Corso CINBO Recent advances in the medical treatment

Corso CINBO Recent advances in the medical treatment of melanoma Roma 21/06/2013 Pre-clinical and clinical aspects of peptide-based melanoma vaccines Giorgio Parmiani San Raffaele Foundation, Milano
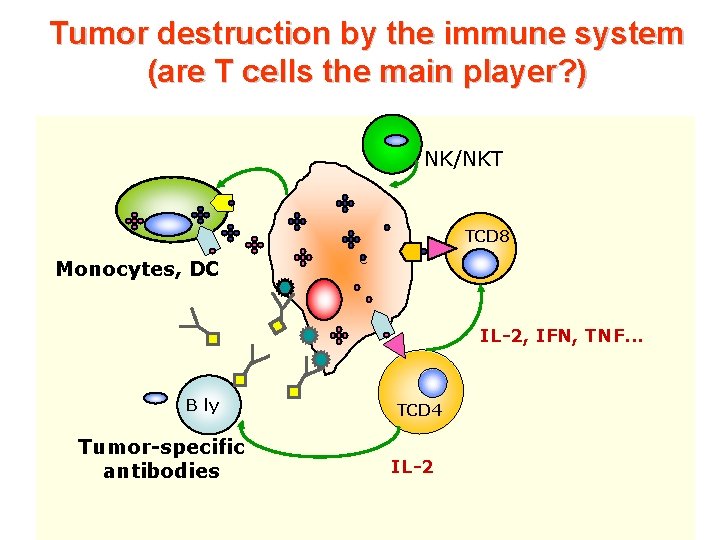
Tumor destruction by the immune system (are T cells the main player? ) NK/NKT TCD 8 Monocytes, DC c IL-2, IFN, TNF. . . B ly Tumor-specific antibodies TCD 4 IL-2

• The Melanoma-Associated Antigens

Melanoma-associated antigens (MAAs) recognized by T cells. Which is the best MAA or combination of? 1. Shared/self/differentiation MAAs (e. g. Mart 1, tyrosinase) 2. Shared/self/cancer testis or germinal MAAs (e. g. MAGE, NY-ESO 1) 3. Universal MAAs (survivin, h. TERT) 4. Mutated, unique MAAs

• These different MAAs have been used as immunogens in clinical trials. • Which was the outcome in terms of toxicity, immune response and clinical response? • The immunogenicity

1 -2. Shared self MAAs • Normal subjects and cancer patients show some form of tolerance to “self” MAAs (immune ignorance, peripheral or central tolerance, low frequency of T cell precursors, T regs). • Tolerance needs to be broken (spontaneously or not) in order to elicit a T cell immune response against “self” MAAs. • Thus, these MAAs are considered to be “weak antigens” though despite being the first to be molecularly characterized and used in patients

In vivo spontaneous tolerance break (immunogenicity) of shared self MAAs. 2 examples
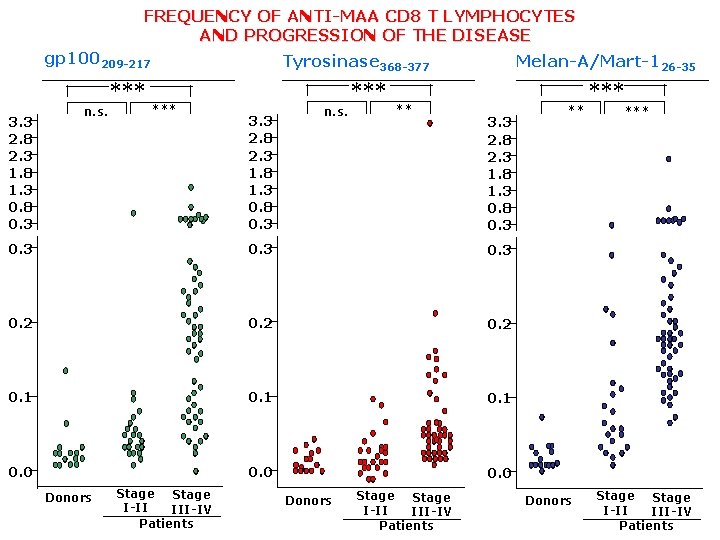
FREQUENCY OF ANTI-MAA CD 8 T LYMPHOCYTES AND PROGRESSION OF THE DISEASE gp 100209 -217 3. 3 2. 8 2. 3 1. 8 1. 3 0. 8 0. 3 n. s. *** Tyrosinase 368 -377 *** 3. 3 2. 8 2. 3 1. 8 1. 3 0. 8 0. 3 n. s. *** ** Melan-A/Mart-126 -35 3. 3 2. 8 2. 3 1. 8 1. 3 0. 8 0. 3 0. 2 0. 1 0. 0 Donors Stage I-II III-IV Patients ** Donors *** Stage I-II III-IV Patients

CONCLUSIONS • A hierarchy exists in the spontaneous recognition of “self” TAAs. • Recognition of “self” TAAs increases with the increased tumor burden (e. g. Melan. A/MART 1)

Break of tolerance to shared self TAAs by vaccination

Peptide-based vaccines • Advantages: • Sequence and biochemical features are known • Easy to synthesize (large availability) • Allow a specific immune-monitoring of the patient response to vaccine • Allow assessing the expression of targeted TAA in patients’ tumor cells

Peptide-based vaccines (cont. ) • Disadvantages: • Easy degradable in absence of adjuvants • Require appropriate HLA allele to be recognized by T cells (patient selection) • Induce T cells that may not recognize tumor cells • Costs (40 -160’ 000€/each)
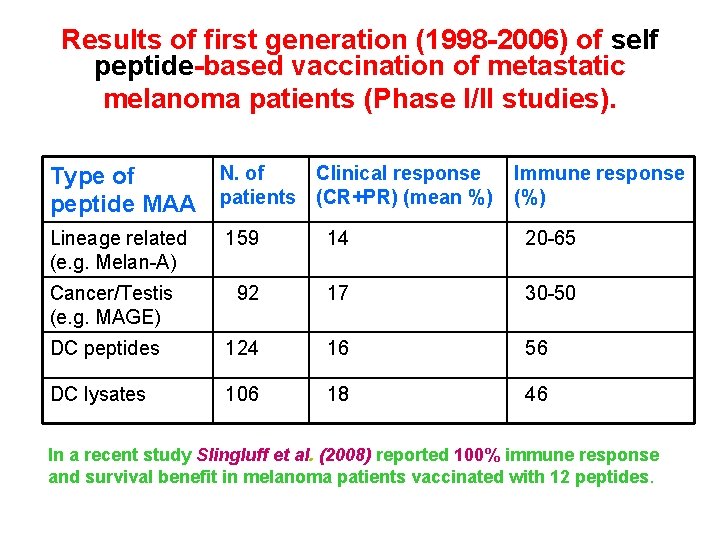
Results of first generation (1998 -2006) of self peptide-based vaccination of metastatic melanoma patients (Phase I/II studies). N. of Clinical response Immune response Type of peptide MAA patients (CR+PR) (mean %) (%) Lineage related (e. g. Melan-A) 159 14 20 -65 92 17 30 -50 DC peptides 124 16 56 DC lysates 106 18 46 Cancer/Testis (e. g. MAGE) In a recent study Slingluff et al. (2008) reported 100% immune response and survival benefit in melanoma patients vaccinated with 12 peptides.
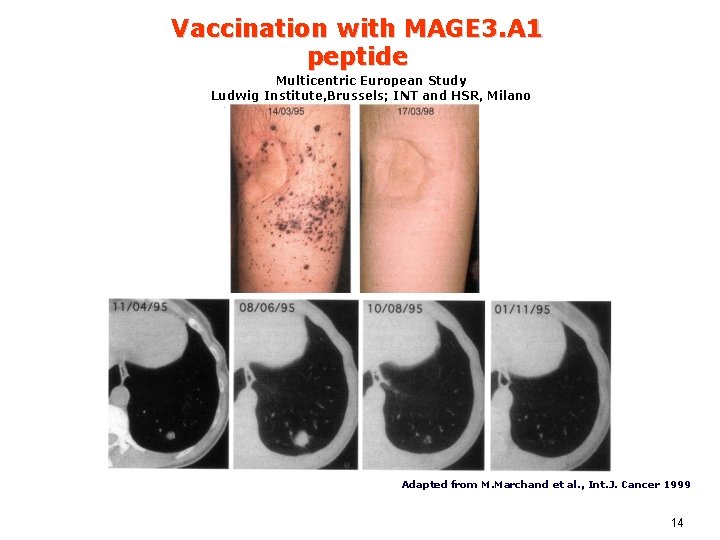
Vaccination with MAGE 3. A 1 peptide Multicentric European Study Ludwig Institute, Brussels; INT and HSR, Milano Adapted from M. Marchand et al. , Int. J. Cancer 1999 14

