Coordination Polymerization Ziegler Natta Processes Stereoregular Polymerization Cationic










- Slides: 31

Coordination Polymerization Ziegler Natta Processes

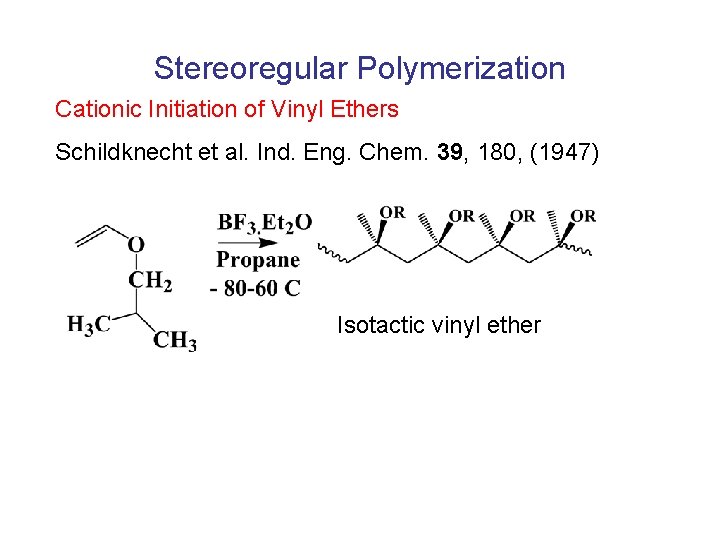
Stereoregular Polymerization Cationic Initiation of Vinyl Ethers Schildknecht et al. Ind. Eng. Chem. 39, 180, (1947) Isotactic vinyl ether

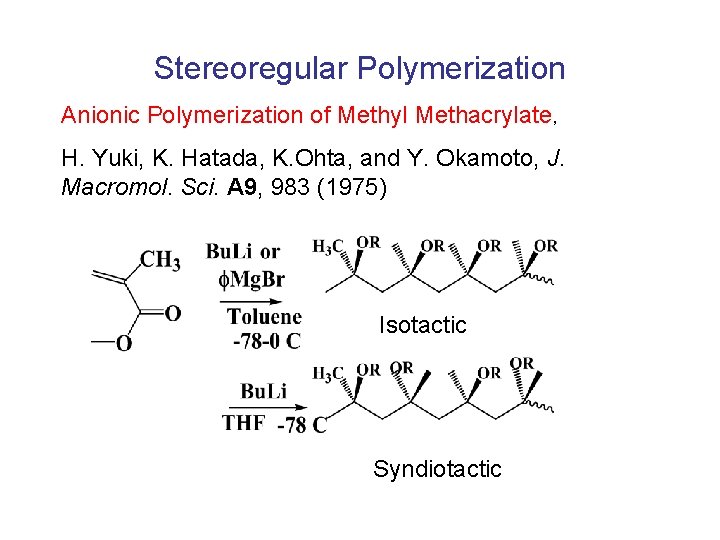
Stereoregular Polymerization Anionic Polymerization of Methyl Methacrylate, H. Yuki, K. Hatada, K. Ohta, and Y. Okamoto, J. Macromol. Sci. A 9, 983 (1975) Isotactic Syndiotactic

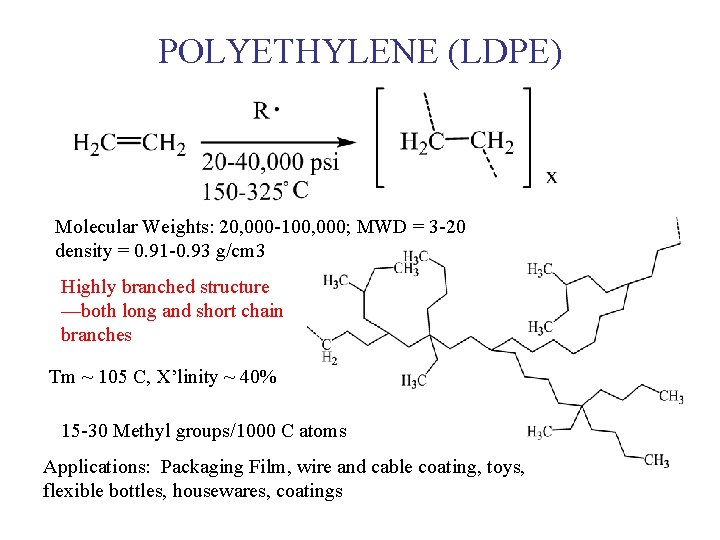
POLYETHYLENE (LDPE) Molecular Weights: 20, 000 -100, 000; MWD = 3 -20 density = 0. 91 -0. 93 g/cm 3 Highly branched structure —both long and short chain branches Tm ~ 105 C, X’linity ~ 40% 15 -30 Methyl groups/1000 C atoms Applications: Packaging Film, wire and cable coating, toys, flexible bottles, housewares, coatings

