CORYNEBACTERIU M DIPTHERIAE GRAM POSITIVE RODS AEROBIC CORYNEBACTERIUM

CORYNEBACTERIU M DIPTHERIAE

GRAM POSITIVE RODS: • AEROBIC

CORYNEBACTERIUM DIPTHERIAE • 1826 (Bretonneau) recognised it as a clinical entity – ”diphtherite” , diptheros meaning leather • Tough leathery pseudomembrane formed in disease • Klebs–Loffler bacilli • 1888 – Roux and Yersin discovered the diphtheria exotoxin and established its pathogenic effect

DIPTHERIA: • TOXIN MEDIATED DISEASE. • TOXEMIA, NONINVASIVE BACILLI, TOXIN SPREADS THROUGH BLOODSTREAM TO VARIOUS ORGANS. • TYPES: 1) Respiratory 2) Cutaneous 3) systemic complications

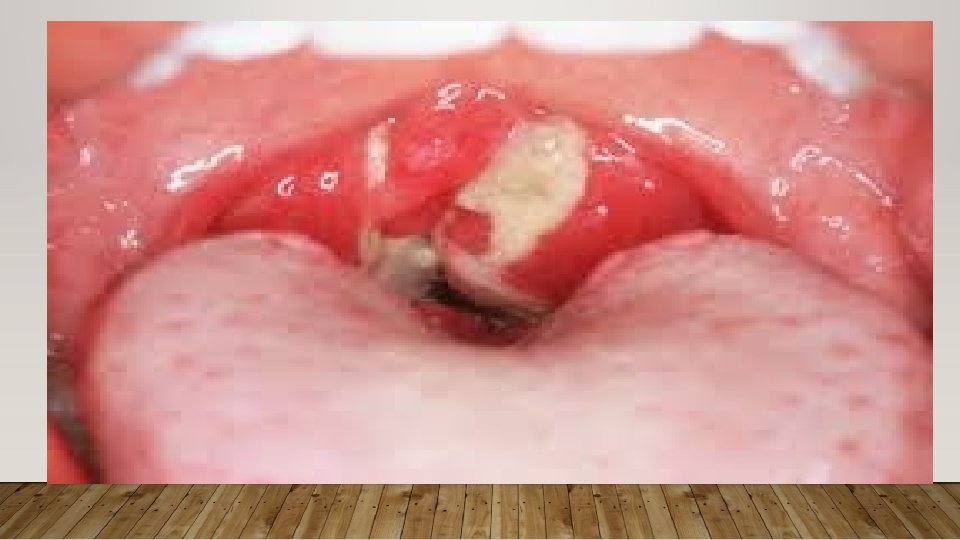
• Respiratory diphtheria: • Most common • Tonsils, pharynx, nose, larynx, conjunctiva, vulvovaginal • IP is 3 -4 days • Toxin inflammatory response Necrosis of epith. lined by tough leathery greyish white PSEUDOMEMBRANE • Exudate formation Mucosal ulcers pseudomembrane made up of inner band of fibrin surrounded by neutrophils, RBC’s and bacteria • Adherent to mucosal base and bleeds on removal • In severe cases extends upto larynx and bronchial airway obstruction fatal airway • Bull neck appearance due to massive tonsillar swelling. Patient presents with foul breath, thick speech and stridor.
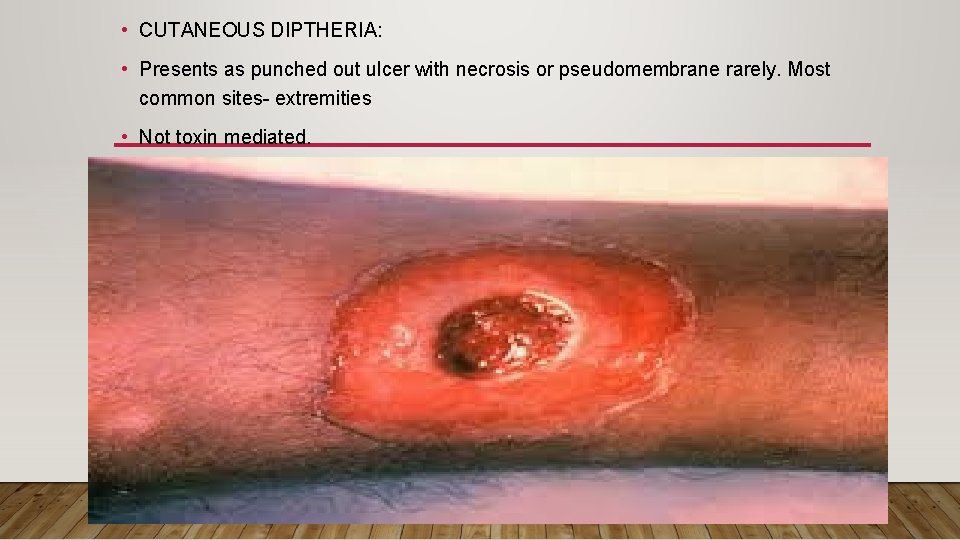
• CUTANEOUS DIPTHERIA: • Presents as punched out ulcer with necrosis or pseudomembrane rarely. Most common sites- extremities • Not toxin mediated.

