Copyright 2007 Pearson Benjamin Cummings All rights reserved

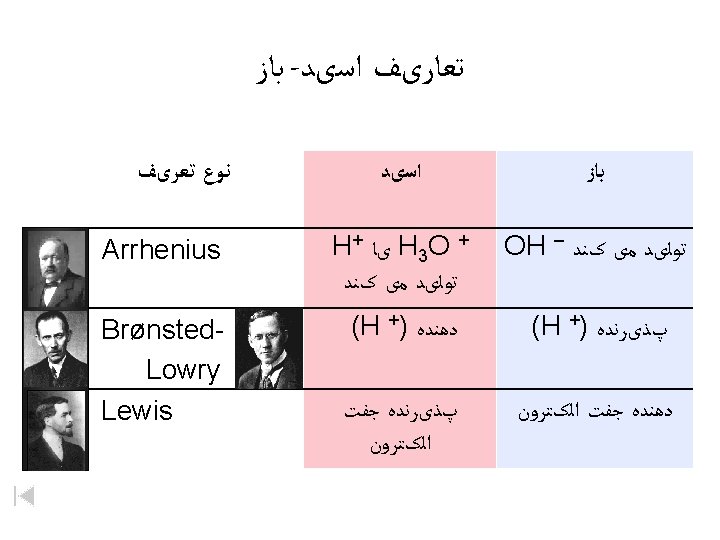
ﺍﺳیﺪ ﻭ ﺑﺎﺯ Copyright © 2007 Pearson Benjamin Cummings. All rights reserved.
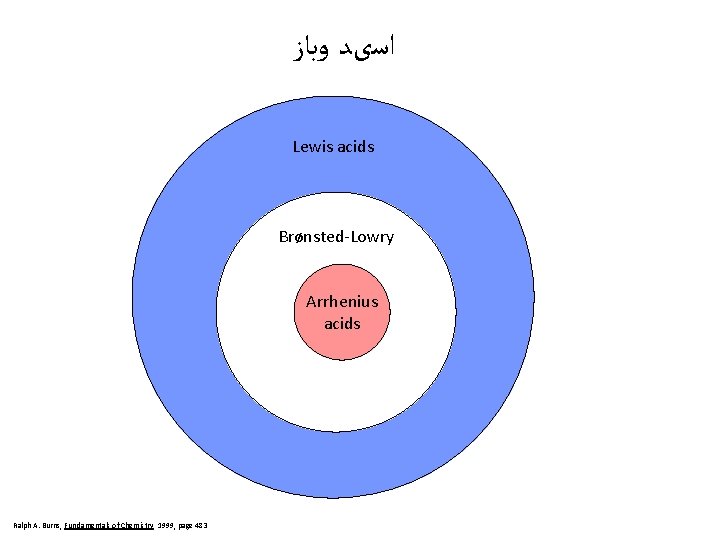
ﺍﺳیﺪ ﻭﺑﺎﺯ Lewis acids Brønsted-Lowry Arrhenius acids Ralph A. Burns, Fundamentals of Chemistry 1999, page 483
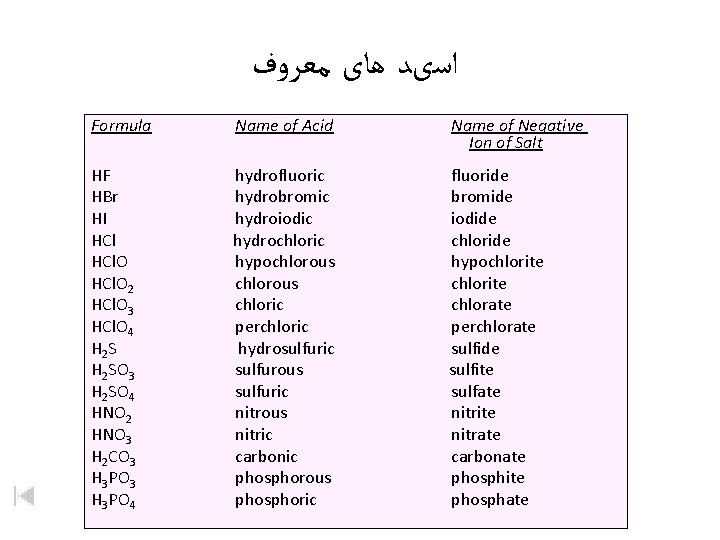
ﺍﺳیﺪ ﻫﺎی ﻣﻌﺮﻭﻑ Formula Name of Acid Name of Negative Ion of Salt HF HBr HI HCl. O 2 HCl. O 3 HCl. O 4 H 2 SO 3 H 2 SO 4 HNO 2 HNO 3 H 2 CO 3 H 3 PO 4 hydrofluoric hydrobromic hydroiodic hydrochloric hypochlorous chloric perchloric hydrosulfuric sulfurous sulfuric nitrous nitric carbonic phosphorous phosphoric fluoride bromide iodide chloride hypochlorite chlorate perchlorate sulfide sulfite sulfate nitrite nitrate carbonate phosphite phosphate
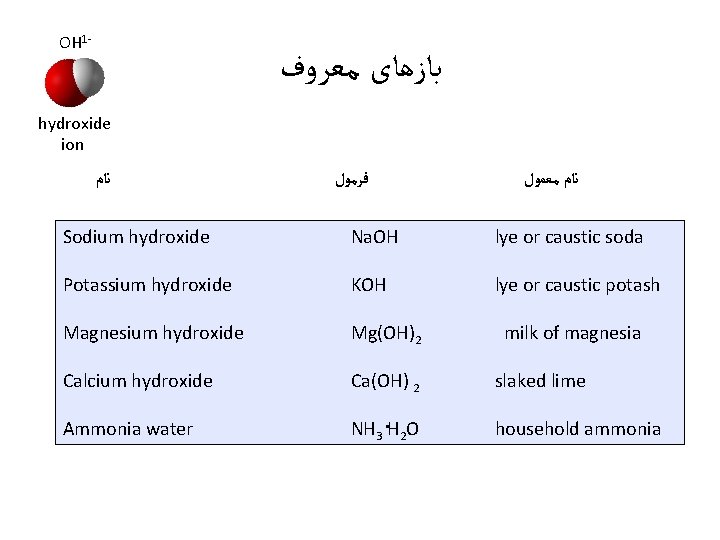
OH 1 - ﺑﺎﺯﻫﺎی ﻣﻌﺮﻭﻑ hydroxide ion ﻧﺎﻡ ﻓﺮﻣﻮﻝ ﻧﺎﻡ ﻣﻌﻤﻮﻝ Sodium hydroxide Na. OH lye or caustic soda Potassium hydroxide KOH lye or caustic potash Magnesium hydroxide Mg(OH)2 Calcium hydroxide Ca(OH) 2 slaked lime Ammonia water NH 3. H 2 O household ammonia milk of magnesia
