Corrosion by Hazardous Chemicals Otto Drescher P E

Corrosion by Hazardous Chemicals Otto Drescher, P. E.

Summary • Discuss Uses & Some Properties • Case History • Materials Selection Guidelines

Specific Chemicals • Sulfuric Acid • • Nitric Acid Phosphoric Acid Chlorine HCl • • HF Organic Acids Caustic Ammonia & Amines

Sulfuric Acid • Largest Tonnage of Inorganic Chemicals • Major Use in Petroleum Refining, Fertilizers, Paints, Pigments, Dyes, Explosives, and p. H Control • Powerful Oxidizing Agent

Sulfuric Acid • Conc. Acid is 70% to 100% • Oleum (Fuming) – 100% + excess SO 3 • Dilute Acid - < 25% • Contaminants Greatly Effect Corrosion

Erosion-corrosion in 93% H 2 SO 4 Carbon Steel Nozzle 4 - 6 fps (1. 2 – 1. 8 m/s) at RT Design Velocity Limit is <2 fps (0. 6 m/s)

Sulfuric Acid – Guidelines Materials Selection °F °C Tantalum Glass Si-Iron Boiling Point Curve Alloy B-2, B-3, B-4 316 825 20 Cb-3 904 L 316 Steel Concentration, Wt. %

Specific Chemicals • Sulfuric Acid • Nitric Acid • • • Phosphoric Acid Chlorine HCl • • HF Organic Acids Caustic Ammonia & Amines

Nitric Acid • Major Use in Ammonium Nitrate and Phosphate Fertilizers, Nitro Explosives, Plastics, and Dyes • Always an Oxidizing Acid

Nitric Acid • Concentrated Acid is 67% to 95% • Fuming Grades are > 99% • HF Addition Makes It Reducing - • Cl Contaminantion Releases Nascent Cl 2

Nitric-HF Acid Pickling Line Alloy 33 (33 Cr-32 Fe-31 Ni-1. 6 Mo) Tank

Nitric Acid – Guidelines Materials Selection °C °F Boiling Point Curve Tantalum Si-Iron 1815 LC Si 304 L 310 LCN Titanium 67% Aluminum Concentration, Wt. %

Specific Chemicals • Sulfuric Acid • Nitric Acid • Phosphoric Acid • Chlorine • HCl • • HF Organic Acids Caustic Ammonia & Amines

Phosphoric Acid • Major Use in Phosphate Fertilizers, Food & Beverages, Drying Agents, Plasticizers, Oil Additives, Fire Retardants, and Insecticides • Is a Reducing Acid, More So When Contaminated With Cl and F

Phosphoric Acid • Two Types of Pure Acid (85%): o C. P. Grade o Food Grade • Superphosphoric Acid - > 105% • Industrial Grades May Be Contaminated With Cl-, F-, and Fe+++

Preferential Attack in H 3 PO 4 Ambient Temperature, Steam Traced 304 Compare Thickness 316

Phosphoric Acid – Guidelines Materials Selection °C C-276 G-30 G-35 Boiling Point Curve 317 L 316 L Concentration, Wt. % °F Alloy 28, 31 825/904 L

Specific Chemicals • • • Sulfuric Acid Nitric Acid Phosphoric Acid • Chlorine • HCl • • HF Organic Acids Caustic Ammonia & Amines

Chlorine Uses - Inorganic • • • Chlorides Salts Bleaching Chlorination Dyes Rockets Explosives Uses - Organic • • • Drugs Plastics Solvents Insecticides PVC Deodorants

Chlorine • Poisonous, greenish-yellow gas • Does Not Attack Metals < 400 °F (204 °C), Except Ti • Moisture aggravates corrosivity • HOCl is a Powerful Oxidizing Acid

Chlorine – Guidelines Materials Selection • Liquid Cl 2 – Carbon Steel ( Dry! ) • Cl 2 Gas o Carbon Steel to 250 °F (121 °C) Wall Temp o Caution! Carbon Steel May Ignite o Alloy 400 to 570 °F (299 °C) Wall Temp