Use of dendritic cells: which progress ? see Ridolfi’s presentation

Disappointment followed by a drop in the interest in new studies • Which are the reasons of such negative clinical results of active immunotherapy of cancer?

Reasons for the limited clinical response

Factors that interfere with the T cellmediated anti-tumor response Tumor (Immunosubversion) • Lack of antigen or downregulation of HLA • Dysfunction of antigen presentation • Release of immunesuppressive factors (IL-10, TGFβ, VEGF) • Tumour counterattack (Fas/Fas. L) • IDO, SPARC • Expression of Fox. P 3, CTLA 4 • Tumor ER stress • NFAT 1, Exosomes Immune system • Immune anergy or ignorance • Lack of tissue homing molecules; defective adhesion • T-cell receptor dysfunction • Inactivation of T-cells within the tumor environment (granzyme B) • T-regulatory cells • MDSC • Epithelial/mesenchimal transition • Tie+ Monocytes dysfunction

• …but, despite this long list of obstacles, under some conditions immune cells can manage to find and sometime destroy cancer cells. • The understanding of the escape mechanism helps to inhibit or downregulate them in the clinic thus improving efficacy.

• Possible examples
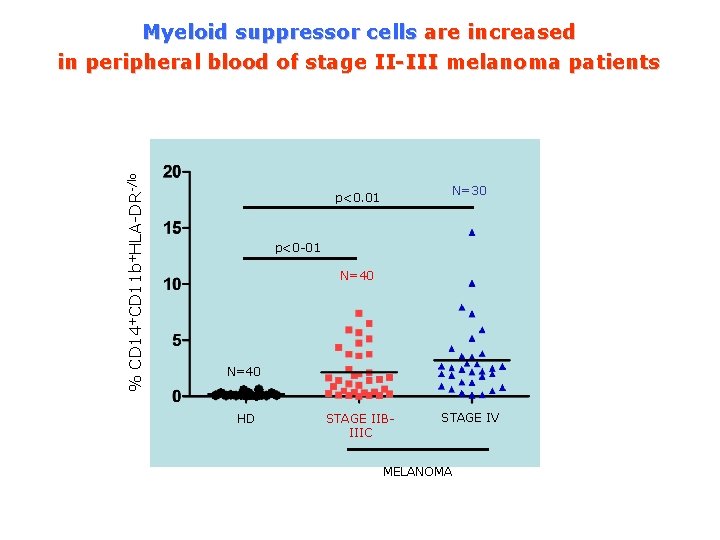
% CD 14+CD 11 b+HLA-DR-/lo Myeloid suppressor cells are increased in peripheral blood of stage II-III melanoma patients N=30 p<0. 01 p<0 -01 N=40 HD STAGE IIBIIIC STAGE IV MELANOMA
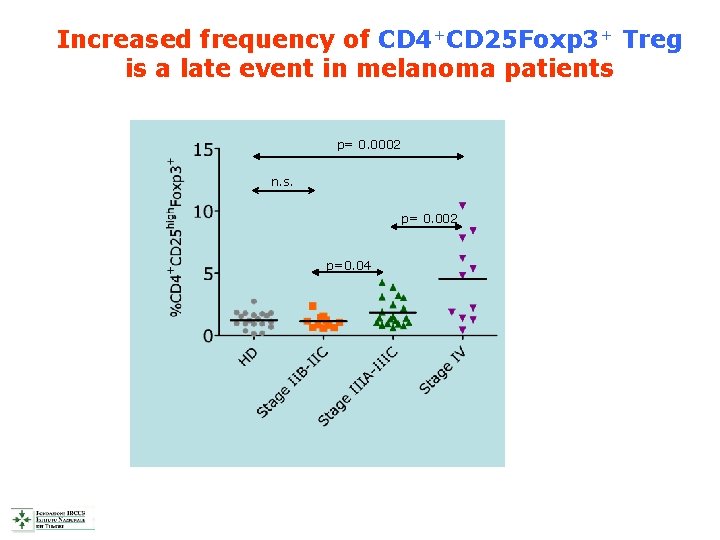
Increased frequency of CD 4+CD 25 Foxp 3+ Treg is a late event in melanoma patients p= 0. 0002 n. s. p= 0. 002 p=0. 04
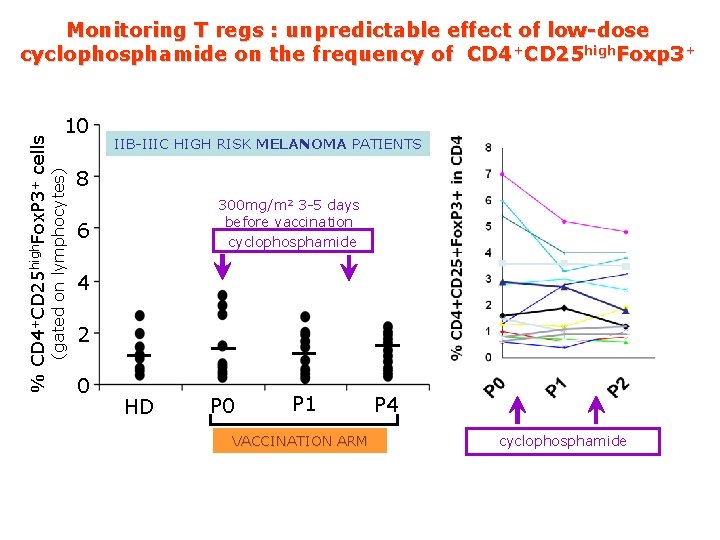
10 (gated on lymphocytes) % CD 4+CD 25 high. Fox. P 3+ cells Monitoring T regs : unpredictable effect of low-dose cyclophosphamide on the frequency of CD 4+CD 25 high. Foxp 3+ IIB-IIIC HIGH RISK MELANOMA PATIENTS 8 300 mg/m 2 3 -5 days before vaccination cyclophosphamide 6 4 2 0 HD P 0 P 1 VACCINATION ARM P 4 cyclophosphamide