• SYSTEMIC COMPLICATIONS: • Neurological – cranial nerve involvement, peripheral neuropathy, ciliary paralysis • Myocarditis asso with arrhythmias and dilated cardiomyopathy. • Pneumonia • Renal failure • Encephalitis • Cerebral infarction • Pulmonary embolism

LABORATORY DIAGNOSIS: • Diagnosis mainly clinical. • Laboratory diagnosis necessary only for: 1. Confirmation of clinical diagnosis 2. Initiating control measures 3. Epidemiological purposes • Consists of isolation of bacilli and toxin demonstration

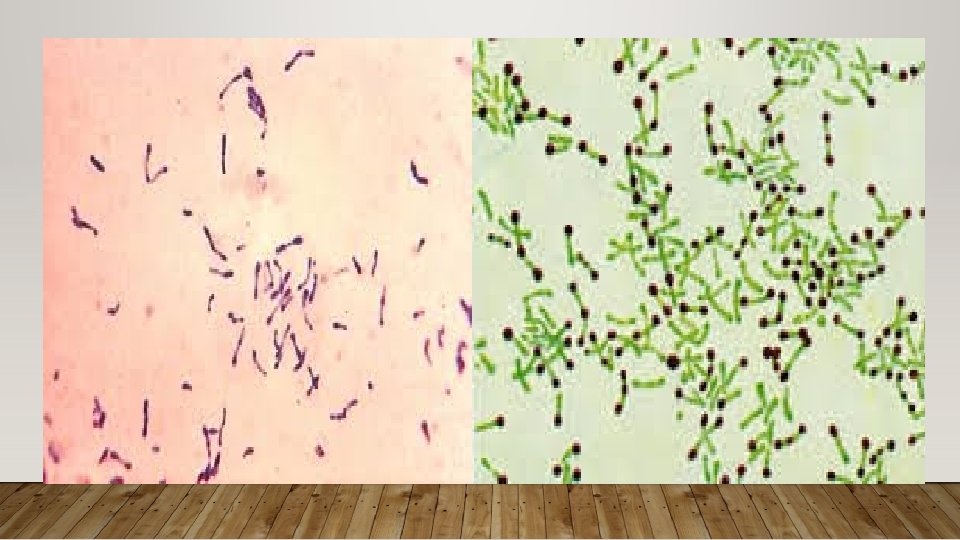
• Specimen: 1. Throat swab containing fibrinous exudate 2. portion of pseudomembrane 3. Nose or skin specimen if infected • Direct smear microscopy: in - Gram stain: irregular stained, club shaped gram positive bacilli, 3 -6 microns length, arranged in CHINESE LETTER or CUNEIFORM arrangement. - Albert’s stain: more specific for C. diptheriae, the bacilli appear green with bluish black metachromatic granules. The granules are also k/as Babes Ernst bodies or Volutin granules. They are storage granules composed of polymetaphosphates - Other stains: Neisser’s stain and Ponder’s stain.

• Culture: Fastidious, aerobic and facultative anaerobe. Grows best in enriched media like blood agar, chocolate agar, Loeffler’s serum slope. On blood agar- small circular white sometimes haemolytic colonies. On Loeffler’s serum slope- small, circular, glistening, white with a yellow tinge in 68 hrs SELECTIVE MEDIUM- Potassium tellurite agar- black coloured colonies. C. ulcerans & C. pseudotuberculosis also produce black coloured colonies. - Tinsdale medium- black colonies surrounded by brown halo.