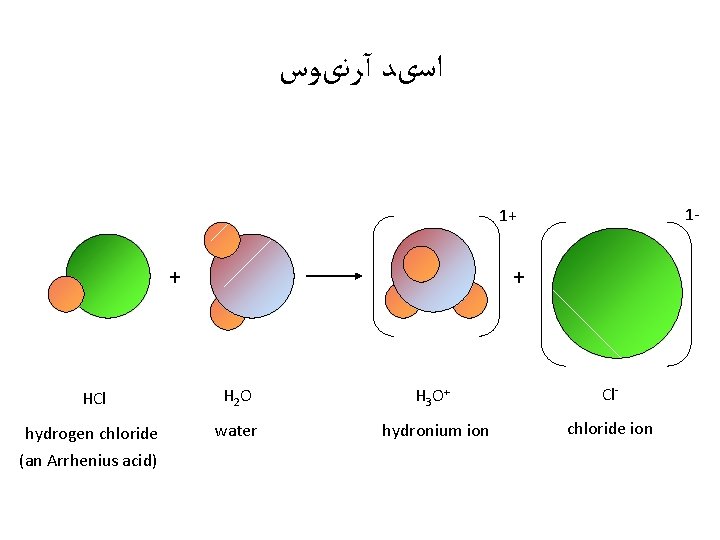
ﺍﺳیﺪ آﺮﻧیﻮﺱ 1 - 1+ + + HCl H 2 O H 3 O+ Cl- hydrogen chloride (an Arrhenius acid) water hydronium ion chloride ion
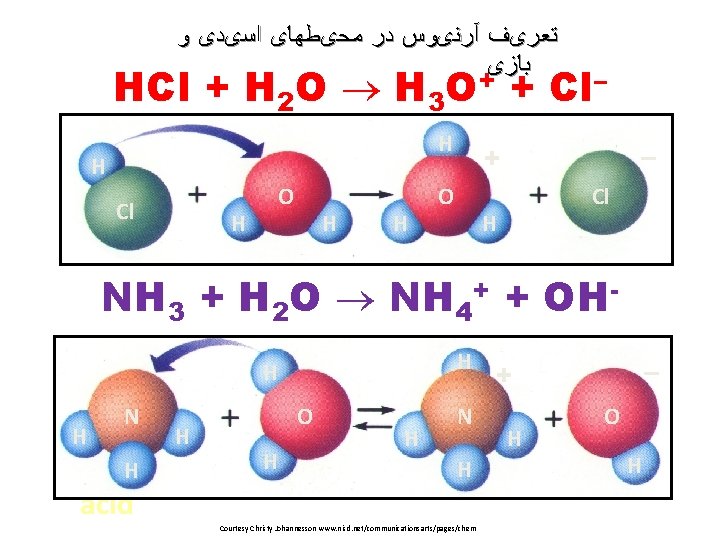
ﺗﻌﺮیﻒ آﺮﻧیﻮﺱ ﺩﺭ ﻣﺤیﻄﻬﺎی ﺍﺳیﺪی ﻭ ﺑﺎﺯی HCl + H 2 O H 3 O+ + Cl– H H Cl H O H H – + O Cl H NH 3 + H 2 O NH 4+ + OHH H H N H acid H O H H N H Courtesy Christy Johannesson www. nisd. net/communicationsarts/pages/chem – + H O H

ﺍﺳیﺪ ﺿﻌیﻒ ﻭ ﻗﻮی Zumdahl, De. Coste, World of Chemistry 2002, page 508


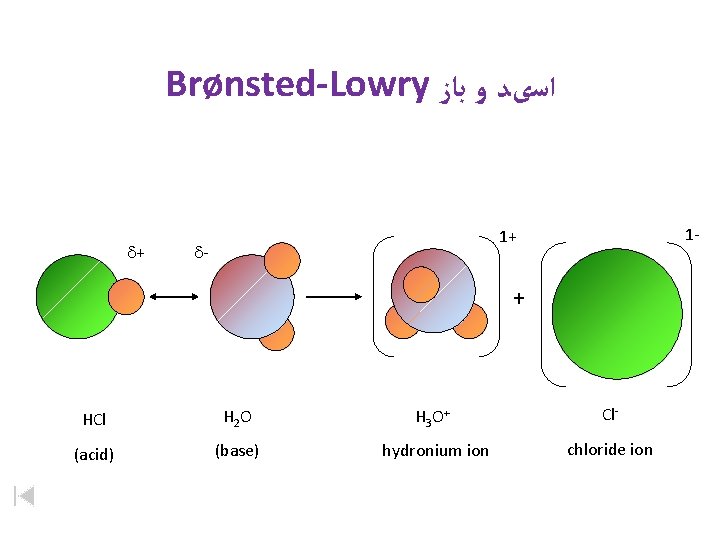
Brønsted-Lowry ﺍﺳیﺪ ﻭ ﺑﺎﺯ d+ 1 - 1+ d- + HCl H 2 O H 3 O+ Cl- (acid) (base) hydronium ion chloride ion
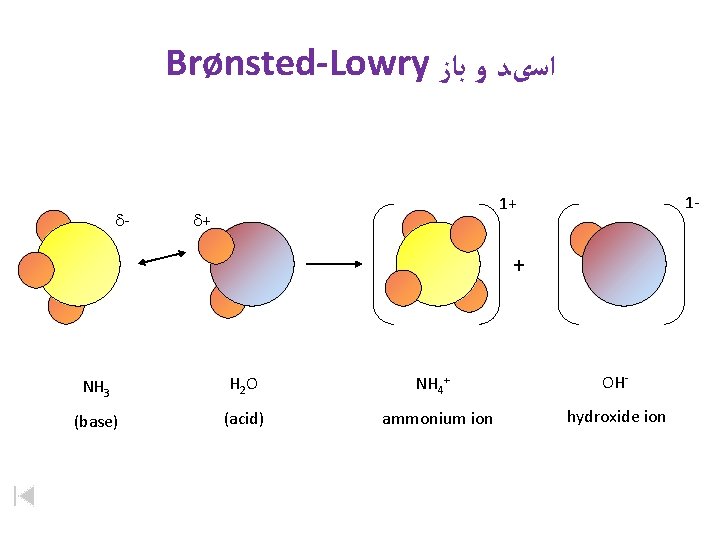
Brønsted-Lowry ﺍﺳیﺪ ﻭ ﺑﺎﺯ d- 1 - 1+ d+ + NH 3 H 2 O (base) (acid) NH 4+ ammonium ion OHhydroxide ion
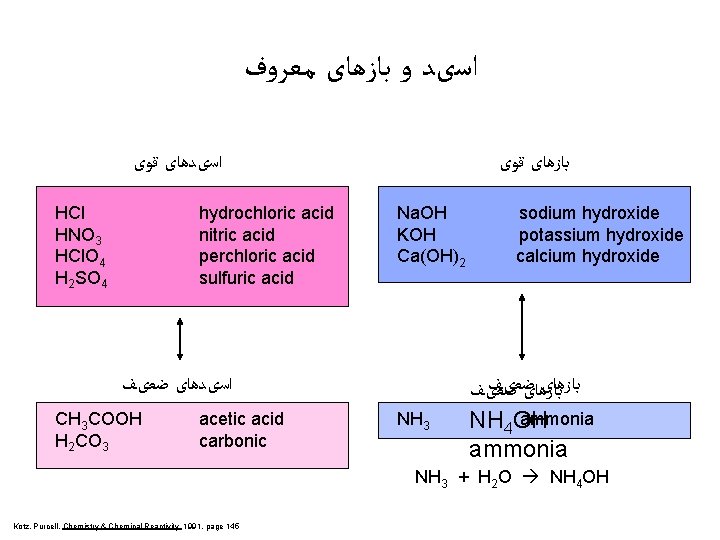