• Cyclophosphamide is effective in a different trial with RCC patients (Walter et al. , Nature Med 2013)
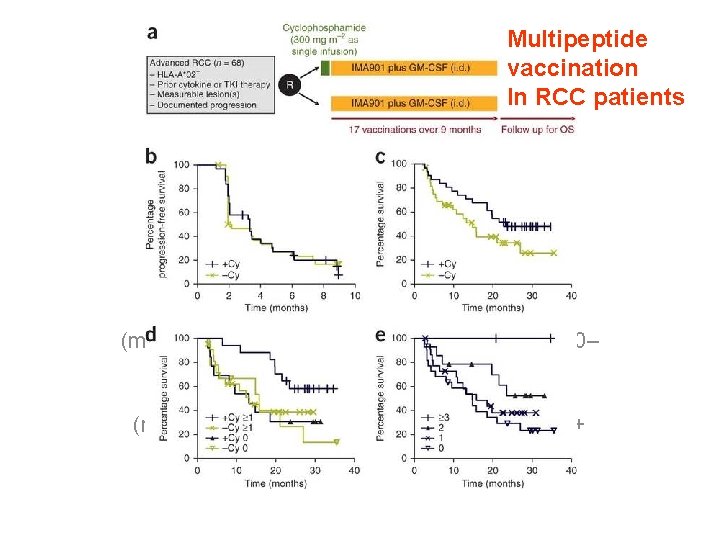
Multipeptide vaccination In RCC patients de 010203040020406080100 Time (months)Percentage survival 3+Cy 1+Cy 0– Cy 1210010203040020406080 Percentage survival 100 Time (months)bc 0246810020406080100 Time (months)Percentageprogression-free survival+

Conclusions 1. Myeloid-derived suppressor cells are increased in the peripheral blood of stage II-III melanoma patients 2. Increased frequency of CD 4+CD 25 Fox. P 3+ Tregs is a late event in melanoma patients Not clear the effect of low-dose cyclophosphamide on the frequency of CD 4+CD 25+Foxp 3+ 3.

Recent progress in cancer vaccines. The Renaissance of cancer immunotherapy : Phase II-III positive randomized trials: • Melanoma (gp 100) • B cell lymphoma • Prostate cancer (Sipuleucel, Provenge) • NSCLC (MAGE) • Renal Cell Carcinoma (IMA 901)

Phase III study of gp 100 peptide vaccine in melanoma • A phase III multi-institutional randomized study of immunization with the gp 100 (210 M) peptide followed by high-dose of IL -2 compared with high dose IL-2 alone in patients with metastatic melanoma. • Schwartzentruber et al. NEJM 2011

Phase III study of gp 100 peptide vaccine (cont. ) • 21 centers; total of 185 patients • Stage IV or locally advanced stage III, HLA -A*0201 • Therapy. IL-2: 720’ 000 IU/kg/dose+/Gp 100 (210 M) peptide+Montanide • Results. High toxicity (IL-2); RR 22. 1% vs. 9. 7% (P=0. 0223); PFS: 2. 9 vs. 1. 6 mos (P=0. 010); Median OS: 17. 6 vs. 12. 9 mos (P=0. 096)
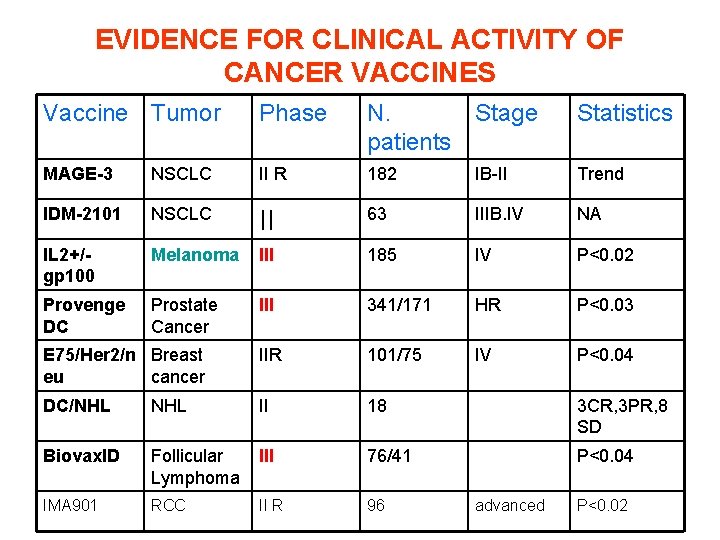
EVIDENCE FOR CLINICAL ACTIVITY OF CANCER VACCINES Vaccine Tumor Phase N. patients Stage Statistics MAGE-3 NSCLC II R 182 IB-II Trend IDM-2101 NSCLC II 63 IIIB. IV NA IL 2+/gp 100 Melanoma III 185 IV P<0. 02 Provenge DC Prostate Cancer III 341/171 HR P<0. 03 E 75/Her 2/n Breast eu cancer IIR 101/75 IV P<0. 04 DC/NHL II 18 3 CR, 3 PR, 8 SD Biovax. ID Follicular Lymphoma III 76/41 P<0. 04 IMA 901 RCC II R 96 advanced P<0. 02
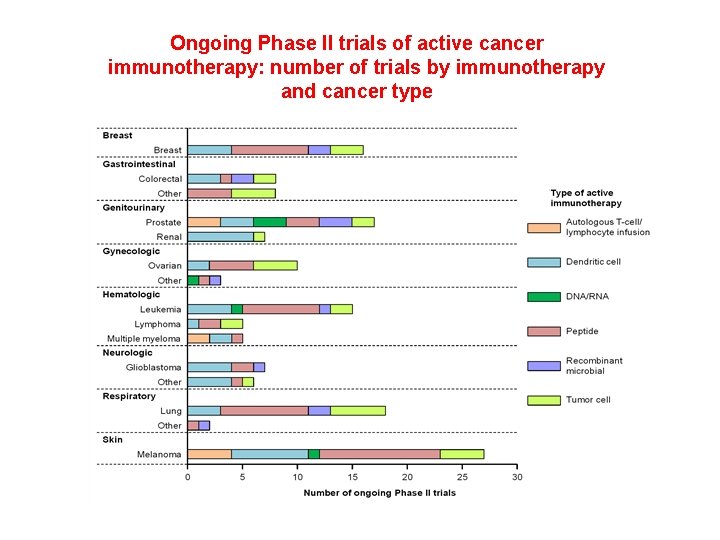
Ongoing Phase II trials of active cancer immunotherapy: number of trials by immunotherapy and cancer type

Self peptide-based melanoma vaccines: Conclusions • A phase III studys showed statistically significant benefit for vaccinated patients. • The use of a) multipeptides, b) patients with limited tumor burden, c) a concomitant modulation of immune suppressive cells, d) combination of chemotherapy/anti-angiogenic agents and vaccines are providing new and promising clinical results.

