Coupling between mass transfer and chemical reactions during

Coupling between mass transfer and chemical reactions during the absorption of CO 2 in a Na. HCO 3 -Na 2 CO 3 brine : experimental and theoretical study CRE XI, August 2007, Bilbao C. WYLOCK (F. N. R. S. Research fellow)(1), P. COLINET(1), T. CARTAGE(2), B. HAUT(1) Chemical Engineering Department, Applied Sciences Faculty, ULB (2) Solvay SA
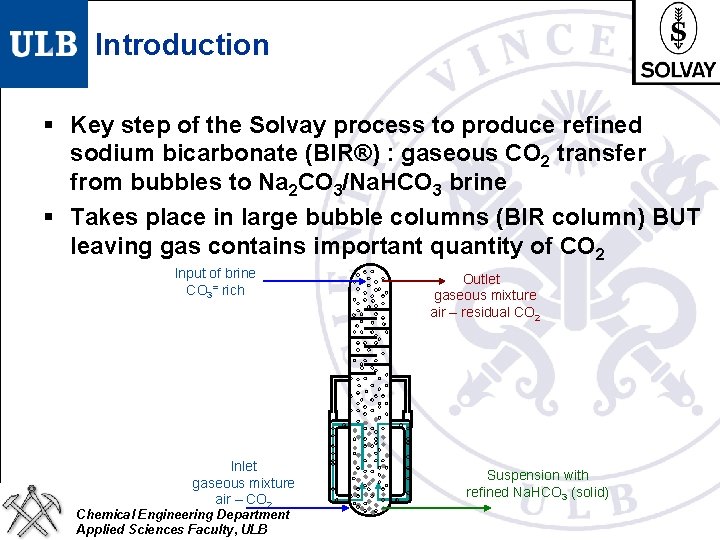
Introduction § Key step of the Solvay process to produce refined sodium bicarbonate (BIR®) : gaseous CO 2 transfer from bubbles to Na 2 CO 3/Na. HCO 3 brine § Takes place in large bubble columns (BIR column) BUT leaving gas contains important quantity of CO 2 Input of brine CO 3= rich Inlet gaseous mixture air – CO 2 Chemical Engineering Department Applied Sciences Faculty, ULB Outlet gaseous mixture air – residual CO 2 Suspension with refined Na. HCO 3 (solid)

Introduction § Key step of the Solvay process to produce refined sodium bicarbonate (BIR®) : gaseous CO 2 transfer from bubbles to Na 2 CO 3/Na. HCO 3 brine § Takes place in large bubble columns (BIR column) BUT leaving gas contains important quantity of CO 2 § CO 2 produced by lime calcination requires large amount of energy § Past optimization of the process : by empiric approach § A more fundamental approach is seeked Chemical Engineering Department Applied Sciences Faculty, ULB

Introduction § Goal : create a complete model, taking into account all the phenomena taking places in BIR column Optimization of the process will be looked for in order to increase mass transfer § Gaseous CO 2 transfer is coupled with chemical reactions in the liquid phase § This work : modelling of the coupling between gasliquid CO 2 transfer and the chemical reactions in the thin layer of liquid near the bubble interface § Multiscale approach is followed : - diffusion boundary layer (quiescent liquid) - gas bubble rising up in the liquid Chemical Engineering Department Applied Sciences Faculty, ULB

Presentation plan § Scale of the diffusion boundary layer 1. Model of the coupling between mass transfer and chemical reactions in a quiescent liquid 2. Mach-Zehnder interferometer § Scale of the bubble 1. Model of the bubble-liquid mass transfer 2. Isothermal stirred tank reactor § Conclusion and perspectives Chemical Engineering Department Applied Sciences Faculty, ULB

Modelling of the coupling between mass transfer and chemical reactions in a quiescent liquid § CO 2 gas-liquid absorption to a Na. HCO 3 -Na 2 CO 3 brine: view of the system Gas-liquid interface Gaseous phase Liquid phase Na. HCO 3/Na 2 CO 3 brine (p. H~10) Gas-liquid equilibrium Diffusion x=0 Chemical Engineering Department Applied Sciences Faculty, ULB Chemical reactions x Axis pointed toward the liquid phase in normal direction of the interface