ﺍﺳیﺪ ﻭ ﺑﺎﺯﻫﺎی ﻣﻌﺮﻭﻑ ﺍﺳیﺪﻫﺎی ﻗﻮی HCl HNO 3 HCl. O 4 H 2 SO 4 hydrochloric acid nitric acid perchloric acid sulfuric acid ﺑﺎﺯﻫﺎی ﻗﻮی Na. OH KOH Ca(OH)2 ﺍﺳیﺪﻫﺎی ﺿﻌیﻒ CH 3 COOH H 2 CO 3 acetic acid carbonic NH 3 sodium hydroxide potassium hydroxide calcium hydroxide ﺑﺎﺯﻫﺎیﺿﻌیﻒ ﺑﺎﺯﻫﺎی ﺿﻌیﻒ ammonia NH 4 OH ammonia NH 3 + H 2 O NH 4 OH Kotz, Purcell, Chemistry & Chemical Reactivity 1991, page 145
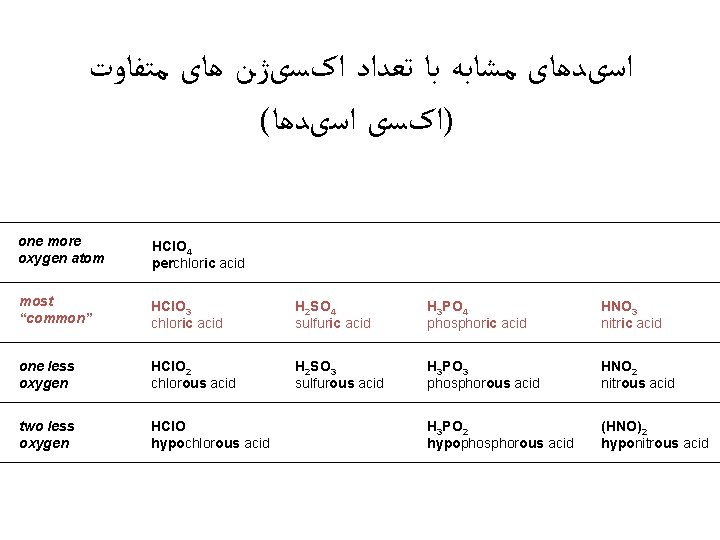
ﺍﺳیﺪﻫﺎی ﻣﺸﺎﺑﻪ ﺑﺎ ﺗﻌﺪﺍﺩ ﺍکﺴیژﻦ ﻫﺎی ﻣﺘﻔﺎﻭﺕ ( )ﺍکﺴی ﺍﺳیﺪﻫﺎ one more oxygen atom HCl. O 4 perchloric acid most “common” HCl. O 3 chloric acid H 2 SO 4 sulfuric acid H 3 PO 4 phosphoric acid HNO 3 nitric acid one less oxygen HCl. O 2 chlorous acid H 2 SO 3 sulfurous acid H 3 PO 3 phosphorous acid HNO 2 nitrous acid two less oxygen HCl. O hypochlorous acid H 3 PO 2 hypophosphorous acid (HNO)2 hyponitrous acid
![[H+] p. H 10 -14 14 1 M Na. OH 10 -13 13 p. [H+] p. H 10 -14 14 1 M Na. OH 10 -13 13 p.](http://slidetodoc.com/presentation_image_h/94ef6f4c8e68bccb876ab5d0e02c00a8/image-15.jpg)
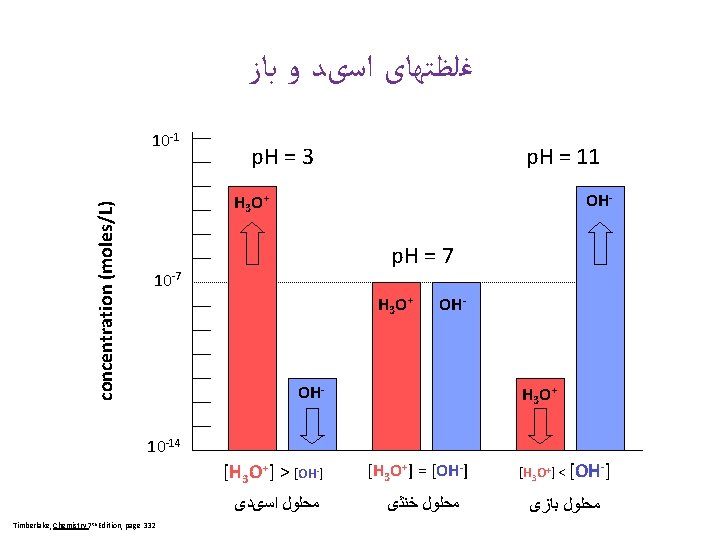
