Coulombs Law SWBAT calculate the electrostatic force acting

Coulomb's Law SWBAT calculate the electrostatic force acting on an object.

Do Now

HW Answers: Practice- Conservation of Charge 1. (-) in knob, (+) on leaves 2. Gains 3. Loses 4. a) 13 b) Negative c) -2. 08 x 10 -18 C 5. a) -1. 5 C b) 9. 38 x 1018 C excess electrons c) +7 C sphere gained electrons 6. a) +6. 5 C b) 4. 06 x 1019 C excess protons c) +9 C sphere gained electrons

Conductors & Insulators A conductor’s electrons are free to move around the object (ex. metals). An insulator’s electrons are not free to move around the object, but can move around inside the individual atoms.
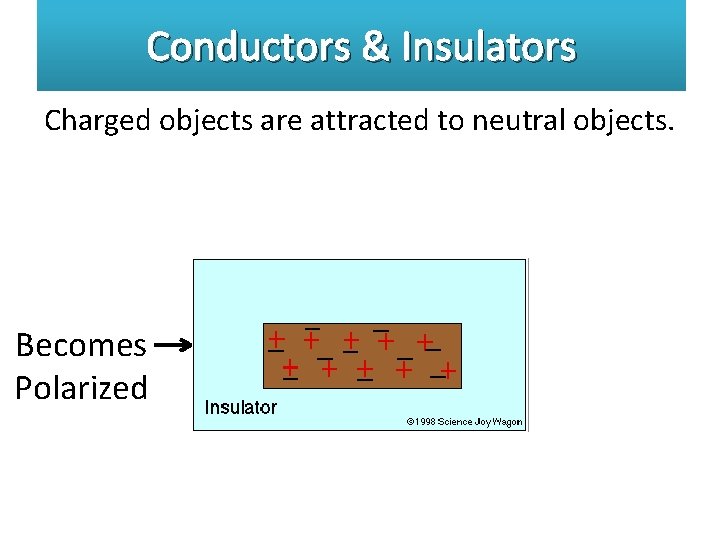
Conductors & Insulators Charged objects are attracted to neutral objects. Becomes Polarized

Why do we get shocked more in the winter?

Critical Thinking Is there a force between two charged objects? How can you tell? Give examples you have seen in this class or in your everyday life.

We can see objects move when charged objects are brought close to them. In other words a mass is being accelerated So there must be some electrostatic force between them.

Coulomb’s Experiment Charles-Augustin Coulomb used a torsion balance to examine the relationship between electrostatic force and 2 different variables: 1. The Magnitude of Charges (q) 2. The Distance between Charges (r) https: //www. youtube. com/watch? v=_5 Vp. Ije-R 54 https: //www. youtube. com/watch? feature=player_detail
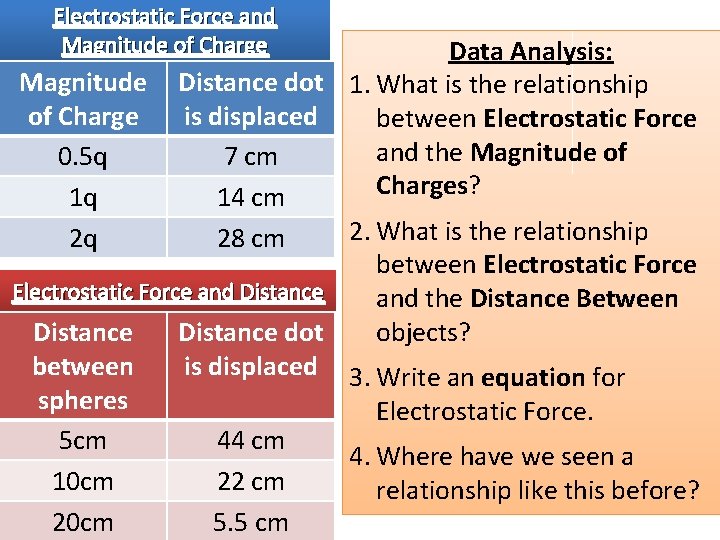
Electrostatic Force and Magnitude of Charge Data Analysis: Magnitude Distance dot 1. What is the relationship of Charge is displaced between Electrostatic Force and the Magnitude of 0. 5 q 7 cm Charges? 1 q 14 cm 2. What is the relationship 2 q 28 cm between Electrostatic Force and Distance and the Distance Between objects? Distance dot between is displaced 3. Write an equation for spheres Electrostatic Force. 5 cm 44 cm 4. Where have we seen a 10 cm 22 cm relationship like this before? 20 cm 5. 5 cm

Coulomb’s Law Fe = Electrostatic Force k = Electrostatic Constant: 8. 99 x 109 N*m 2/C 2 q 1 and q 2 = the charges of the 2 objects r = the distance between the centers of the 2 objects Where have we seen this relationship before?