Modelling of the coupling between mass transfer and chemical reactions in a quiescent liquid § Mass balance on an infinitesimal element of quiescent liquid transfer-reactions PDEs: with : Chemical Engineering Department Applied Sciences Faculty, ULB
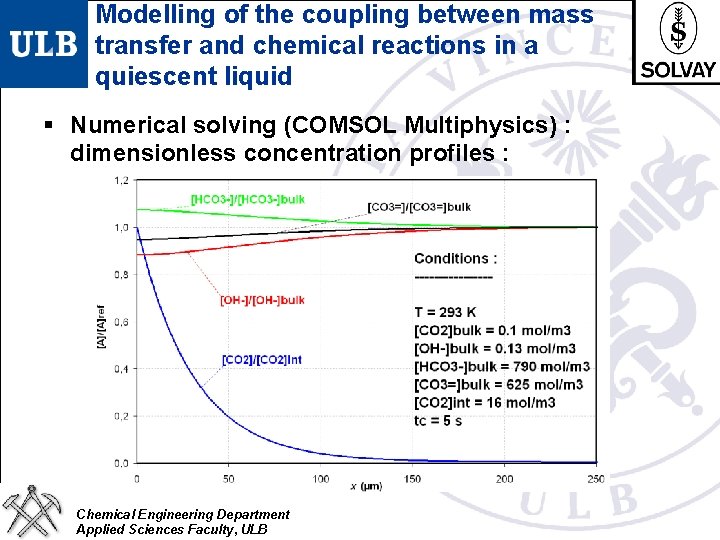
Modelling of the coupling between mass transfer and chemical reactions in a quiescent liquid § Numerical solving (COMSOL Multiphysics) : dimensionless concentration profiles : Chemical Engineering Department Applied Sciences Faculty, ULB

Presentation plan § Scale of the diffusion boundary layer 1. Model of the coupling between mass transfer and chemical reactions in a quiescent liquid 2. Mach-Zehnder interferometer § Scale of the bubble 1. Model of the bubble-liquid mass transfer 2. Isothermal stirred tank reactor § Conclusion and perspectives Chemical Engineering Department Applied Sciences Faculty, ULB

Mach-Zehnder interferometer § § § Aim : validation of the model of the coupling between mass transfer and chemical reactions Brine in a Hele-Shaw cell in contact with gaseous CO 2 set in a Mach-Zehnder interferometer Refractive index variation profiles near the interface, caused by the CO 2 transfer, can be observed interference fringes bend near the interface Mach-Zehnder block diagram Chemical Engineering Department Applied Sciences Faculty, ULB Experimental cell
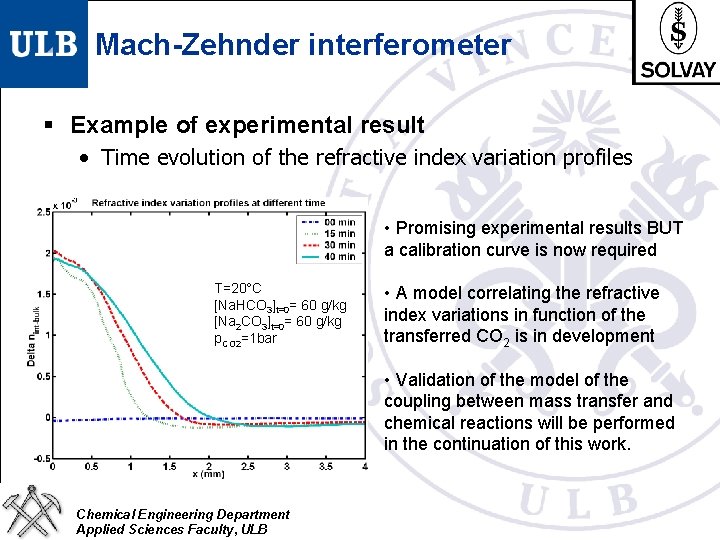
Mach-Zehnder interferometer § Example of experimental result • Time evolution of the refractive index variation profiles • Promising experimental results BUT a calibration curve is now required T=20°C [Na. HCO 3]t=0= 60 g/kg [Na 2 CO 3]t=0= 60 g/kg p. CO 2=1 bar • A model correlating the refractive index variations in function of the transferred CO 2 is in development • Validation of the model of the coupling between mass transfer and chemical reactions will be performed in the continuation of this work. Chemical Engineering Department Applied Sciences Faculty, ULB