New targets in cancer vaccination • Stroma: a site of complex and often immune inhibitory interactions among tumor cells, infiltrating lymphocytes, macrophage and fibroblast and different soluble factors (e. g. chemokines). • Cancer stem cells

The role of stroma • Stromal cells take up tumor-derived exosomes becoming susceptible (H. Schreiber) or resistant ( G. Parmiani, L. Rivoltini) to CTL. • Endothelial cell antigens (e. g. VEGFR-2; D. Schadendorf). • Fibroblast antigens • Inflammatory/Immune cells (Tcells, DCs, Monocytes, MDSC, mast cells? ) • Chemokines (CCL 2 -5, -19, -21; Cx. CL 9 -13; T. F. Gajewski) • Aim: Altering the tumor stroma to the benefit of the host

Cancer stem cells: A new target of immunotherapy? • Objectives: Identification of molecules with immunological relevance expressed by cancer stem cells and validation of their role as target molecules of immunotherapy. • See Ruggero De Maria presentation?

Rationale for a new generation of cancer vaccines • • Early disease Multiple antigens New TLR targeting adjuvants (Cp. G, HSPs) Down-regulation of Tregs and/or Myeloid Derived Suppressor Cells • Immune-monitoring in blood, LNs and tumor tissue. • Assessment of patient polymorphisms and tumor gene signatures • Combination with otherapeutic agents

• Combination with otherapeutic agents (e. g. antivascular agents)
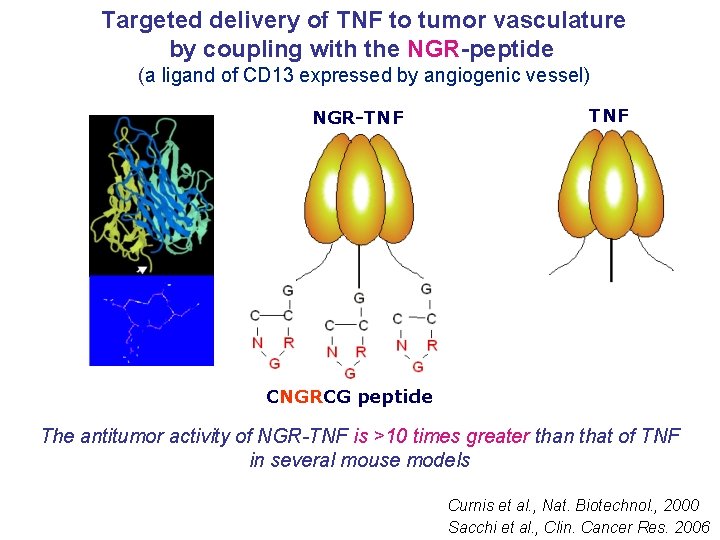
Targeted delivery of TNF to tumor vasculature by coupling with the NGR-peptide (a ligand of CD 13 expressed by angiogenic vessel) NGR-TNF CNGRCG peptide The antitumor activity of NGR-TNF is >10 times greater than that of TNF in several mouse models Curnis et al. , Nat. Biotechnol. , 2000 Sacchi et al. , Clin. Cancer Res. 2006
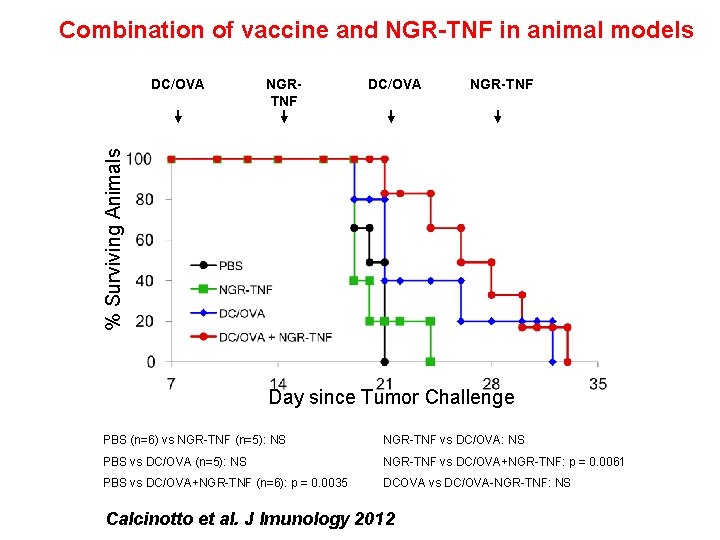
Combination of vaccine and NGR-TNF in animal models NGRTNF DC/OVA NGR-TNF % Surviving Animals DC/OVA Day since Tumor Challenge PBS (n=6) vs NGR-TNF (n=5): NS NGR-TNF vs DC/OVA: NS PBS vs DC/OVA (n=5): NS NGR-TNF vs DC/OVA+NGR-TNF: p = 0. 0061 PBS vs DC/OVA+NGR-TNF (n=6): p = 0. 0035 DCOVA vs DC/OVA-NGR-TNF: NS Calcinotto et al. J Imunology 2012

• A pilot phase I and II study of NGRh. TNF in combination with a peptidebased vaccine (MAGE. A 3, NA 17. A 2) for metastatic melanoma (NGR/Vax/01) • PI: Giorgio Parmiani, MD

Inclusion criteria (cont. ) • HLA-A*0201 - and/or HLA-A 1 -positive typing on PBMCs. • Melanoma cells expressing the NA 17. A 2 or MAGE-3. A 1 melanoma antigens in HLA-A 2 or HLA-A 1 patient, respectively, as defined by PCR analysis on frozen or paraffinized tissue.