Ziegler’s Discovery • 1953 K. Ziegler, E. Holzkamp, H. Breil and H. Martin • Angew. Chemie 67, 426, 541 (1955); 76, 545 (1964). + Ni(Ac. Ac) + Cr(Ac. Ac) + Zr(Ac. Ac) Same result White Ppt. (Not reported by Holzkamp) White Ppt. (Eureka! reported by Breil)

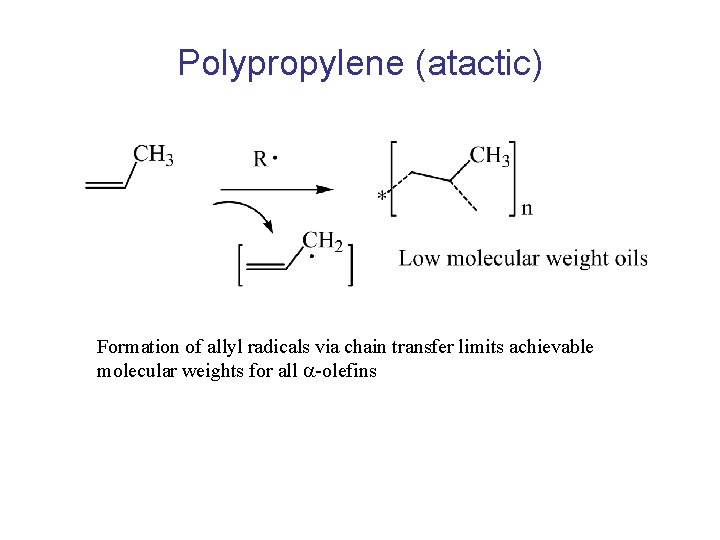
Polypropylene (atactic) Formation of allyl radicals via chain transfer limits achievable molecular weights for all -olefins

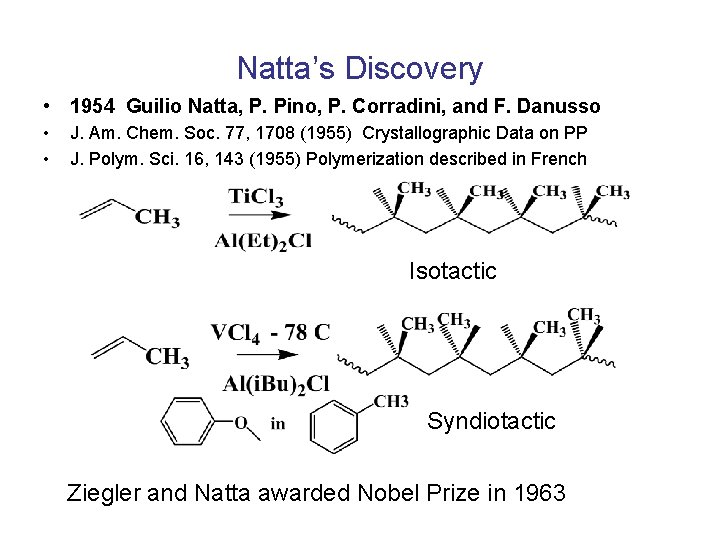
Natta’s Discovery • 1954 Guilio Natta, P. Pino, P. Corradini, and F. Danusso • • J. Am. Chem. Soc. 77, 1708 (1955) Crystallographic Data on PP J. Polym. Sci. 16, 143 (1955) Polymerization described in French Isotactic Syndiotactic Ziegler and Natta awarded Nobel Prize in 1963

Polypropylene (isotactic) Density ~ 0. 9 -0. 91 g/cm 3—very high strength to weight ratio Tm = 165 -175 C: Use temperature up to 120 C Copolymers with 2 -5% ethylene—increases clarity and toughness of films Applications: dishwasher safe plastic ware, carpet yarn, fibers and ropes, webbing, auto parts

Polyethylene (HDPE) Essentially linear structure Few long chain branches, 0. 5 -3 methyl groups/ 1000 C atoms Molecular Weights: 50, 000 -250, 000 for molding compounds 250, 000 -1, 500, 000 for pipe compounds >1, 500, 000 super abrasion resistance—medical implants MWD = 3 -20 density = 0. 94 -0. 96 g/cm 3 Tm ~ 133 -138 C, X’linity ~ 80% Generally opaque Applications: Bottles, drums, pipe, conduit, sheet, film