Presentation plan § Scale of the diffusion boundary layer 1. Model of the coupling between mass transfer and chemical reactions in a quiescent liquid 2. Mach-Zehnder interferometer § Scale of the bubble 1. Model of the bubble-liquid mass transfer 2. Isothermal stirred tank reactor § Conclusion and perspectives Chemical Engineering Department Applied Sciences Faculty, ULB
![Bubble-liquid mass transfer model § [CO 2] profile interfacial CO 2 transfer rate : Bubble-liquid mass transfer model § [CO 2] profile interfacial CO 2 transfer rate :](http://slidetodoc.com/presentation_image_h2/5b0f27a3ce66f0a96d3a2505ff024ee9/image-13.jpg)
Bubble-liquid mass transfer model § [CO 2] profile interfacial CO 2 transfer rate : § Used to estimate interfacial CO 2 transfer rate from rising bubble in a bubble column § Required : representation of the liquid flow around the bubble Higbie model Chemical Engineering Department Applied Sciences Faculty, ULB
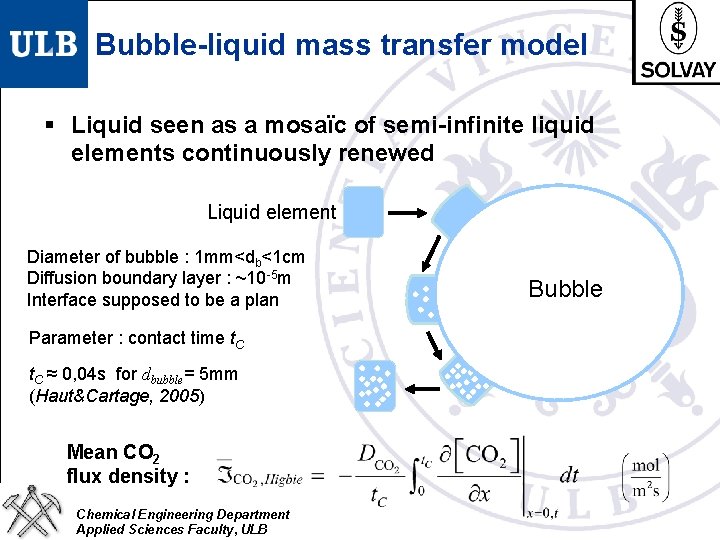
Bubble-liquid mass transfer model § Liquid seen as a mosaïc of semi-infinite liquid elements continuously renewed Liquid element Diameter of bubble : 1 mm<db<1 cm Diffusion boundary layer : ~10 -5 m Interface supposed to be a plan Parameter : contact time t. C ≈ 0, 04 s for dbubble= 5 mm (Haut&Cartage, 2005) Mean CO 2 flux density : Chemical Engineering Department Applied Sciences Faculty, ULB Bubble
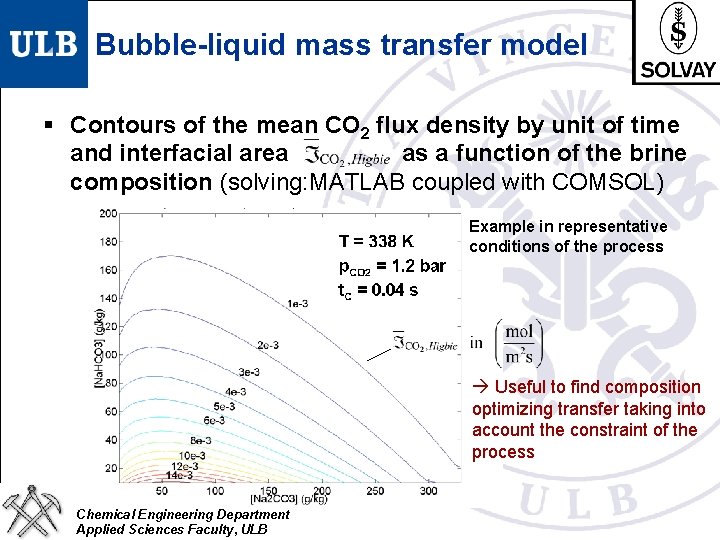
Bubble-liquid mass transfer model § Contours of the mean CO 2 flux density by unit of time and interfacial area as a function of the brine composition (solving: MATLAB coupled with COMSOL) Example in representative conditions of the process Useful to find composition optimizing transfer taking into account the constraint of the process Chemical Engineering Department Applied Sciences Faculty, ULB

