Coulombs Law THE LAW OF FORCE 1 Coulombs

Coulomb’s Law THE LAW OF FORCE! 1
Coulomb’s Law 2

Coulomb’s Law • Any two charged objects will create a • • • force on each other. Opposite charges will produce an attractive force while similar charges will produce a repulsive force. The greater the charges, the greater the force. The greater the distance between them, the smaller the force. 3

Coulomb’s Law k. C q 1 q 2 Fe = 2 r 4

Coulomb’s Law F the force on each charge k the electrostatic constant e q 1 the quantity of charge 1 measured in coulombs q 2 the quantity of charge 2 measured in coulombs r the radius of separation from center of one charge to the center of the other. 5

Coulomb’s Law • The forces are force pairs of each other so they will always be equal in size and opposite in direction. • Newton’s 3 rd Law 6

Coulomb’s Law 1 Coulomb = 6. 3 x 1018 fundamental charges -or 1 fundamental charge = 1. 6 x 10 -19 Coulomb (this is the charge on one electron or one proton) 7

Coulomb’s Law The formula for Coulomb's law is very similar to Newton's Universal Law of Gravitation. 8

Coulomb’s Law Electric Force is a field force Gravity is also a field force Similarities? – Both are proportional to the mass (charge) of the objects involved – Both are inversely proportional to the square of the distance between the objects Differences? – Gravitational forces are only attractive; electrical forces may be either attractive or repulsive. – Electric force is WAY stronger! 9

10

11

According to Coulomb's law, a pair of particles that are moved twice as far apart will experience forces that are A. half as strong. B. one quarter as strong. C. twice as strong. D. four times as strong. 12
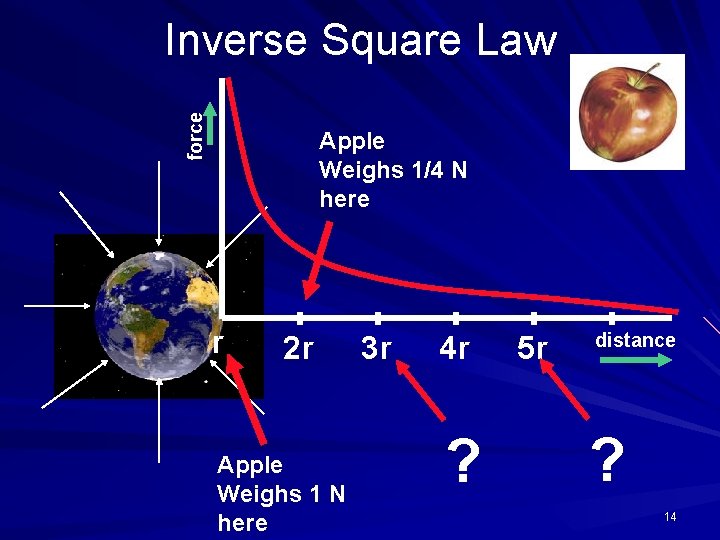
force Inverse Square Law Apple Weighs 1/4 N here r 2 r Apple Weighs 1 N here 3 r 4 r ? 5 r distance ? 14
- Slides: 13