• Biochemical identification: - catalase positive, oxidase negative. - ferments glucose, maltose and starch with production of acid and no gas. - Pyrazinamidase test- positive for C. diptheriae, C. ulcerans, C. pseudotuberculosis. - Urease test- C. diptheriae is negative whereas C. ulcerans and C. pseudotuberculosis are positive

VIRULENCE FACTOR: diphtheria toxin • Primary virulent factor responsible for disease. • Virulent strain produces powerful exotoxin • Pathogenic effects are due to toxin. • The toxin is synthesized in precursor form. • Activation – accomplished by proteases in infected tissues.

TOXIN • Two fragments A (active subunit) and B (binding subunit). • B subunit binds to the host cell receptors like EGF and facilitates entry of factor A. • Factor A causes ADP ribosylation of EF-2 (elongation factor 2). • This leads to inhibition of EF-2, further inhibits translation and halts the protein synthesis.

• Toxin acts by inhibiting protein synthesis • Toxin fragment ‘A’ inhibits polypeptide chain elongation in the presence of NAD, by activating the elongation factor EF-2


• Factors regulating toxin production: 1. Phage coded- DT is coded by bacteriophage called beta corynephage carrying tox gene. 2. Iron concentration- optimum concentration 0. 1 mg/L. higher levels inhibit toxin synthesis. 3. DT repressor gene 4. Biotypes

Toxoid for vaccination: • Toxin is antigenic but cant be given directly as it is virulent. • Toxoid is form of toxin in which virulence is lost while antigenicity is retained. • It is toxoided by formalin, acidic p. H, prolonged storage. • Park William 8 strain used as source of toxin for vaccine production.

Demonstration of toxin: • Diagnosis is incomplete without demostration of toxin. It can be done by two methods: A. In vivo tests: 1. Subcutaneous test-0. 8 ml culture broth injected SC in 2 guinea pigs. One protected with antitoxin 500 units. If test strain virulent, unprotected animal dies in 4 days. 2. Intracutaneous test- 0. 1 ml culture broth injected intracutaneously in 2 guinea pigs. One protected with 500 units of antitoxin acts as control. Other animal is given 50 units antitoxin intraperitoneally after 4 hrs of skin test. Toxigenicity indicated by necrosis of inoculated site in 48 -72 hrs and no change in control animals.

B. In vitro tests: 1. ELEK’S gel precipitation test: -immunodiffusion type of test. - rectangular strip of filter paper soaked in diphtheria antitoxin 1000 units/ml. -Placed on surface of 20% horse serum agar. -Test strain streaked at right angles to filter paper strip and incubated at 37 o. C for 24 hrs. -If toxigenic, toxin diffuses in the agar meets with the antitoxin and produces arrow shaped precipitation band. -Non toxigenic strains produces no lines of precipitation.


2. Other tests: a. detection of tox gene by PCR b. detection of diphtheria toxin by ELISA or ICT c. cytotoxicity produced on cell lines

Typing of C. diptheriae: • Biotyping/ Macleod’s classification- divided into 4 biotypes gravis, intermedius, mitis, belfanti • Serotyping- based on antigens such as mycolic acid, polysaccharide antigen etc • Bacteriophage typing- based on sensitivity of strains to lytic action of standard phages • Bacteriocin typing- ability of test strain producing bacteriocin to kill indicator strains of bacteria • Molecular typing – PFGE, RFLP

• Three biotypes • Gravis- short, no granules. Daisy head colony. Non emulsifiable colony, starch fermentation positive. 100% toxigenic. • Intermedius- long barred, poor granules, frogs egg colony, starch fermentation negative, moderate virulence, 90 -95% toxigenic. • Mitis- long curved, prominent granules, poached egg colony, soft buttery easily emulsifiable colony, mild virulence, 80 -85% toxigenic.

TREATMENT: • Started immediately on clinical suspicion • Passive immunisation with anti diptheritic serum is the treatment of choice • Antibiotics have no major role once the toxin is secreted. If initiated early i. e. within 6 hrs of infection Penicillin or Erythromycin is the drug of choice. It helps in precvention of further release of toxin by killing the bacteria. It is useful in treatment of cutaneous diphtheria. In carriers erythromycin is the drug of choice.