The Law of Universal Gravitation Fg = Gravitational Force G = Gravitational Constant: 6. 67 x 10 -11 N*m 2/kg 2 m 1 and m 2 = the masses of the 2 objects r = the distance between the centers of the 2 objects This relationship comes up again and again in physics!!!

Reference Table Where is Coulomb’s Law? Where is the Electrostatic Constant (k)?
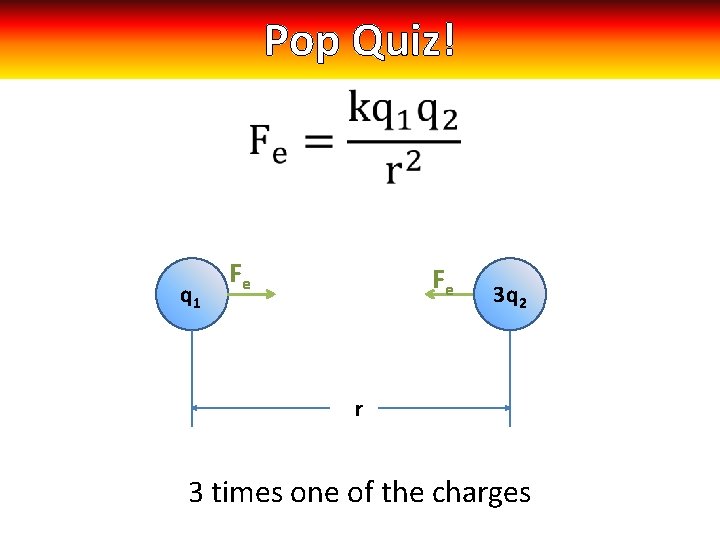
Pop Quiz! q 1 Fe Fe 3 q 3 q 22 r 3 times one of the charges
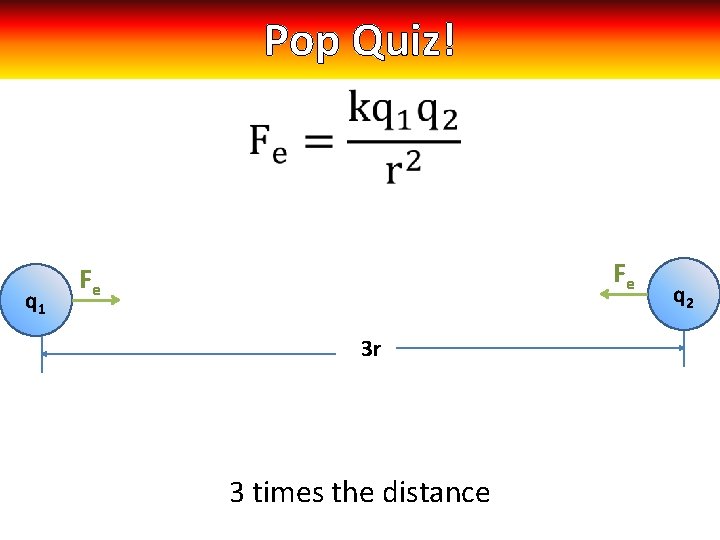
Pop Quiz! q 1 Fe Fe 3 r 3 times the distance q 2
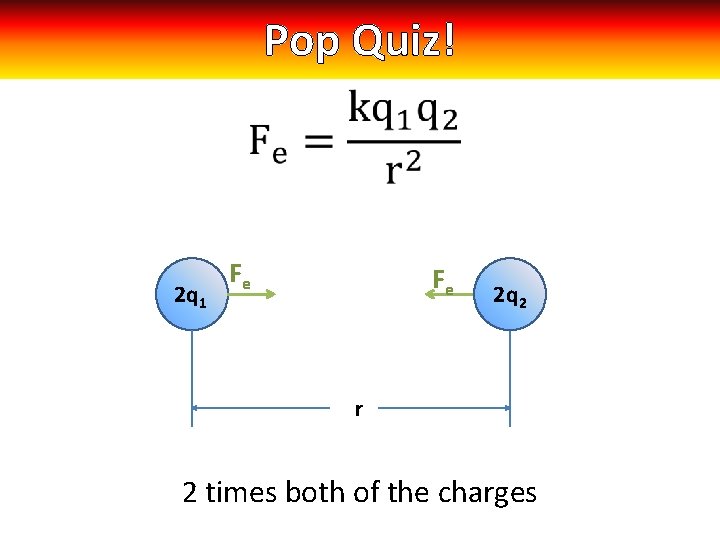
Pop Quiz! 2 q 1 Fe Fe 2 q 3 q 22 r 2 times both of the charges
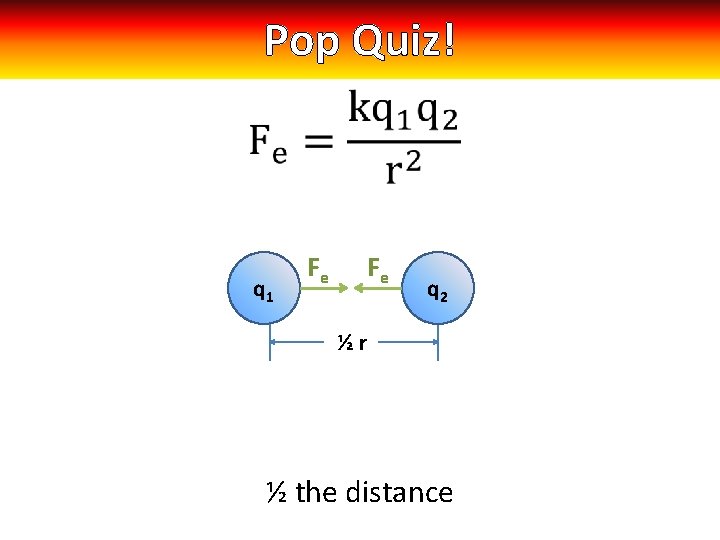
Pop Quiz! q 1 Fe Fe q 2 ½r ½ the distance
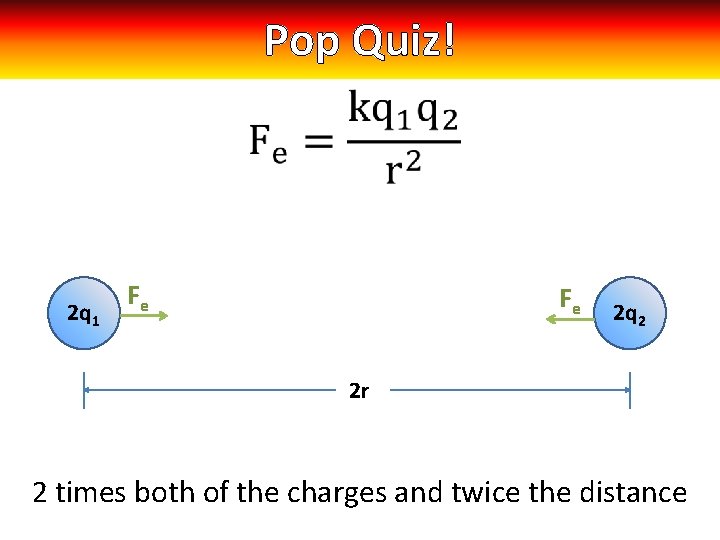
Pop Quiz! 2 q 1 Fe Fe 2 q 2 2 r 2 times both of the charges and twice the distance
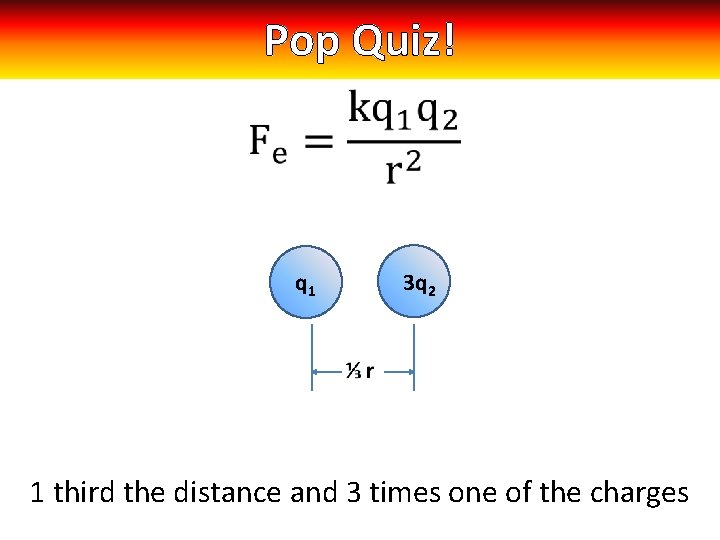
Pop Quiz! q 1 3 q 2 1 third the distance and 3 times one of the charges

Pop Quiz! ½ q 1 Fe Fe 4 q 2 2 r ½ one charge, 4 times the other, and twice as far

Example 1 Two identical conducting spheres are placed with their centers 0. 30 m apart. One is given a charge of +12 x 10 -9 C and the other is given a charge of -18 x 10 -9 C. a. Find the electric force exerted on one sphere by the other. b. The spheres are connected by a conducting wire. After equilibrium has occurred, find the electric force between the two spheres.

Example 2 Two identical conducting spheres are placed with their centers 1. 2 m apart. One is given a charge of +7. 3 μC and the other is given a charge of -4. 5 μC. a. Find the electric force exerted on one sphere by the other. b. The spheres are connected by a conducting wire. After equilibrium has occurred, find the electric force between the two spheres.

Exit Ticket What is the electrostatic force between the two charged spheres shown below? +3 C Fe Fe -73 q. C 2 2 m What would happen to the electrostatic force is the distance between the spheres is doubled and one charge is tripled?

Practice- Coulomb’s Law
- Slides: 24