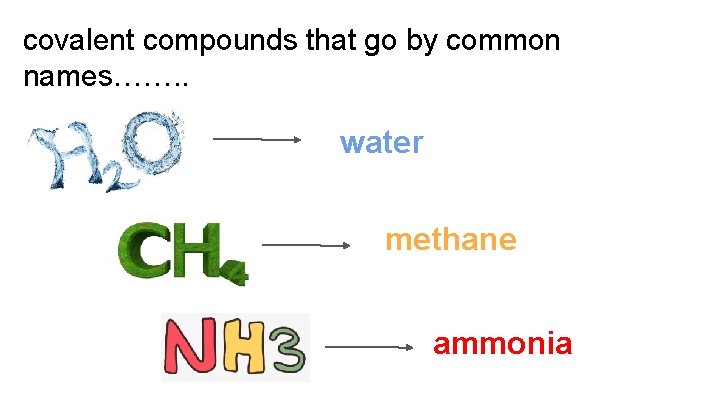
covalent compounds that go by common names……. . water methane ammonia

diatomic molecules…. . . when two of same elements (nonmetals) bond covalently to make one molecule these are named using only the name of the element oxygen

other diatomics……. there are 7 common diatomics you should recognize * find atomic #7, go right till group 7 A then go down to make the number 7 ** add hydrogen to make 7
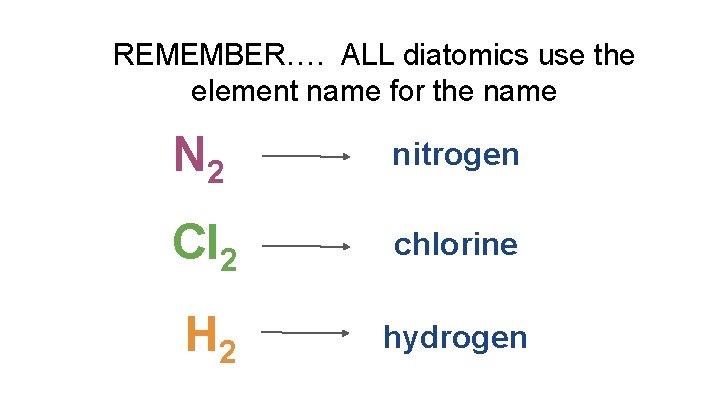
REMEMBER…. ALL diatomics use the element name for the name N 2 nitrogen Cl 2 chlorine H 2 hydrogen

Writing the Formula 1. Write the prefix in the first name as a subscript beside the element symbol. If there is not a prefix on the first name there is only one of the element. If there is only 1, NEVER write 1 as a subscript 2. Write the prefix in the second name as a subscript beside the element symbol. If mono- is the prefix, do NOT include a subscript. Lack of a subscript assumes 1.

Writing the Formula 1. Write the prefix in the first name as a subscript beside the element symbol. If there is not a prefix on the first name there is only one of the element. If there is only 1, NEVER write 1 as a subscript disulfur monoxide S 2 O 2. Write the prefix in the second name as a subscript beside the element symbol. If mono- is the prefix, do NOT include a subscript. Lack of a subscript assumes 1.

Writing the Formula 1. Write the prefix in the first name as a subscript beside the element symbol. If there is not a prefix on the first name there is only one of the element. If there is only 1, NEVER write 1 as a subscript carbon dioxide CO 2 2. Write the prefix in the second name as a subscript beside the element symbol. If mono- is the prefix, do NOT include a subscript. Lack of a subscript assumes 1.

Writing the Formula 1. Write the prefix in the first name as a subscript beside the element symbol. If there is not a prefix on the first name there is only one of the element. If there is only 1, NEVER write 1 as a subscript 2. Write the prefix in the second name as a subscript beside the element symbol. tetraphosphorus decasulfide P 4 S 10 If mono- is the prefix, do NOT include a subscript. Lack of a subscript assumes 1.

Writing the Formula 1. Write the prefix in the first name as a subscript beside the element symbol. If there is not a prefix on the first name there is only one of the element. If there is only 1, NEVER write 1 as a subscript iodine heptaflouride I F 7 2. Write the prefix in the second name as a subscript beside the element symbol. If mono- is the prefix, do NOT include a subscript. Lack of a subscript assumes 1.
- Slides: 21