Counting Atoms 1 2 Atomic Symbol 15 9994

Counting Atoms 1 2
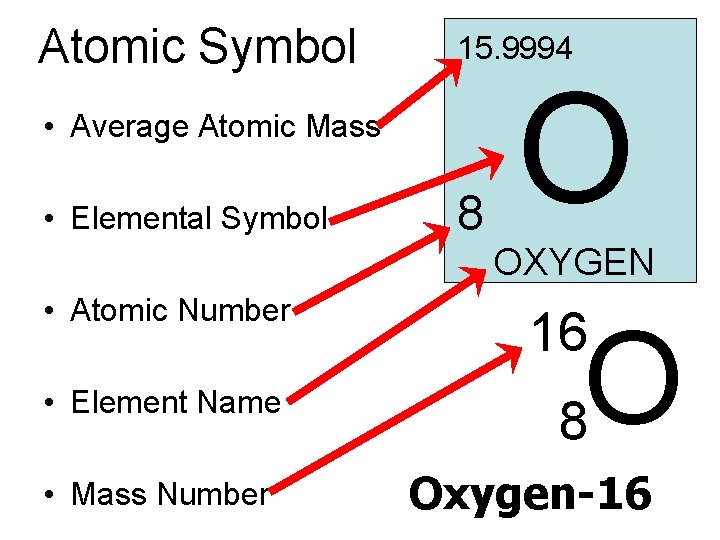
Atomic Symbol 15. 9994 • Average Atomic Mass • Elemental Symbol • Atomic Number • Element Name • Mass Number 8 O OXYGEN 16 O 8 Oxygen-16

So What is the… • Average Atomic Mass ? ? – The weighted avg. of the atomic masses of the naturally occurring isotopes of the element • Atomic Number ? ? – The nbr. of protons in the nucleus • Elemental Symbol ? ? – One or two letter symbol given to element, based on name • Mass Number ? ? – The total nbr. of protons and neutrons in the nucleus of an isotope

Are You Neutral? • All atoms, unless otherwise noted, are neutral. Meaning that they have the same number of _______ as ____. • Protons • Electrons • Remember that the nbr. of p+ is equal to the atomic nbr. • Every element has a specific atomic nbr. , no two element have the same A. N.
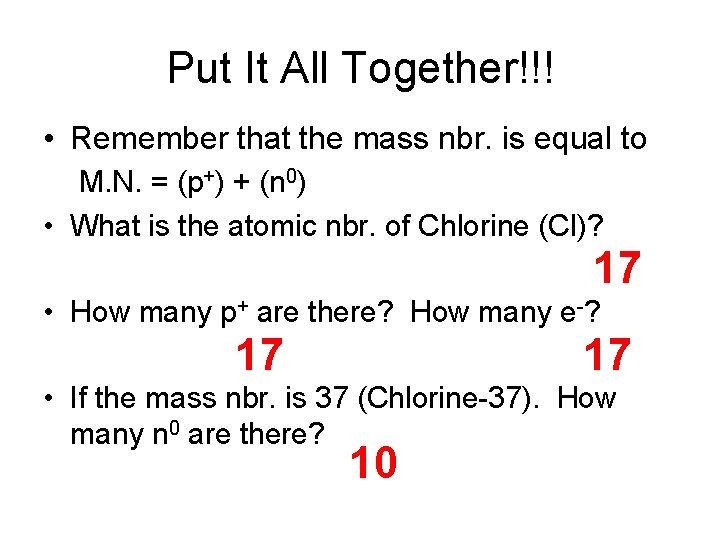
Put It All Together!!! • Remember that the mass nbr. is equal to M. N. = (p+) + (n 0) • What is the atomic nbr. of Chlorine (Cl)? 17 • How many p+ are there? How many e-? 17 17 • If the mass nbr. is 37 (Chlorine-37). How many n 0 are there? 10

Try this… • Determine the number of protons, neutrons and electrons in an atom of each of the following: Carbon-14 Uraniuim-234

Isotopes • Atoms of the same element the have different masses • A modification to Dalton’s original Atomic Theory • Nuclide – a general term for any isotope

Try this… • Hydrogen has three isotopes/nuclides, Hydrogen 1 (Protium), Hydrogen-2 (Deuterium) and Hydrogen-3 (Tritium) • Diagram a model showing the number and location of protons (p+), Neutrons (n 0) and electrons (e-) Isotope Hydrogen-1 Hydrogen-2 Hydrogen-3 Nuclear Symbol 1 H 1 2 H 1 3 H 1 p+ 1 n 0 0 e 1 1 1 2 1

How do we determine the Avg. Atomic Mass? • How do you determine your grade for this class. • Its based on percentages. • Tests/Homework/Participations… every category doesn’t carry the same weight! • It’s a weighted average! • The same idea applies to calculating Avg. Atomic Masses.

• Each isotope has its own percentage or abundance – How much is naturally occurring • Each isotope has its own atomic mass • When calculating the average atomic mass, you use the atomic mass of the isotopes NOT the mass number • The atomic mass is measured in amu’s (atomic mass units) • 1 amu = 1/12 the mass of the carbon-12 atom or 1. 660540 x 10 -27 g

Try this… • Oxygen has three isotopes… Isotope mass (amu) Oxygen-16 15. 995 Oxygen-17 16. 999 Oxygen-18 17. 999 abundance 99. 762 0. 03800 0. 2000 Calculate the average atomic mass of oxygen. Round the avg. atomic mass to the least accurate measurement.

IONS • Take it Mr. F
- Slides: 12