Chlorine – Guidelines Materials Selection • Chlorinated Organics o Carbon Steel to 200 °F (93 °C) Wall Temp o Alloys 400 & 200 to B. P. • Chlorinated Waters – Titanium, C-276

Specific Chemicals • • Sulfuric Acid Nitric Acid Phosphoric Acid Chlorine • HCl • • HF Organic Acids Caustic Ammonia & Amines

Hydrochloric Acid • 2 nd in Importance to Sulfuric Acid • Major Use in Pickling Steels, Oil Well p. H, Chemical Cleaning, Ore Reduction, Food Processing, Synthetic Rubber, and Regeneration of Ion Exchange Resins

Hydrochloric Acid • Anhydrous is Non-corrosive, Very Hygroscopic • Strong, Reducing Acid • Attacks Most Metals Below H 2 in EMF Series • Forms Metal Chlorides and Liberates H 2

Hydrochloric Acid • Attacks Noble Metals Only If DO or Oxidizing +++ Contaminants Are Present (Fe ) • Water Solution is 28% – 35% • Commercial Grade is Muriatic Acid • Constant Boiling Mixture is 20. 2%

HCl Pitting Corrosion Alloy 825 Pickling Hook in Heated Acid +++ Contaminated With Fe

Hydrochloric Acid – Guidelines Materials Selection °C °F Boiling Point Curve B-2, B-3 Zirconium Tantalum 400 Air Free C-Family 200, 400 825 Concentration, Wt. %

Specific Chemicals • • • Sulfuric Acid Nitric Acid Phosphoric Acid Chlorine HCl • HF • • • Organic Acids Caustic Ammonia & Amines

Hydrofluoric Acid • Major Uses are Refrigerants, Fluorocarbons, Etch/Polish Glass, Pickling, Ore Processing Acidification of Oil Wells • Is a Weak, Reducing Acid • Anhydrous HF and HF Acid are Very Hazardous • Commercial Grades are 48% & 70% • Fluoride Ions Less Aggressive Than Chloride

SCC of Alloy 400 in HF Bubble Caps Exposed to Aerated Vapors

HF Acid – Guidelines Materials Selection °C Boiling Point Curve 400 200, 400, C-276, C-22, 686 Concentration, Wt. % °F

Specific Chemicals • • • Sulfuric Acid Nitric Acid Phosphoric Acid Chlorine HCl • HF • Organic Acids • Caustic • Ammonia & Amines

Organic Acids • Major Uses are Dyeing, Pigments, Pesticides, Tanning, Esters, Alcohols, Organic Chemicals • Are Weak, Reducing Acids • Less Corrosive as Carbon Chain Lengthens (or as Molecular Weight Increases)

Organic Acids • Rank in Corrosivity: Formic > Acetic > Propionic > Butyric • Aeration & Temperature Increase Corrosion - • Contaminants (e. g. Cl ) Increase Corrosion
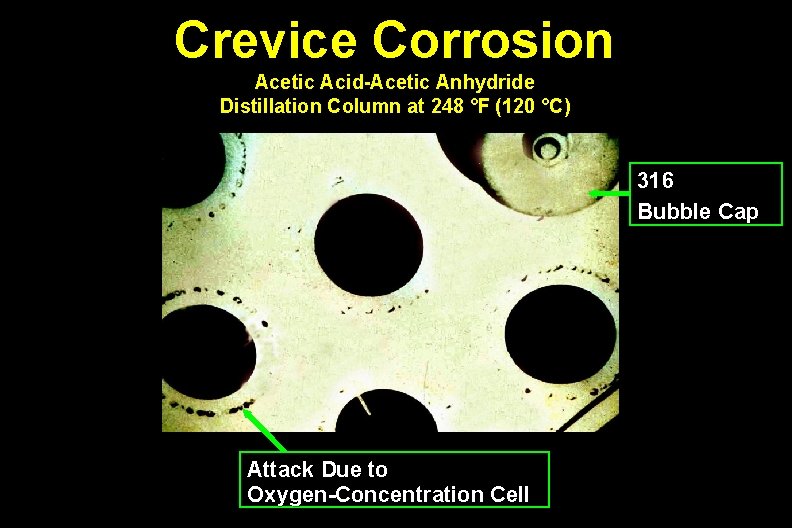
Crevice Corrosion Acetic Acid-Acetic Anhydride Distillation Column at 248 °F (120 °C) 316 Bubble Cap Attack Due to Oxygen-Concentration Cell