NGR/Vax/01. Early results in the clinic • Phase I-II of combination of NGR-h. TNF and melanoma peptide vaccine • 9 patients enrolled; • No SAE ; grade I-II local erythema, fever, chills. • Ex-vivo immune response to vaccine 6/8; • Clinical response 5 Long Term SD (4, 5, 8, 11, 15+, 16+, 17+, 22+)
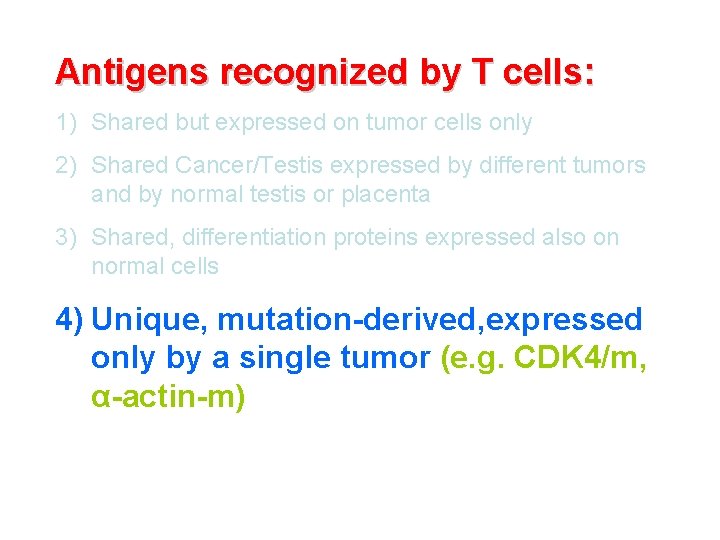
Antigens recognized by T cells: 1) Shared but expressed on tumor cells only 2) Shared Cancer/Testis expressed by different tumors and by normal testis or placenta 3) Shared, differentiation proteins expressed also on normal cells 4) Unique, mutation-derived, expressed only by a single tumor (e. g. CDK 4/m, α-actin-m)
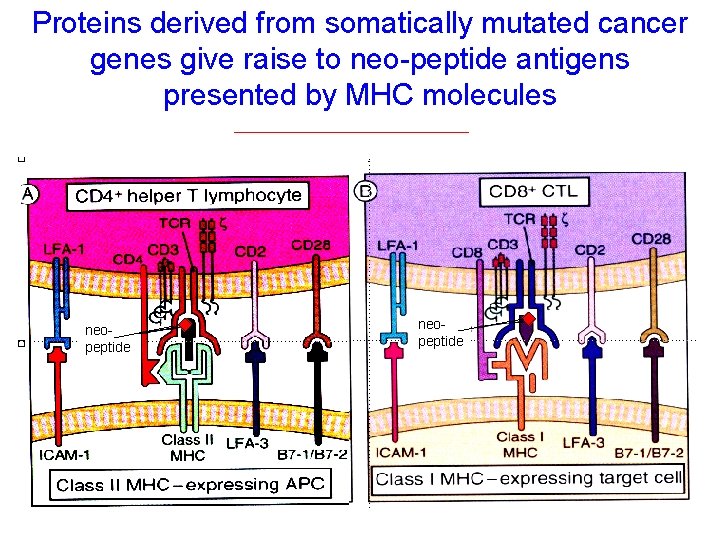
Proteins derived from somatically mutated cancer genes give raise to neo-peptide antigens presented by MHC molecules neopeptide
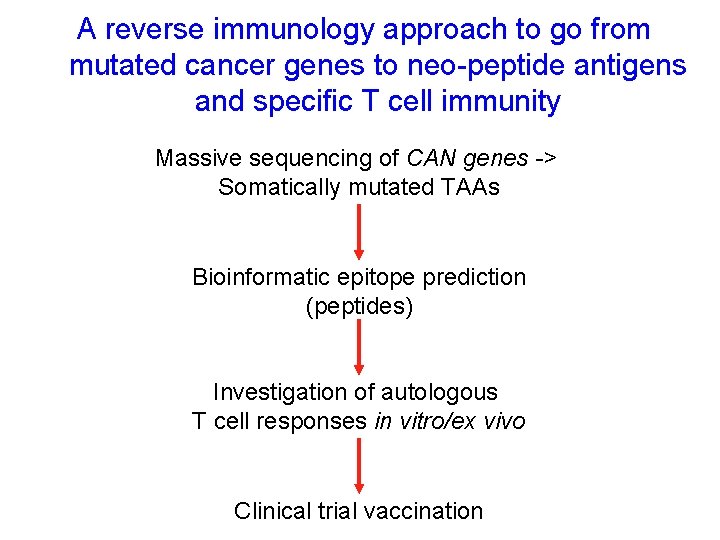
A reverse immunology approach to go from mutated cancer genes to neo-peptide antigens and specific T cell immunity Massive sequencing of CAN genes -> Somatically mutated TAAs Bioinformatic epitope prediction (peptides) Investigation of autologous T cell responses in vitro/ex vivo Clinical trial vaccination

PREDICTED EPITOPES DERIVED FROM APC MUTATION MISSENSE A 3079 T (Y->L) BINDING SOME RAPRESENTATIVE HLA-A ALLELES MUTATED SEQ HLA-A*01 DTPINLSLKY ELDTPINLSL HLA-A*01 11 LDTPINLSLKY ELDTPINLSLK HLA-A*0201 ELDTPINLSLKYSDEQ HLA-A*03 NLSLKYSDE TPINLSLKY LDTPINLSLK ELDTPINLSL HLA-A*1101 DTPINLSLK HLA-A*26 DTPINLSLKY ELDTPINLSL DGELDTPINL HLA-A*6801 DTPINLSLK score W. T. SEQ score 25 14 DTPINYSLKY ELDTPINYSL 25 11 20 14 LDTPINYSLKY ELDTPINYSLK 20 13 20 12 ELDTPINYSLKYSDEQ 20 2 15 13 16 15 NYSLKYSDE TPINYSLKY LDTPINYSLK ELDTPINYSL 5 13 16 12 22 DTPINYSLK 22 28 20 17 DTPINYSLKY ELDTPINYSL DGELDTPINY 28 19 19 24 DTPINYSLK 20 The maximal score for peptides binding HLA-A*0201 is 36. Influenza A matrix GILGFVFTL scores 30

Vaccination with unique TAAs. A new genomic strategy • Cancer cells contain somatic mutations detectable by new sequencing technology (Sjoblom et al. , Science 2006; Parsons et al. , Science 2008). These mutations can generate candidate new T cell epitopes. • Breast and colorectal cancers may accumulate 7 -10 new HLA-A*0201 epitopes that may define an individual tailored polyvalent vaccine (Segal et al. , Cancer Res 2008) • Driver mutations/each melanoma in 120 genes

P. Robbins Trial? • At the moment I am not aware of any ongoing cancer vaccination program based on individually mutated cancer gene -products but I know that several research groups are pursuing this idea.

Acknowledgments • Financial support: • • AIRC, Milano Alleanza contro il Cancro (ACC), Rome Italian Network for Biotherapy of Tumors (NIBIT), Siena European Commission (ATTACK) • Collaborators: • Cristina Maccalli, Vincenzo Russo, Paolo Dellabona, Gloria Sovena, (San Raffaele Scientific Institute, Milano) • Licia Rivoltini, Chiara Castelli, Mario Santinami, (National Tumor Institute, Milano)

• END OF THE PRESENTATION. • Thank you for your attention!



• A pilot phase I and II study of NGRh. TNF in combination with a peptidebased vaccine for metastatic melanoma. • PI: Giorgio Parmiani

Expression of MHC and APM molecules and NKG 2 DLs in GBM-derived CSCs and FBS tumor cells • The expression of: MHC class I and II; Antigen processing machinery (APM), using 21 different m. Abs directed against HLA molecules, their heavy chains, b 2 -microglobulin immunoproteasome, constitutive proteasome subunits, chaperon molecules, TAPs etc. ; NKG 2 DLs; has been tested in 11 different GBM CSCs and, for 5 of them, in their paired tumor cells grown in the presence of FBS (FBS tumor cells).