[H+] p. H 10 -14 14 1 M Na. OH 10 -13 13 p. H Scale 14 Acidic Neutral 0 Base Basic Acid 10 -12 12 7 Zumdahl, De. Coste, World of Chemistry 2002, page 515 10 -11 11 Ammonia (household cleaner) 10 -10 10 10 -9 9 10 -8 8 10 -7 7 10 -6 6 10 -5 5 10 -4 4 10 -3 3 10 -2 2 10 -1 1 100 0 Blood Pure water Milk Vinegar Lemon juice Stomach acid 1 M HCl

ﺑﺮﺧی ﻣﻮﺍﺩ p. H gastric juice 1. 6 vinegar 2. 8 carbonated beverage 3. 0 0 1 2 acidic Timberlake, Chemistry 7 th Edition, page 335 urine 6. 0 4 5 bile 8. 0 6 7 neutral [H+] = [OH-] 8 ammonia 11. 0 bleach 12. 0 seawater 8. 5 9 1. 0 M Na. OH (lye) 14. 0 milk of magnesia 10. 5 detergents 8. 0 - 9. 0 milk 6. 4 tomato 4. 2 coffee 5. 0 3 blood 7. 4 potato 5. 8 apple juice 3. 8 lemon juice 2. 2 drinking water 7. 2 bread 5. 5 orange 3. 5 1. 0 M HCl 0 water (pure) 7. 0 soil 5. 5 10 11 basic 12 13 14
![p. H = -log [H 1+] Kelter, Carr, Scott, Chemistry A World of Choices p. H = -log [H 1+] Kelter, Carr, Scott, Chemistry A World of Choices](http://slidetodoc.com/presentation_image_h/94ef6f4c8e68bccb876ab5d0e02c00a8/image-19.jpg)
p. H = -log [H 1+] Kelter, Carr, Scott, Chemistry A World of Choices 1999, page 285