Presentation plan § Scale of the diffusion boundary layer 1. Model of the coupling between mass transfer and chemical reactions in a quiescent liquid 2. Mach-Zehnder interferometer § Scale of the bubble 1. Model of the bubble-liquid mass transfer 2. Isothermal stirred tank reactor § Conclusion and perspectives Chemical Engineering Department Applied Sciences Faculty, ULB

Isothermal stirred tank reactor § Aim : validation of the bubble-liquid mass transfer model § Pure CO 2 bubbles pass through a brine § Continuous p. H and [CO 2]bulk measurement Bubble-liquid mass transfer model coupled with mass balances in the stirred tank reactor - Se (total interfacial area) adjustable parameter - contact time tc posed to 0. 02 s (tc for bubble of diameter 1. 5 mm) Þ Able to reproduce time evolution of the p. H and transferred CO 2 for different brine compositions Chemical Engineering Department Applied Sciences Faculty, ULB
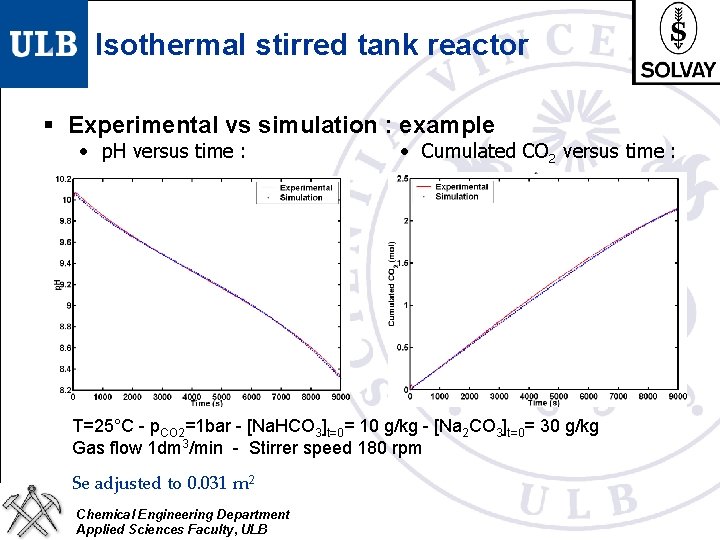
Isothermal stirred tank reactor § Experimental vs simulation : example • p. H versus time : • Cumulated CO 2 versus time : T=25°C - p. CO 2=1 bar - [Na. HCO 3]t=0= 10 g/kg - [Na 2 CO 3]t=0= 30 g/kg Gas flow 1 dm 3/min - Stirrer speed 180 rpm Se adjusted to 0. 031 m 2 Chemical Engineering Department Applied Sciences Faculty, ULB

Presentation plan § Scale of the diffusion boundary layer 1. Model of the coupling between mass transfer and chemical reactions in a quiescent liquid 2. Mach-Zehnder interferometer § Scale of the bubble 1. Model of the bubble-liquid mass transfer 2. Isothermal stirred tank reactor § Conclusion and perspectives Chemical Engineering Department Applied Sciences Faculty, ULB

Conclusion and perspectives § Multiscale approach : § Scale of the diffusion boundary layer • Mathematical model of the coupling between mass transfer and chemical reactions in a quiescent liquid § Scale of the bubble • Model of the coupling completed by the Higbie representation of the liquid flow around a gas bubble • Model of the bubble-liquid mass transfer to calculate the mean CO 2 flux density by unit of time and interfacial area in a BIR bubble column Chemical Engineering Department Applied Sciences Faculty, ULB

Conclusion and perspectives § An experimental device proposed for each scale § Mach-Zehnder interferometer • Allow to visualize refractive index variation profiles caused by the CO 2 transfer • Correlation between transferred CO 2 and refractive index variations in development • Validation of the model of the coupling : to be continued § Isothermal stirred tank reactor • Able to reproduce time evolution of the p. H and transferred CO 2 for different brine composition • Cross validation has to be performed • After this step : validation of the bubble-liquid mass transfer model Chemical Engineering Department Applied Sciences Faculty, ULB

Thank you for your attention Chemical Engineering Department Applied Sciences Faculty, ULB
- Slides: 22