Antigens recognized by T cells: 1) Shared, self differentiation proteins expressed also on normal cells (e. g. MART-1, CEA, PSA) 2) Shared self Cancer/Testis expressed by different tumors and by normal testis or placenta (e. g. MAGE, NY-ESO-1) 3) Shared, universal TAAs predominantly expressed by tumor cells (e. g. h. TERT, survivin) 4) Unique, expressed only by a single tumor (e. g. CDK 4/m, α-actin-m)

Exploiting known frequently somatically mutated CAN genes in CRC - From massive sequencing of each exons of about 20. 000 protein-coding genes - In 22 + 48 (only some genes) tumor samples - Identified 142 genes that bear non-synonymous mutations with driver characteristics - On average of about 15 CAN-genes are mutated in each CRC Wood LD et al Science 2007
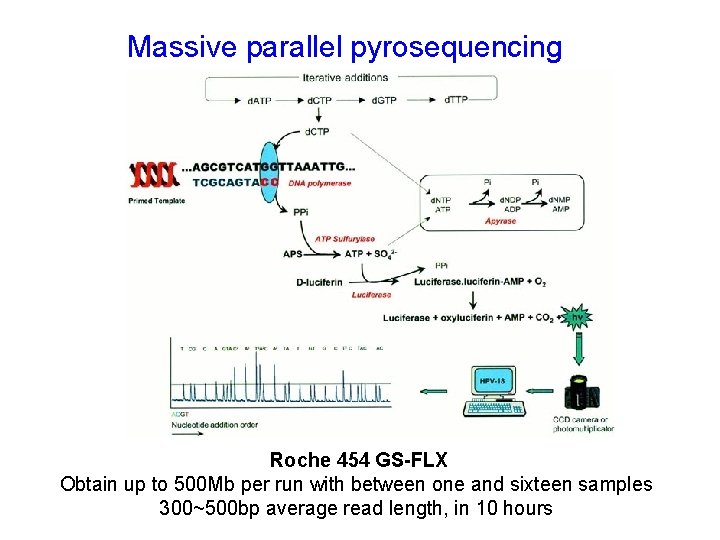
Massive parallel pyrosequencing Roche 454 GS-FLX Obtain up to 500 Mb per run with between one and sixteen samples 300~500 bp average read length, in 10 hours

20 most frequently mutated CAN-gene in CRC Mutated in 100% to 20% of CRCs Mutated in fewer than 5% of CRCs
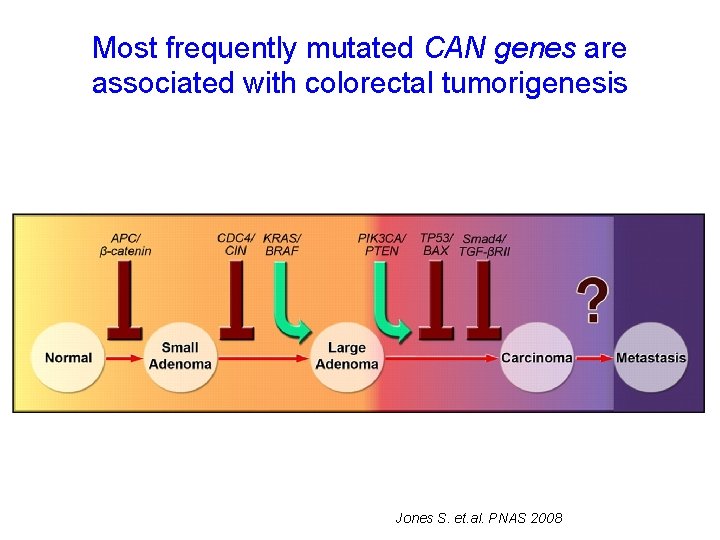
Most frequently mutated CAN genes are associated with colorectal tumorigenesis Jones S. et. al. PNAS 2008

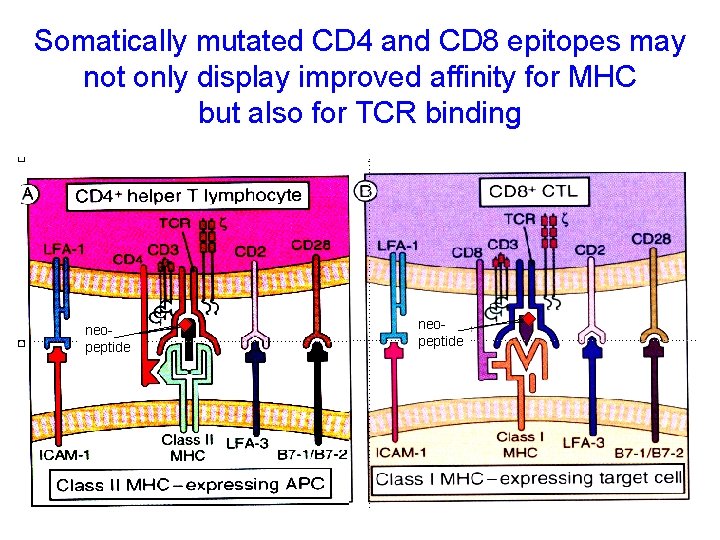
Somatically mutated CD 4 and CD 8 epitopes may not only display improved affinity for MHC but also for TCR binding neopeptide

Risultati e conclusione del primo anno di attività. Studi pre-clinici • Sono state identificate 9 sequenze di SVV e sintetizzati i peptidi sintetici corrispondenti. • I peptidi sono stati utilizzati in vitro per stimolare, in colture a breve termine, linfociti T CD 4+ isolati dal sangue di 7 soggetti normali e 7 pazienti con melanoma metastatico. • Linfociti CD 4 di 2/7 pazienti e 4/7 donatori hanno risposto. Nel caso dei pazienti con melanoma la reattività anti-SVV dei linfociti T CD 4+ appare quindi ridotta rispetto a quella dei soggetti sani.