![p. H = -log [H 1+] Acid Base [H+] Acidic [H+] = [OH-] Neutral p. H = -log [H 1+] Acid Base [H+] Acidic [H+] = [OH-] Neutral](http://slidetodoc.com/presentation_image_h/94ef6f4c8e68bccb876ab5d0e02c00a8/image-20.jpg)
p. H = -log [H 1+] Acid Base [H+] Acidic [H+] = [OH-] Neutral p. H = 7 Basic [OH-]
![p. H ﻣﺤﺎﺳﺒﺎﺕ p. H = -log[H 3 O+] = 10 -p. H + p. H ﻣﺤﺎﺳﺒﺎﺕ p. H = -log[H 3 O+] = 10 -p. H +](http://slidetodoc.com/presentation_image_h/94ef6f4c8e68bccb876ab5d0e02c00a8/image-21.jpg)
p. H ﻣﺤﺎﺳﺒﺎﺕ p. H = -log[H 3 O+] = 10 -p. H + p. OH = 14 p. OH [H 3 O+] [OH-] = 1 x 10 -14 p. OH = -log[OH-] = 10 -p. OH [OH-]
![p. H = - log [H+] Given: p. H = 4. 6 p. H p. H = - log [H+] Given: p. H = 4. 6 p. H](http://slidetodoc.com/presentation_image_h/94ef6f4c8e68bccb876ab5d0e02c00a8/image-22.jpg)
p. H = - log [H+] Given: p. H = 4. 6 p. H = - log [H+] choose proper equation 4. 6 = - log [H+] substitute p. H value in equation - 4. 6 = log [H+] 2 nd log - 4. 6 = log [H+] = 2. 51 x 10 -5 M 10 x antilog determine the [hydronium ion] multiply both sides by -1 take antilog of both sides Recall, [H+] = [H 3 O+] You can check your answer by working backwards. p. H = - log [H+] p. H = - log [2. 51 x 10 -5 M] p. H = 4. 6