Selective Corrosion Boiling Acetic Acid Weld Metal Corroded More Than Base Metal Corroded More Than Weld Metal

Organic Acids – Guidelines Materials Selection • IGA Probable, if Low C Not Used • SCC Possible, if Chlorides Present • 304 L For Storage and Low Temperatures • 316 L For Higher Temperatures - • 6% Mo Alloys if Crevices & Cl Present • May Need 6% or Higher Mo Alloy Welds

Specific Chemicals • • • Sulfuric Acid Nitric Acid Phosphoric Acid Chlorine HCl • HF • Organic Acids • Caustic • Ammonia & Amines

Sodium Hydroxide Caustic Soda • Major Uses are Sodium Compounds, Metallurgical (Aluminum), Glass, Rayon Fibers, Cellophane, Paper Industry, Detergents, Bleaching, Whitening Products, Soaps, Cleaning (Dissolves Organics), p. H Control • Strong Alkali • KOH Is Very Similar to Na. OH

Caustic Cracking of Steel Heating Coil in 50% Na. OH General Corrosion Stress Corrosion Cracking
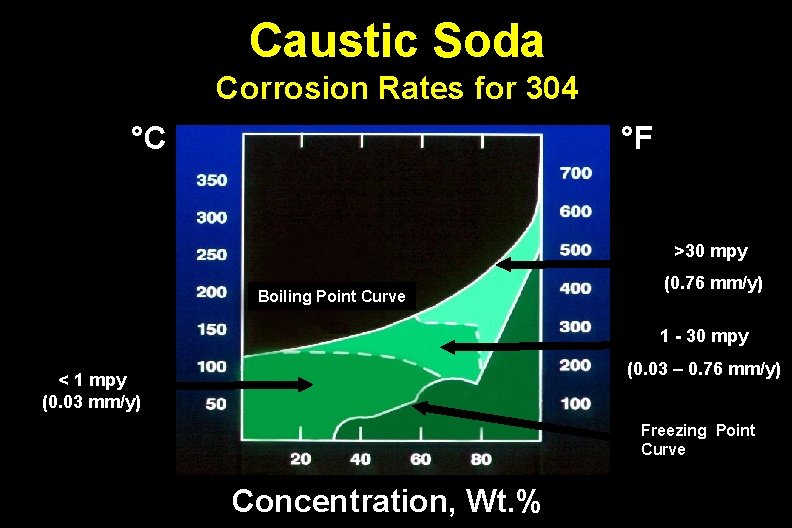
Caustic Soda Corrosion Rates for 304 °C °F >30 mpy Boiling Point Curve (0. 76 mm/y) 1 - 30 mpy (0. 03 – 0. 76 mm/y) < 1 mpy (0. 03 mm/y) Freezing Point Curve Concentration, Wt. %
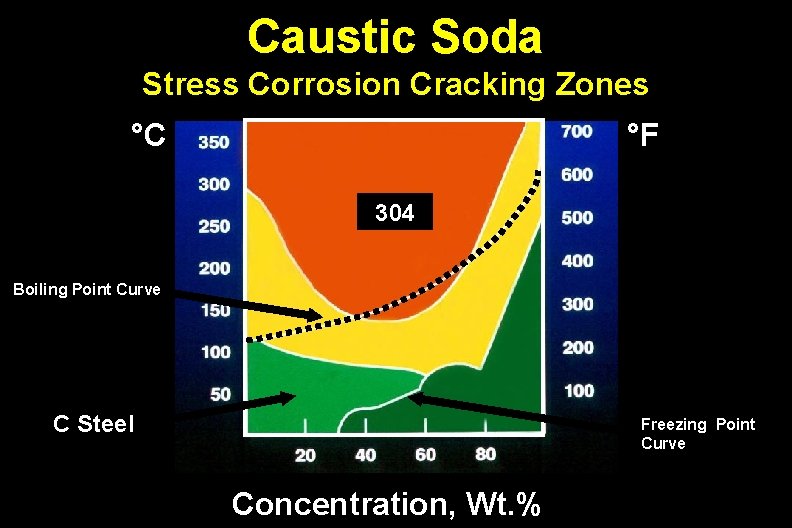
Caustic Soda Stress Corrosion Cracking Zones °C °F 304 Boiling Point Curve C Steel Freezing Point Curve Concentration, Wt. %