Risultati del primo anno di attività. Studi pre-clinici • Unità di Biologia dei Tumori e Targeting Vascolare HSR (A. Corti); • Obbiettivo: Verificare se la combinazione di NGR-TNF con vaccinazione può indurre, in modelli animali, una più forte e duratura risposta immune e una migliore risposta clinica in confronto alla sola vaccinazione.
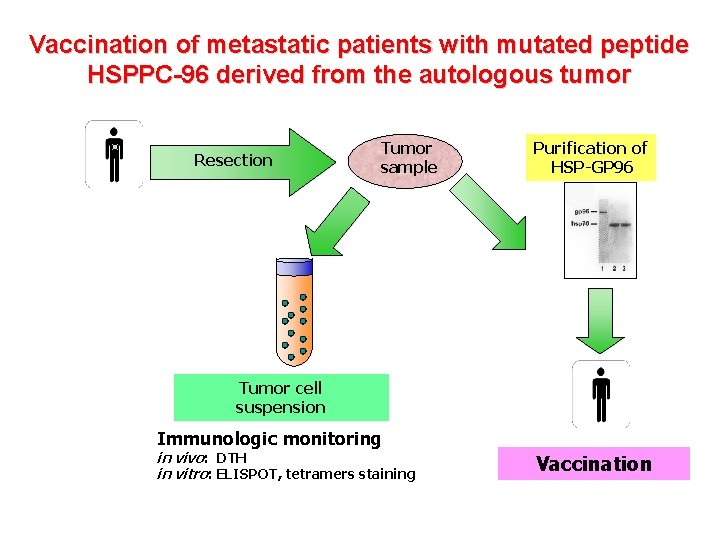
Vaccination of metastatic patients with mutated peptide HSPPC-96 derived from the autologous tumor Resection Tumor sample Purification of HSP-GP 96 Tumor cell suspension Immunologic monitoring in vivo: DTH in vitro: ELISPOT, tetramers staining Vaccination
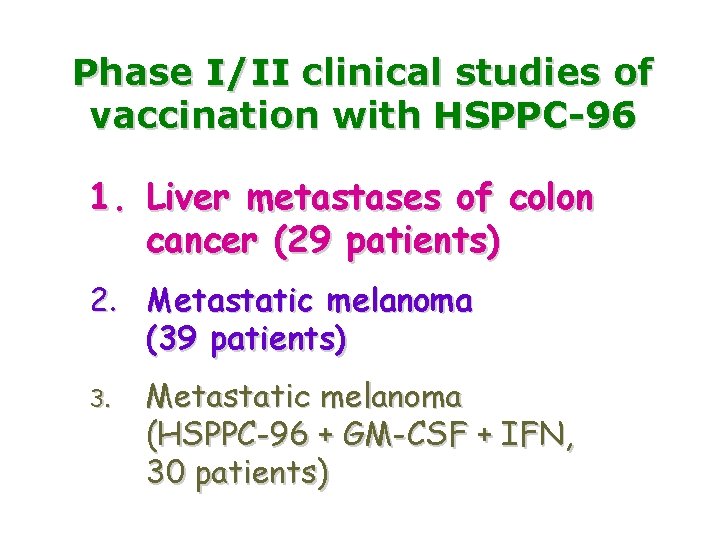
Phase I/II clinical studies of vaccination with HSPPC-96 1. Liver metastases of colon cancer (29 patients) 2. Metastatic melanoma (39 patients) 3. Metastatic melanoma (HSPPC-96 + GM-CSF + IFN, 30 patients)
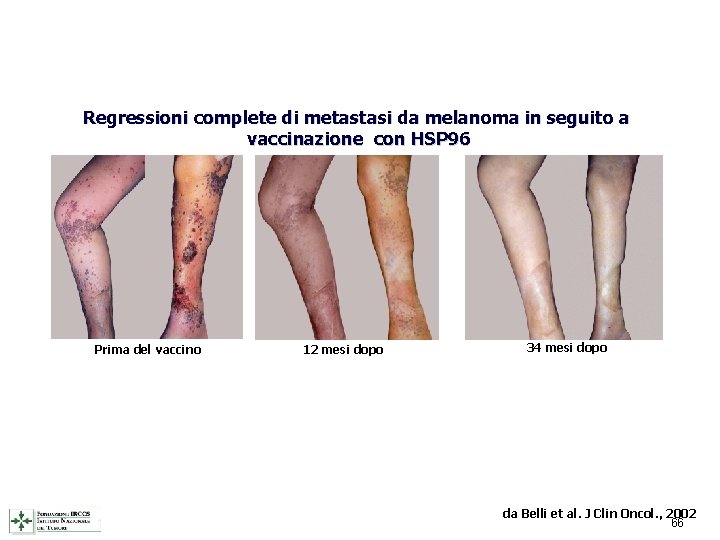
Regressioni complete di metastasi da melanoma in seguito a vaccinazione con HSP 96 Prima del vaccino 12 mesi dopo 34 mesi dopo da Belli et al. J Clin Oncol. , 2002 66
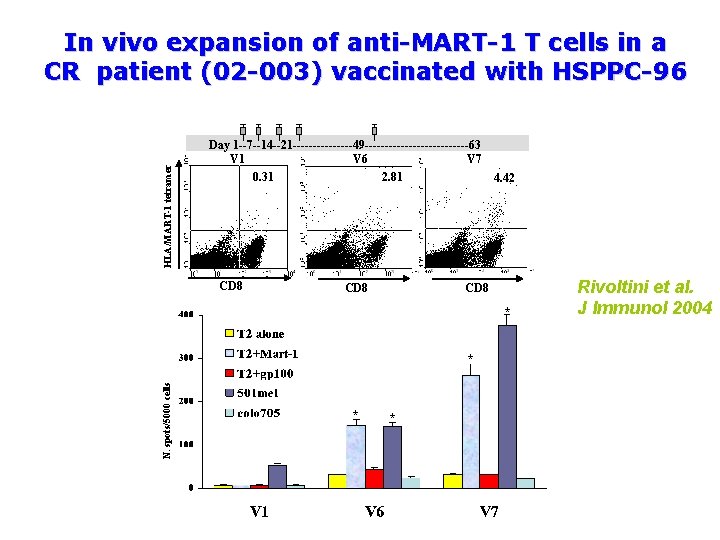
HLA/MART-1 tetramer In vivo expansion of anti-MART-1 T cells in a CR patient (02 -003) vaccinated with HSPPC-96 Day 1 --7 --14 --21 --------49 -------------63 V 1 V 6 V 7 0. 31 2. 81 CD 8 4. 42 CD 8 * N. spots/5000 cells * * * Rivoltini et al. J Immunol 2004
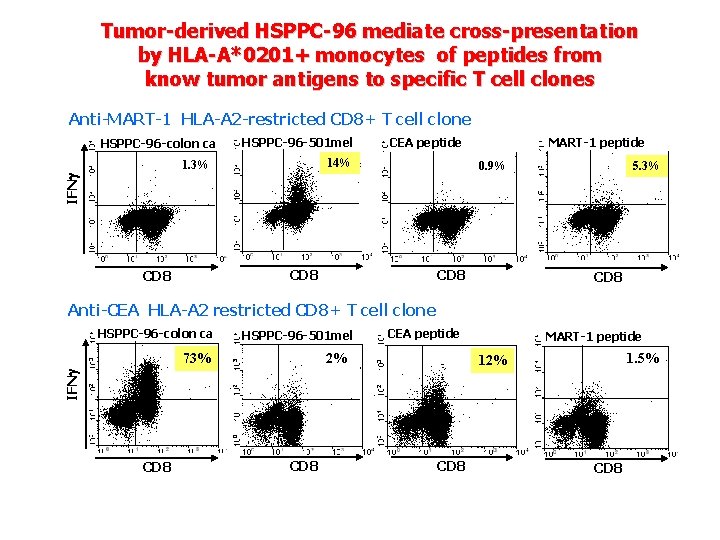
Tumor-derived HSPPC-96 mediate cross-presentation by HLA-A*0201+ monocytes of peptides from know tumor antigens to specific T cell clones Anti-MART-1 HLA-A 2 -restricted CD 8+ T cell clone HSPPC-96 -colon ca HSPPC-96 -501 mel CEA peptide 14% 0. 9% 5. 3% IFN 1. 3% MART-1 peptide CD 8 Anti-CEA HLA-A 2 restricted CD 8+ T cell clone HSPPC-96 -colon ca HSPPC-96 -501 mel 2% MART-1 peptide 1. 5% 12% IFN 73% CEA peptide CD 8

PHASE III RANDOMIZED TRIAL OF AUTOLOGOUS TUMOR-DERIVED HSPPC-96 vs. PHYSICIAN CHOICE IN METASTATIC MELANOMA (Testori A, Parmiani G et al. JCO 2008) • Trial features: Randomization 2: 1 favoring vaccination (215 vs. 107 patients). Physician choice included IL-2 and/or dacarbazine/temozolomide-based therapy and/or surgery • Results. Overall, patient in ITT vaccination arm fared similarly to those in the physician choice arm in terms of survival. . Subset of patients who received at least 10 doses of vaccine showed an extension in median survival of 29% compared with those receiving physician choice treatment. Oncophage® was associated with clinical benefit (P = 0. 017) in stage M 1 a and M 1 b patients who received at least 10 doses. • Conclusion: Signs of potential survival benefit in M 1 a/b patients.