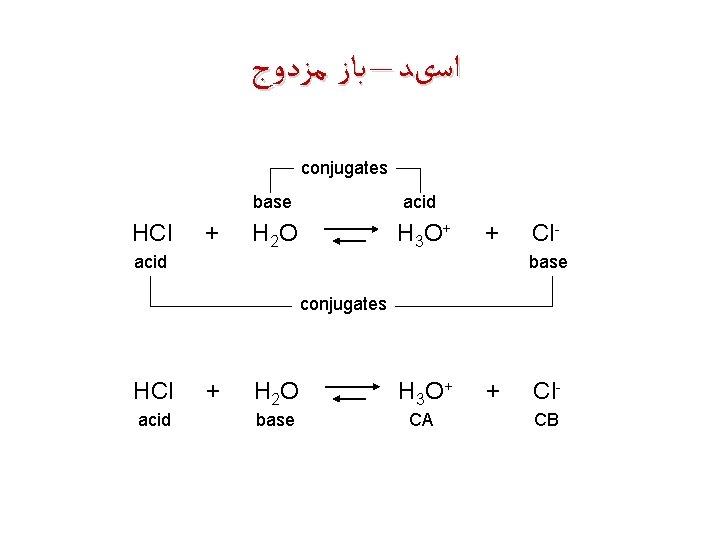
ﺍﺳیﺪ – ﺑﺎﺯ ﻣﺰﺩﻭﺝ conjugates HCl + base acid H 2 O H 3 O + + acid Clbase conjugates HCl acid + H 2 O H 3 O + base CA + Cl. CB
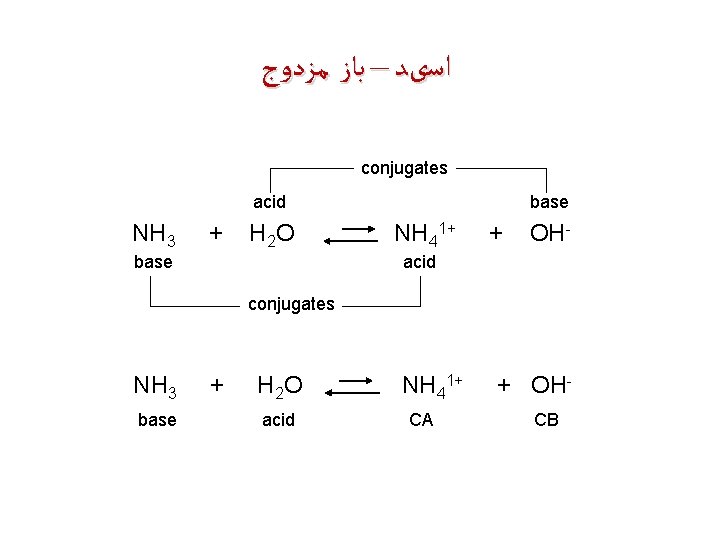
ﺍﺳیﺪ – ﺑﺎﺯ ﻣﺰﺩﻭﺝ conjugates acid NH 3 + H 2 O base NH 41+ + OH- acid conjugates NH 3 base + H 2 O acid NH 41+ CA + OHCB
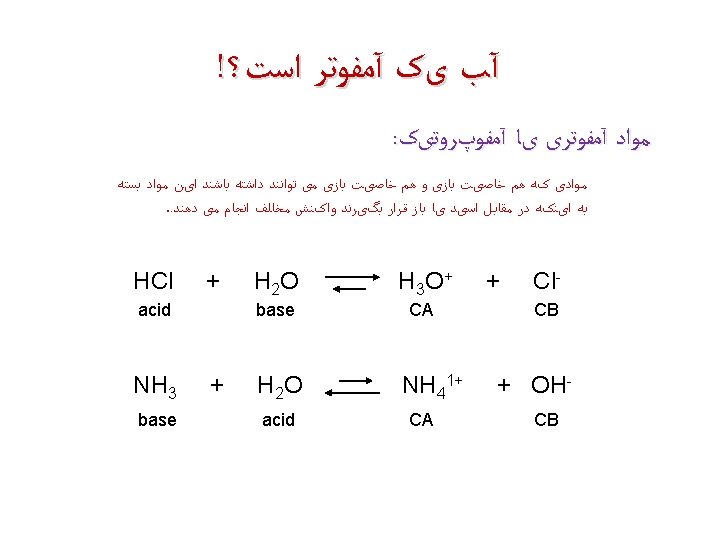
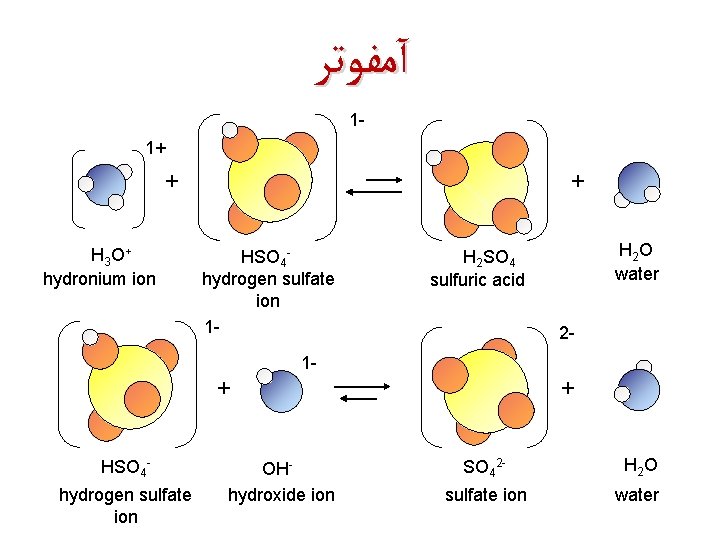
آﻤﻔﻮﺗﺮ 11+ + H 3 O + hydronium ion + HSO 4 hydrogen sulfate ion 1 - H 2 O water H 2 SO 4 sulfuric acid 2 - 1 - + HSO 4 hydrogen sulfate ion OHhydroxide ion + SO 42 - H 2 O sulfate ion water
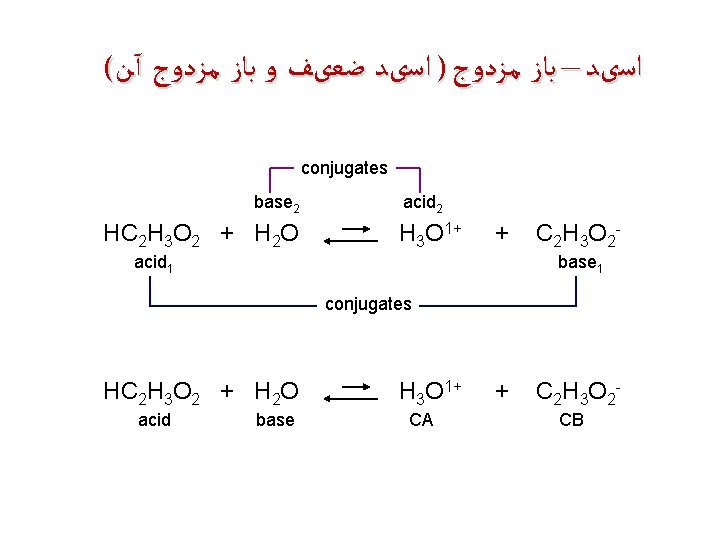
( ﺍﺳیﺪ – ﺑﺎﺯ ﻣﺰﺩﻭﺝ ) ﺍﺳیﺪ ﺿﻌیﻒ ﻭ ﺑﺎﺯ ﻣﺰﺩﻭﺝ آﻦ conjugates base 2 HC 2 H 3 O 2 + H 2 O acid 2 H 3 O 1+ + acid 1 C 2 H 3 O 2 base 1 conjugates HC 2 H 3 O 2 + H 2 O acid base H 3 O 1+ CA + C 2 H 3 O 2 CB

کﺎﻏﺬ ﻟیﺘﻤﻮﺱ Copyright © 2007 Pearson Benjamin Cummings. All rights reserved.
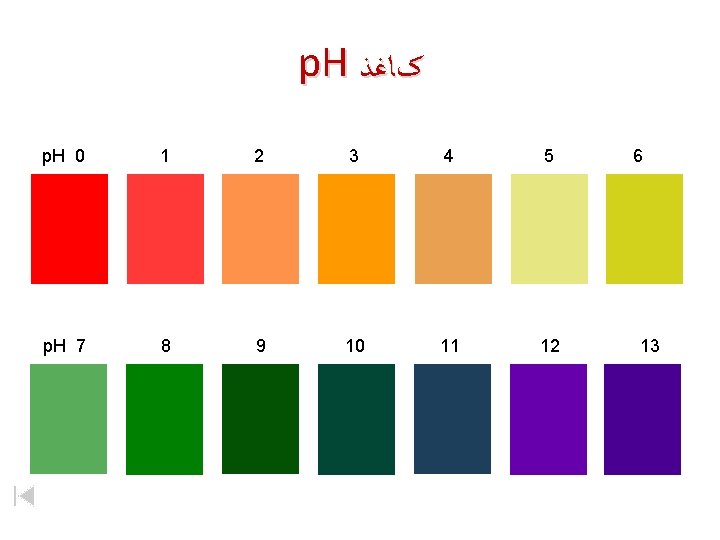
p. H کﺎﻏﺬ p. H 0 1 2 3 4 5 p. H 7 8 9 10 11 12 6 13
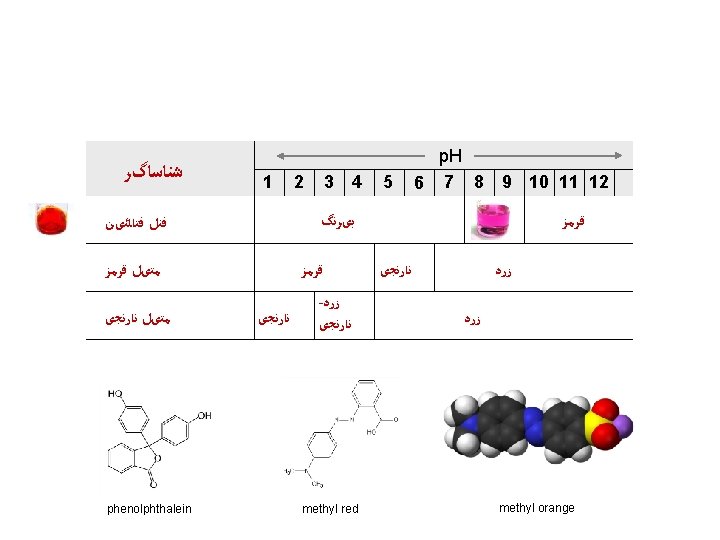
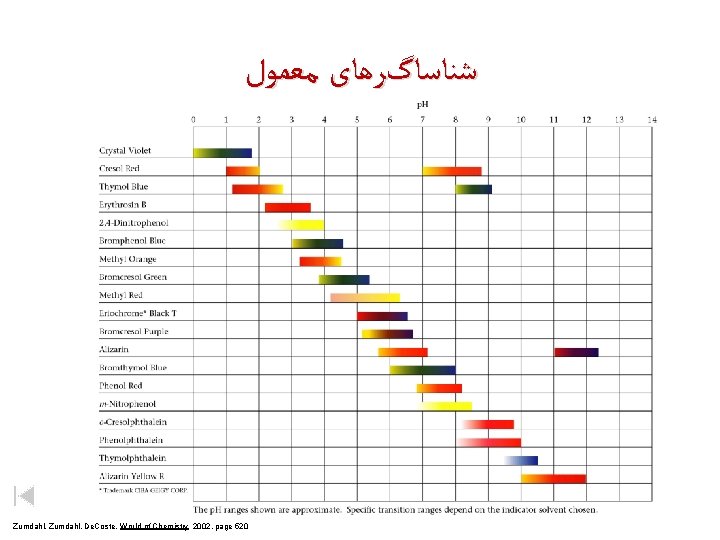
ﺷﻨﺎﺳﺎگﺮﻫﺎی ﻣﻌﻤﻮﻝ Zumdahl, De. Coste, World of Chemistry 2002, page 520
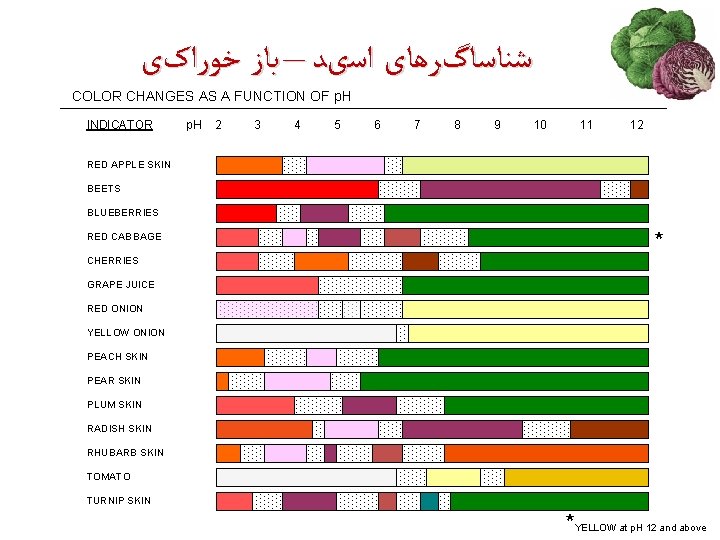
ﺷﻨﺎﺳﺎگﺮﻫﺎی ﺍﺳیﺪ – ﺑﺎﺯ ﺧﻮﺭﺍکی COLOR CHANGES AS A FUNCTION OF p. H INDICATOR p. H 2 3 4 5 6 7 8 9 10 11 12 RED APPLE SKIN BEETS BLUEBERRIES RED CABBAGE * CHERRIES GRAPE