Counting Atoms and Balancing Chemical Equations Identifying Compounds

Counting Atoms and Balancing Chemical Equations

Identifying Compounds Each new element is identified by a capital letter l Example: H 2 SO 4 l l The elements in Sulfuric Acid l Hydrogen l Sulfur l Oxygen

Subscripts l C 12 H 22 O 11 l There are 12 atoms of Carbon l There are 22 atoms of Hydrogen l There are 11 atoms of Oxygen If there is not a subscript listed, it is understood to be 1. l Example: Na. Cl l l There is one atom of Sodium l There is one atom of Chlorine

You Practice! Na. HCO 3 HCl Sodium – 1 Hydrogen – 1 Carbon – 1 Oxygen -3 Hydrogen – 1 Chlorine - 1

There are times you will see a compound with parenthesis. Pb(NO 3)2 The 2 after the parenthesis indicates there are two sets of the parenthesis. Pb(NO 3) So, in counting the atoms, you would have the following: Lead – 1 Oxygen - 6 Nitrogen -2

You Practice!! (NH 4)3 PO 4 Mg(OH)2 Nitrogen – 3 Hydrogen – 12 Phosphorus – 1 Oxygen – 4 Magnesium – 1 Oxygen – 2 Hydrogen – 2

Coefficient 2 H 2 SO 4 This means there are 2 compounds of Sulfuric Acid. Think: H 2 SO 4 Counting the atoms: Hydrogen – 4 Sulfur – 2 Oxygen - 8

You Practice!! 3 H 3 PO 4 2 H 2 O Hydrogen – 9 Phosphorus – 3 Oxygen - 12 Hydrogen – 4 Oxygen - 2

Got It? ?


Chemical Equations Chemical equations express what is happening in a chemical reaction using symbols. 2 K + Cl 2 Reactant yields 2 KCl Product

Law of Conservation of Mass In a chemical reaction, matter cannot be created or destroyed. It can only be changed. Therefore the Reactants must equal the Products for the equation to be balanced

What does that mean? ? The reactants MUST contain the same elements and the same number of each element as the product. BUT they CAN be in different compounds.

HUH? ? ? H 2 + O 2 Reactants H=2 O=2 H 2 O Products H =2 O=1 This cannot happen. An oxygen atom cannot be destroyed.

So what do we do? The chemical equation must be written according to the law. AGAIN --- HUH? ? ? H 2 + O 2 H 2 O is the production of water. Well, how it actually happens in nature is this…. 2 H 2 + O 2 Reactants: H=4 O=2 2 H 2 O Products: H=4 O=2

Do I Need to Know How to Balance? NOT YET. You need to be able to recognize if the chemical reaction follows the law of conservation of mass. Na + O 2 Mg + 2 HCl Na 2 O NOPE Mg. Cl 2 + H 2 YUP!!

You Practice!!! Hg. O Hg + O 2 Hg = O= NOPE YUP N 2 + 3 H 2 2 NH 3 YUP Zn + 2 HCl Zn. Cl 2 + H 2
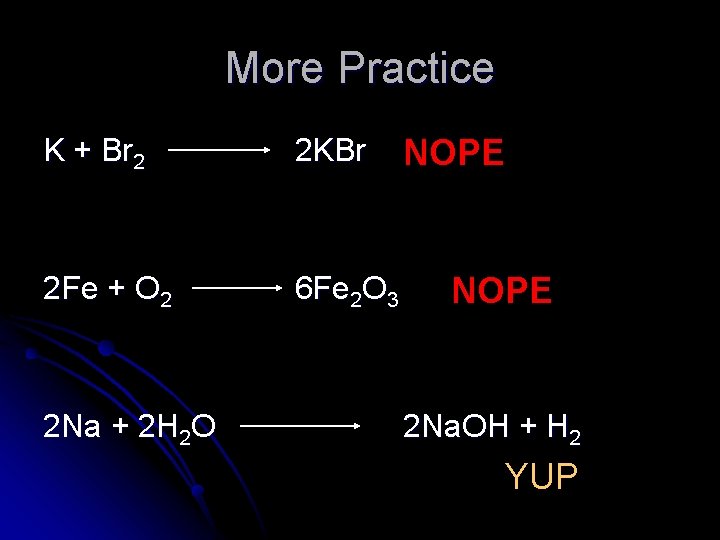
More Practice K + Br 2 2 KBr 2 Fe + O 2 6 Fe 2 O 3 2 Na + 2 H 2 O NOPE 2 Na. OH + H 2 YUP

Got It? ? ?

Balancing Chemical Equations H 2 + O 2 Reactants: H = 2 O=2 H 2 O Products: H = 2 O=1 What do we multiply by 1 to get 2?

Keep Going…. H 2 + O 2 Reactants: H = 2 O=2 2 H 2 O Products: H = 4 O=2 Now the Oxygen is balanced but Hydrogen is not, so what do we multiply by 2 to get 4?

Almost There!!! 2 H 2 + O 2 2 H 2 O Reactants: H = 4 O=2 Products: H = 4 O=2 Now it is balanced!!! Congratulations, you have now learned the hardest thing we will do all year!!
- Slides: 22