Potential therapeutic targets for metastatic melanoma • Target • BRAF • RAS • • • VEGF CTLA 4 PI 3 K/AKT PTEN loss BCL 2 PTK • • • Agent Sorafenib Farnesyl transferase inhibitors Bevacizumab Anti-CTLA 4 (Ipilimumab) CCI-779 17 -AAG Antisense (Oblimersen) Gleevec Trial + + + + With the possible exception of Anti-CTLA$ none of the trials showed a statistically significant clinical benefit.
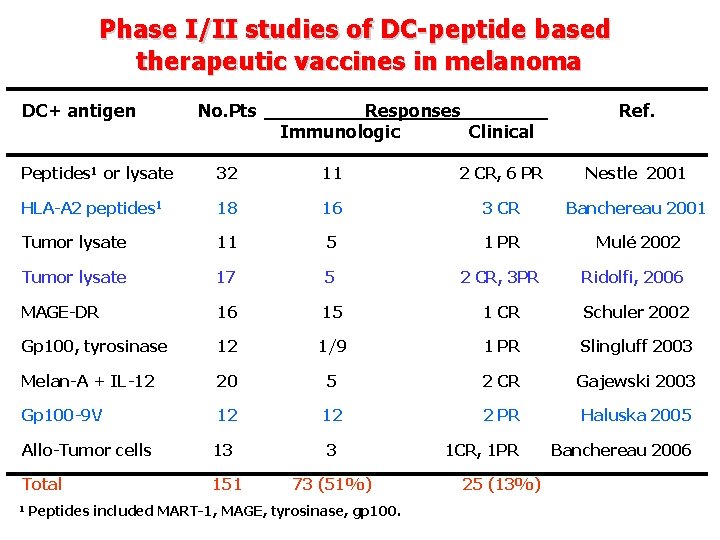
Phase I/II studies of DC-peptide based therapeutic vaccines in melanoma DC+ antigen No. Pts Responses Immunologic Clinical Ref. Peptides 1 or lysate 32 11 2 CR, 6 PR Nestle 2001 HLA-A 2 peptides 1 18 16 3 CR Banchereau 2001 Tumor lysate 11 5 1 PR Mulé 2002 Tumor lysate 17 5 2 CR, 3 PR Ridolfi, 2006 MAGE-DR 16 15 1 CR Schuler 2002 Gp 100, tyrosinase 12 1/9 1 PR Slingluff 2003 Melan-A + IL-12 20 5 2 CR Gajewski 2003 Gp 100 -9 V 12 12 2 PR Haluska 2005 Allo-Tumor cells 13 3 1 CR, 1 PR Banchereau 2006 Total 151 73 (51%) 1 Peptides included MART-1, MAGE, tyrosinase, gp 100. 25 (13%)

DC-peptide based cancer vaccines • Ex-vivo DCs+ tumor cell lysate/apoptotic cells • Ex-vivo DCs + purified MAA (proteins, peptides) • DC+m. RNA • DCs Gene-modifed to express MAA DCs delivered ex-vivo (Ab-mediated) or in vitro

Phase III prospective trial with Sepuleucel-T in prostate cancer • Vaccine: Autologous DC pulsed with PAP/GM-CSF hybrid protein. • Statistically significant benefit in survival at 3 years of observation.

Publications • Sacchi et al. Phase I Study of NGR-h. TNF, a Selective Vascular Targeting Agent, in Combination with Cisplatin in Refractory Solid Tumors. Clin Cancer Res 2011; 17: 1964; Calcinotto A, Grioni M, Jachetti E, Curnis F, Mondino A, Parmiani G, Corti A, Bellone M. Targeting tumor necrosis factor–α to neoangiogenesis vessels enhances lymphocyte infiltration in tumors and increases therapeutic potential of immunotherapy. J Immunol, 2012; 188: www. jimmunol. org.



CSCs FBS CSCs

Conclusions A Low immunogenic profile was found in both CSCs and FBS tumor cells isolated from GBM patients, with higher defective APC pattern in CSCs; the immune profile can be rescued, though more efficiently in FBS tumor cells, by treatment of GBM cell lines with IFNs or 5 -Aza-Cd. R ; T cell-mediated immune responses can be obtained from GBM patients, though mostly in the TH 2 -mediated subset; Differential gene signature, including immune related genes, was detected in CSCs vs FBS tumor cells.

Reclutamento pazienti 1. Disponibilità dei pazienti in rapporto a trattamenti competitivi (es. GSK 1021202 e Vemurafenib nel melanoma); 2. Complessità del trattamento (interdisciplinarietà degli operatori, ecc. ) e costi.

Inclusion criteria • Histologically confirmed AJCC (modified) stage IV melanoma; • ECOG performance status 0 -1; • Life expectancy of at least 6 months; • Hematopoietic, liver and renal normal functions • No brain mets • Signed written informed consent. •

Obiettivi e Razionale • Il progetto è basato sull’attivazione di studi clinici di combinazione tra farmaci biologici e vaccinazione con peptidi antigenici, oltre a studi associati o paralleli di laboratorio destinati: 1) a migliorare le conoscenze sui meccanismi alla base della combinazione proposta; 2) al monitoraggio immuno/biologico dei pazienti; c) a valutare polimorfismi di geni che possono influire sulla risposta al trattamento.

Immune response as assessed by 4 different assays • Tabella (Silvia)

Criticità per il trasferimento clinico (cont. ) • Scarsa partecipazione industriale dovuta a difficoltà di standardizzazione della combinazione di farmaci e brevettabilità (fornitura gratuita del farmaco NGRh. TNF da parte di Mol. Med, Milano).

Somatically mutated tumor antigens: Best candidates with limitations 1. Somatically mutated TAAs are in principle the best antigens to use for cancer immunotherapy; 2. Surprisingly, still a limited number of such antigens have been identified. Cumbersome, lengthy and poorly processing approaches for the identification of mutated TAAs (shared or unique) have prevented as yet their massive characterization 3. As a consequence, the T cell responses specific for mutated TAAs remains largely undefined

Somatic mutations in CAN genes fulfil two critical requirements for cancer immunotherapy 1. They involve driver mutations: – causally implicated in cancer development. – confer growth advantage. – positively selected in the microenvironment of the tissue in which the cancer arise This is relevant to avoid immune escape 2. They (at least some) seem to be expressed by cancer stem/initiating cells

Probability to generate novel unique class I epitopes from somatically mutated CAN genes in CRC - Individual CRC accumulate, on average, 10 novel and unique HLAA*0201 epitopes, including genes implicated in the neoplastic process.
- Slides: 86