JUICE RED ONION YELLOW ONION PEACH SKIN PEAR SKIN PLUM SKIN RADISH SKIN RHUBARB SKIN TOMATO TURNIP SKIN *YELLOW at p. H 12 and above

کﻠﻢ ﺑﻨﻔﺶ Copyright © 2007 Pearson Benjamin Cummings. All rights reserved.
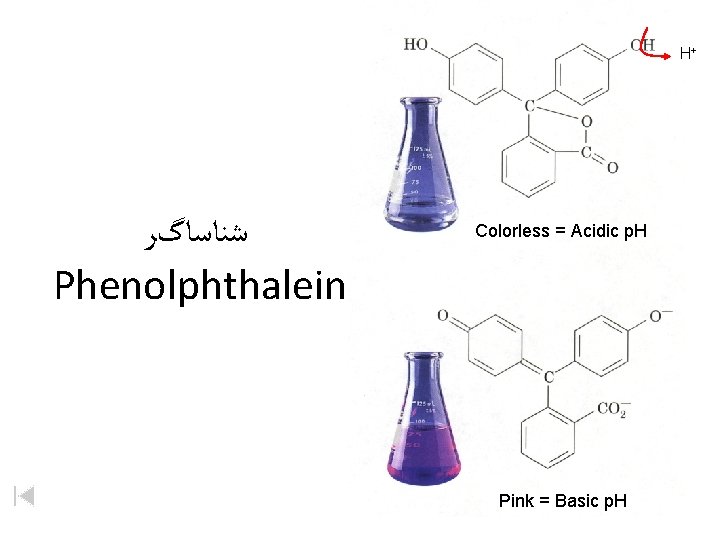
H+ ﺷﻨﺎﺳﺎگﺮ Phenolphthalein Colorless = Acidic p. H Pink = Basic p. H
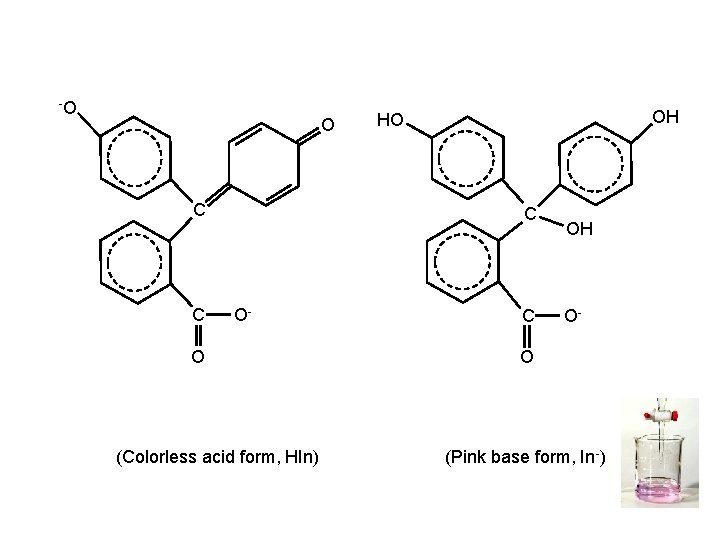
-O O C C OH HO C O- O (Colorless acid form, HIn) C OH O- O (Pink base form, In-)
- Slides: 35