Counting Orbitals I Atomic Orbitals n and m

Counting Orbitals I Atomic Orbitals: n, ℓ and mℓ Joseph F. Lomax U. S. Naval Academy Copy permission with this footer

Counting up Orbitals: In subshells Number of orbitals in a subshell Seven f-orbitals Five d-orbitals Three p-orbitals One s-orbital Joseph F. Lomax U. S. Naval Academy Copy permission with this footer
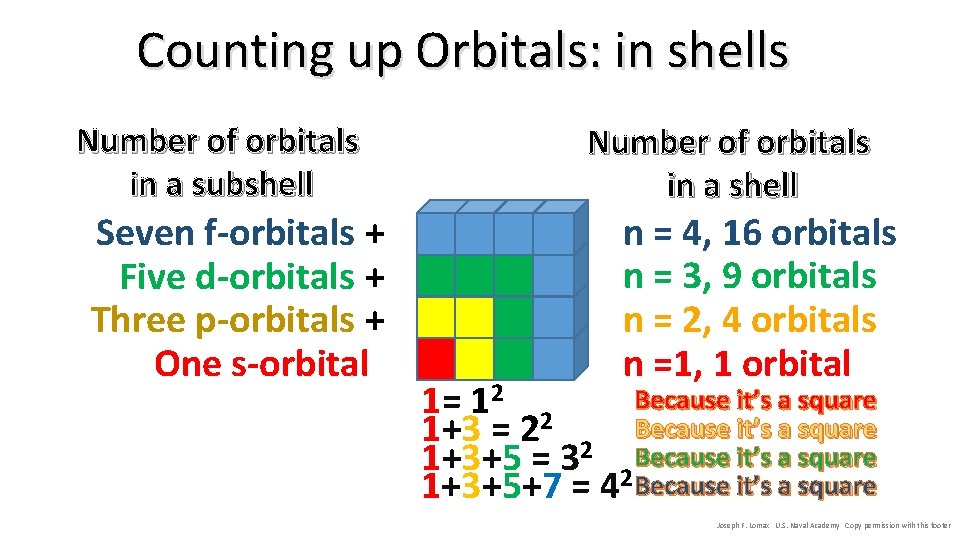
Counting up Orbitals: in shells Number of orbitals in a subshell Seven f-orbitals + Five d-orbitals + Three p-orbitals + One s-orbital Number of orbitals in a shell n = 4, 16 orbitals n = 3, 9 orbitals n = 2, 4 orbitals n =1, 1 orbital Because it’s a square 1= 12 2 1+3 = 2 2 Because it’s a square 1+3+5 = 3 2 Because it’s a square 1+3+5+7 = 4 Because it’s a square Joseph F. Lomax U. S. Naval Academy Copy permission with this footer
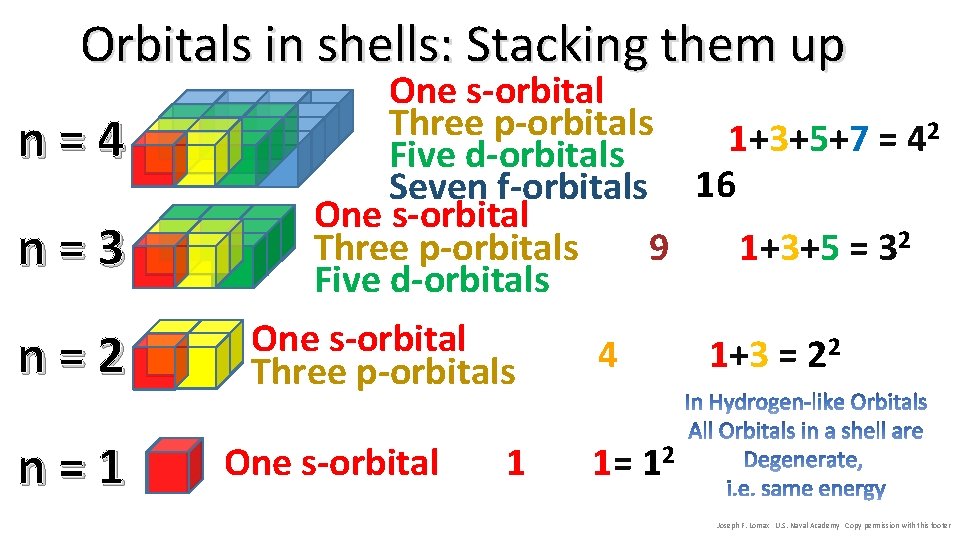
Orbitals in shells: Stacking them up n=4 n=3 n=2 n=1 One s-orbital Three p-orbitals 2 1+3+5+7 = 4 Five d-orbitals Seven f-orbitals 16 One s-orbital 9 1+3+5 = 32 Three p-orbitals Five d-orbitals One s-orbital 2 4 1+3 = 2 Three p-orbitals One s-orbital 1 1= 12 Joseph F. Lomax U. S. Naval Academy Copy permission with this footer
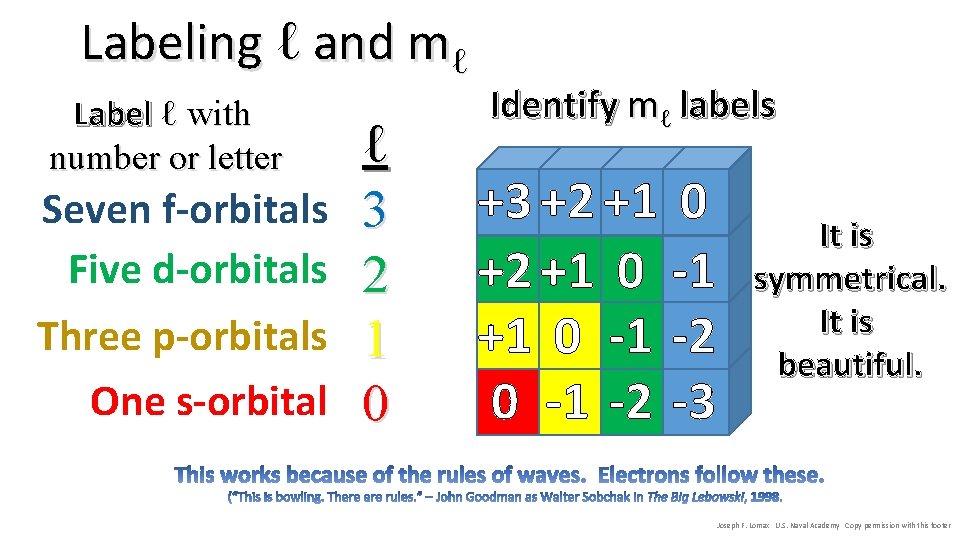
Labeling ℓ and mℓ Label ℓ with number or letter Seven f-orbitals Five d-orbitals Three p-orbitals One s-orbital ℓ 3 2 1 0 Identify mℓ labels +3 +2 +1 0 -1 -2 -3 It is symmetrical. It is beautiful. Joseph F. Lomax U. S. Naval Academy Copy permission with this footer
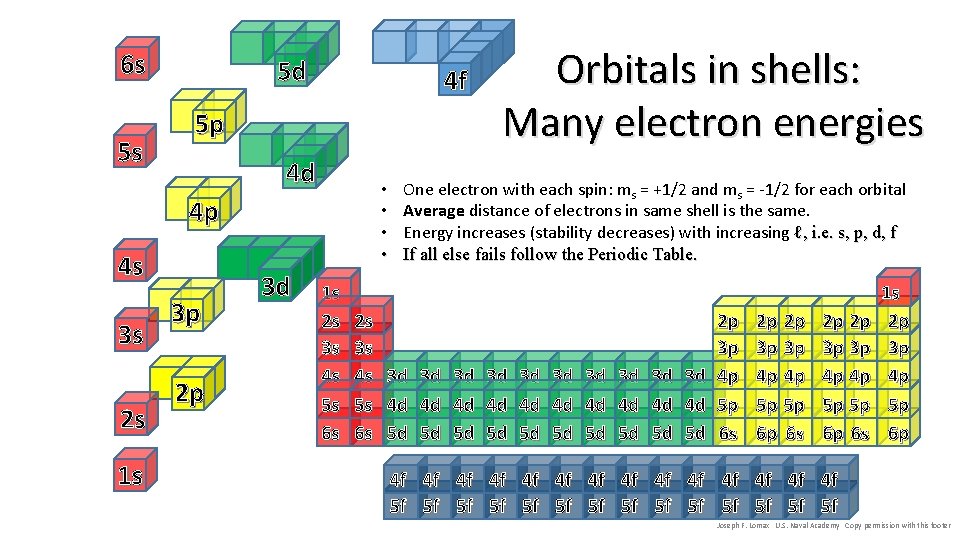
6 s 5 s 5 d 4 f 5 p 4 d • • 4 p 4 s 3 s 2 s 1 s 3 p 2 p 3 d 1 s 2 s 3 s 4 s 5 s 6 s Orbitals in shells: Many electron energies One electron with each spin: ms = +1/2 and ms = -1/2 for each orbital Average distance of electrons in same shell is the same. Energy increases (stability decreases) with increasing ℓ, i. e. s, p, d, f If all else fails follow the Periodic Table. 3 d 4 d 5 d 3 d 4 d 5 d 3 d 4 d 5 d 2 p 3 p 4 p 5 p 6 s 2 p 2 p 3 p 3 p 4 p 4 p 5 p 5 p 6 p 6 s 1 s 2 p 3 p 4 p 5 p 6 p 4 f 4 f 4 f 4 f 5 f 5 f 5 f 5 f Joseph F. Lomax U. S. Naval Academy Copy permission with this footer
- Slides: 6