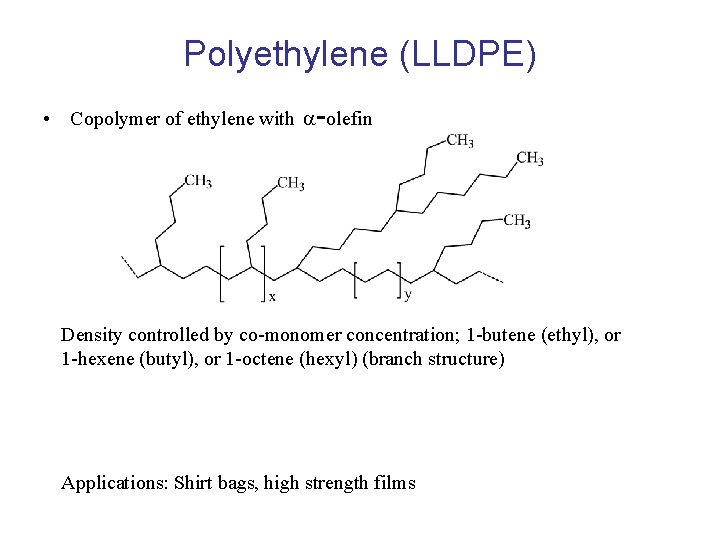
Polyethylene (LLDPE) • Copolymer of ethylene with -olefin Density controlled by co-monomer concentration; 1 -butene (ethyl), or 1 -hexene (butyl), or 1 -octene (hexyl) (branch structure) Applications: Shirt bags, high strength films

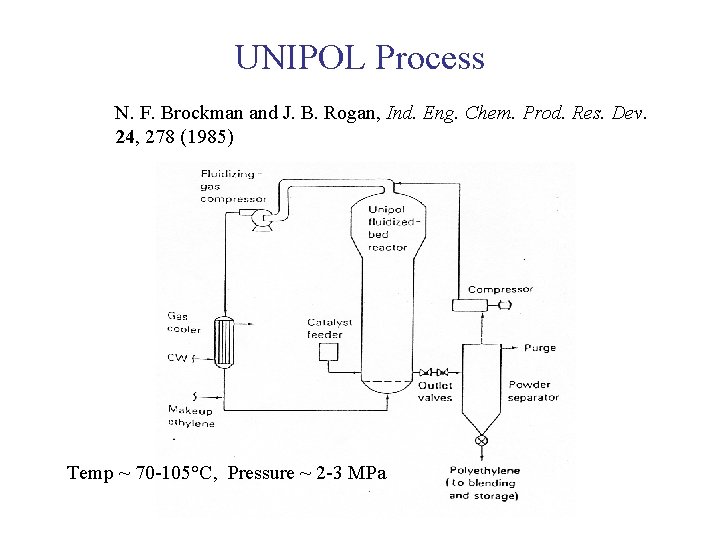
UNIPOL Process N. F. Brockman and J. B. Rogan, Ind. Eng. Chem. Prod. Res. Dev. 24, 278 (1985) Temp ~ 70 -105°C, Pressure ~ 2 -3 MPa

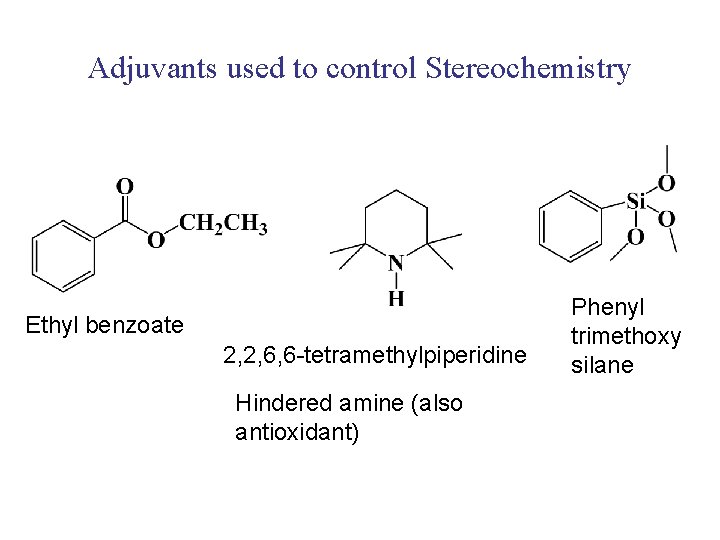
CATALYST PREPARATION Ball mill Mg. Cl 2 (support) with Ti. Cl 4 to produce maximum surface area and incorporate Ti atoms in Mg. Cl 2 crystals Add Al(Et)3 along with Lewis base like ethyl benzoate Al(Et)3 reduces Ti. Cl 4 to form active complex Ethyl Benzoate modifies active sites to enhance stereoselectivity Catalyst activity 50 -2000 kg polypropylene/g Ti with isospecificity of > 90%

Catalyst Formation Al. Et 3 + Ti. Cl 4 → Et. Ti. Cl 3 + Et 2 Al. Cl + Ti. Cl 4 → Et. Ti. Cl 3 + Et. Al. Cl 2 Et. Ti. Cl 3 + Al. Et 3 → Et 2 Ti. Cl 2 + Et. Al. Cl 2 Et. Ti. Cl 3 → Ti. Cl 3 + Et. (source of radical products) Et. + