Coral Reefs in Acid Lab Directions Follow the

Coral Reefs in Acid Lab Directions: Follow the steps in each box. Use the pictures to help answer the questions. Answer the questions using both pictures & written response. Write your answers on a separate piece of paper. Modified from: In. STEP ‘Dude –where’d the reef go? ’ 1

1. What are Coral Reefs? • Where are they located? Zone? • What are they made of? • Who uses them? • Why are they important? 2

1. What are Coral Reefs? • • Where are they located? Zone? Shallow sunlight in saltwater, Photic zone What are they made of? Calcium carbonate Who uses them? Fish, sponges, sea stars, turtles, crabs, Why are they important? Provide food & habitat to other marine organisms 3

2. Coral Cover Observation • What has happened to Coral Cover % from 1977 to 2002? • Why do you think this happened? • How does this relate to question 1? • Graph of Coral Cover • See Figure 6. at http: //www. climateshifts. org/? p=6348 4

2. Coral Cover Observation • What has happened to Coral Cover % from 1977 to 2002? • Declined • Why do you think this happened? • Answers will vary • How does this relate to question 1? • Answers will vary; If lots of marine organisms use them, but they are disappearing, you will see a decrease in biodiveristy 5

3. Demo • Add 5 to 8 drops of vinegar onto a shell or coral. Write your observations. You may need to use a microscope or hand-lens. • How does this relate to question 2? • Why is this happening? What is vinegar? What is the shell made of? (hint- think p. H) 6

3. Demo • Add 5 to 8 drops of vinegar onto a shell or coral. Write your observations. You may need to use a microscope or hand-lens. • How does this relate to question 2? – It bubbles and fizzes • Why is this happening? What is vinegar? What is the shell made of? (hint- think p. H) – Vinegar is an acid. – Shell is calcium carbonate – The vinegar is breaking down the shell. – This helps explain why the coral reef is disappearing. Something acid in the water is breaking down the coral reef, which is also made from calcium carbonate 7

4. Demo • Take a small cup of salt water. Add p. H indicator into it. What is the p. H? • Then, using a straw, blow bubbles into the cup for 15 to 20 seconds. What do you notice about the p. H of Salt water after blowing “bubbles” into it? • What are the “bubbles” made out of? 8

4. Demo • Take a small cup of salt water. Add p. H indicator into it. What is the p. H? • p. H = about 8. 2 • Then, using a straw, blow bubbles into the cup for 15 to 20 seconds. What do you notice about the p. H of Salt water after blowing “bubbles” into it? • The color changes resulting in a p. H change. The p. H has decreased • What are the “bubbles” made out of? • Carbon dioxide 9

5. CO 2 + Water= ? • See Carbon dioxide entering the water picture at • http: //askascientist. co. uk/chemistry/oceanacidity-what-is-phanyway/ • What is happening to CO 2 in this picture? • How does this relate to question 4? 10

5. CO 2 + Water= ? • What is happening to CO 2 in this picture? – Carbon dioxide is being absorbed in the ocean. It is mixing with the water molecule to form a new compound H 2 CO 3 (called ‘Carbonic Acid’) • How does this relate to question 4? – Blowing bubbles is adding Carbon to water and the result was a decrease in p. H – Having Carbonic Acid form must be a reason has to why the p. H decreased. 11

6. CO 2 concentration • See Mauna Loa Carbon dioxide data at • http: //celebrating 200 ye ars. noaa. gov/datasets/ mauna/ • What is the trend? • What is causing this trend? • Where is some of the extra CO 2 going? • How does this relate to question 5? 12

6. CO 2 concentration • What is the trend? • Carbon dioxide concentration is increasing over the last 50 years • What is causing this trend? • Answers will vary; increased human activity such as cars, airplanes, burning of fossil fuels, etc. • Where is some of the extra CO 2 going? • Atmosphere and then into the ocean • How does this relate to question 5? • Because there is more carbon dioxide in the atmosphere, more is entering the ocean, and therefore, it is mixing with water to become carbonic acid. More carbonic acid means the p. H of the ocean is decreasing. 13
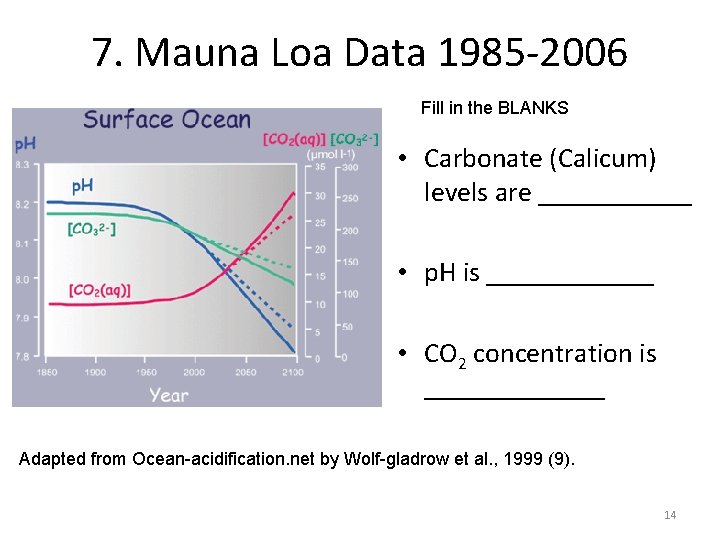
7. Mauna Loa Data 1985 -2006 Fill in the BLANKS • Carbonate (Calicum) levels are ______ • p. H is ______ • CO 2 concentration is _______ Adapted from Ocean-acidification. net by Wolf-gladrow et al. , 1999 (9). 14

7. Mauna Loa Data 1985 -2006 • Carbonate (Calicum) levels are DECREASING • p. H is DECREASING • CO 2 concentration is INCREASING 15

8. CONCLUSION • Because Carbon Dioxide levels are…. • This is causing the ocean to become…. • This is affecting coral reefs because… 16

8. CONCLUSION • Because Carbon Dioxide levels are…. INCREASING • This is causing the ocean to become…. MORE ACIDIC • This is affecting coral reefs because… THE ACIDIC ENVIRONMENT EATS AWAY AT THE CALICUM CARBONATE STRUCTURE 17

9. What else depends on carbonate? List 5 18

9. What else depends on carbonate? • • Mollusk- shelled gastropods, Arthropods- exoskeletons Algae- red algae Echinoderms- calcium bodies • Anything that eats or lives among those organisms. 19

10. Acidification Projections What if the CO 2 amount trend continues, as is, into our future? from Hoegh-Guldberg et al (2007) 20

10. Acidification Projections • What if the CO 2 amount trend continues, as is, into our future? • It is projected that coral reefs will completely die off if Carbon Dioxide levels reach 500 ppm. 21

11. What can we (you) do? List 5 22

11. What can we (you) do? • • • Reduce your carbon foot print Ride a bike instead of drive Carpool Alternative Energy: wind or solar Replanting coral programs 23
- Slides: 23