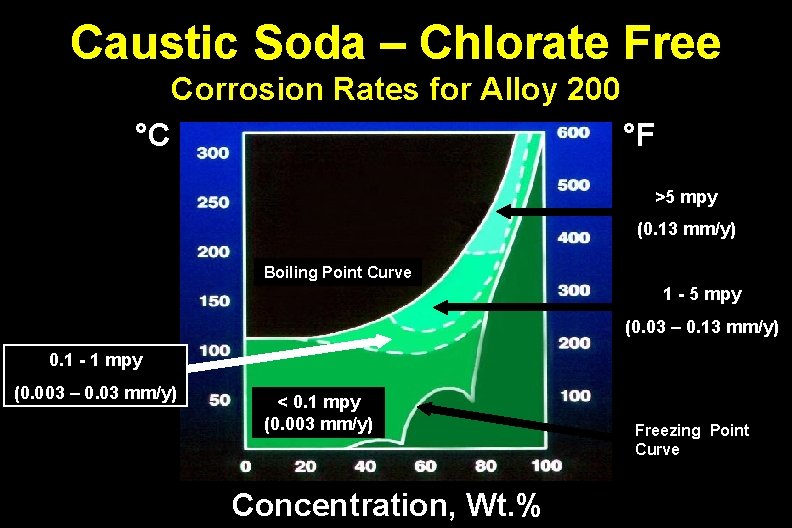
Caustic Soda – Chlorate Free Corrosion Rates for Alloy 200 °C °F >5 mpy (0. 13 mm/y) Boiling Point Curve 1 - 5 mpy (0. 03 – 0. 13 mm/y) 0. 1 - 1 mpy (0. 003 – 0. 03 mm/y) < 0. 1 mpy (0. 003 mm/y) Concentration, Wt. % Freezing Point Curve

Specific Chemicals • • • Sulfuric Acid Nitric Acid Phosphoric Acid Chlorine HCl • • • HF Organic Acids Caustic • Ammonia & Amines

Ammonia • Major Uses are Organic Compounds, Drugs, Plastics, Dyes, Fertilizers, Explosives, Production of Nitric Acid, Ammonium Salts, Refrigerant, Cleaning Agents • 4 th Most Important Chemical

Ammonia – Guidelines Materials Selection • Anhydrous Ammonia Is Non-corrosive Except for SCC of Steel • NH 3 + H 2 O NH 4 OH Water Dilution Forms Ammonium Hydroxide - Inhibits SCC of Steel • Ingress of Water and O 2 Corrosive to Cu and Cr-free Ni Alloys (Even ppm)

Ammonia – Guidelines Materials Selection • Corrosion of Steels Through Stainless Steels May Be Caused By CO 2 Contamination (Ammonium Carbamate) • Chloride Contamination Can Reduce Life of Stainless Steel Equipment

Summary • Discussed Specific Chemicals • Reviewed Use & Some Properties • Presented Materials Selection Guidelines for Each Chemical

The End Questions?
- Slides: 50