PROPHYLAXIS: • Active immunisation done with diphtheria toxoid- induces antitoxin formation. • Vaccine not useful for prevention of cutaneous infection and elimination of carrier state. • Two types of vaccine- single and combined. • Combined vaccines- DPT, Da. PT, DT. • DPT- choice for vaccinating infants. Pertussis component acts as adjuvant and increases the immunogenicity of DT and TT. • Plain formol toxoid or alum adsorbed toxoid

• Administration of vaccine: • Included under National Immunisation Schedule. • Total 5 doses- 3 doses at 6, 10 and 14 weeks of birth. Two booster doses at 1624 months and 5 years respectively. • Site- deep IM at anterolateral aspect of thigh. Gluteal region not preferred • Preservative- Thiomersal • Storage- 2 -8 o C. • Dose – 0. 5 ml. • Protective titre of >= 0. 01 units/ml of antitoxin is protective.

• Adverse reactions 1. Mild- fever and local reaction 2. Severe- the pertussis component is encephalitogenic. So the vaccine not recommended after 6 years of age. • Acellular pertussis is recommended after 6 years of age. • Contraindications 1. Hypersensitivity to previous dose 2. Progressive Neurological disorder.

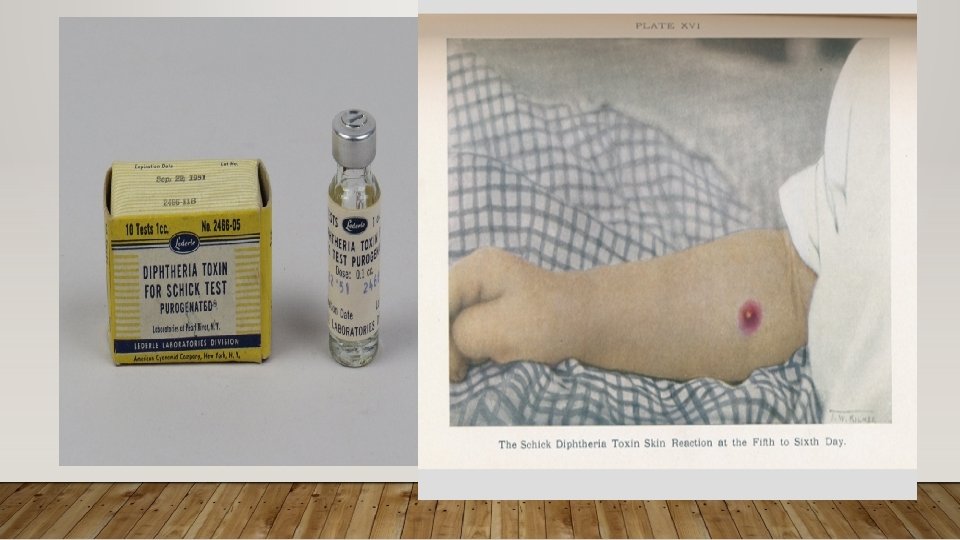
• SCHICK TEST: • Named after the discoverer Bela Schick. • Toxin-antitoxin mediated test. • Carried out on people before starting immunisation to determine susceptible individuals. • 0. 1 ml of diphtheria toxin injected intradermally in the arm. • Positive reaction indicated by development of erythema and induration aound the site of injection.

DIPTHEROIDS: • Nondiptherial corynebacteria. • Exist as normal commensals in throat, skin and conjunctiva. • Cause invasive infections in immunocompromised host. • Can be differentiated from C. diptheriae by following features: 1. stain uniformly 2. Palisade arrangement 3. Absence of metachromatic granules except C. xerosis. 4. Toxigenicity test is negative. 5. Ferment sucrose also.

Coryneforms rarely pathogenic to man are: • C. ulcerans and C. pseudetuberculosis caused localised infections in throat. • C. ulcerans cause infection in cows. C. pseudotuberculosis cause pseudotuberculosis in sheep and suppurative lymphadenitis in horses. • C. minutissimum- Erythrasma , infection of skin of groin and axilla. • C. tenuis- c/by formation of pigmented nodules around axillary and pubic hair shaft. • C. jeikeium- colonizes skin of hospitalised patients. Associated with bacteremia. • C. urealyticum- skin commensal, rarely causes UTI in immunocompromised and renal transplant patients. • C. amycolatum- rarely causes catheter related and surgical site infection • C. pseudodiptheriticum- commensal in throat, causes exudative pharyngitis and endocarditis in immunocompromised host

- Slides: 35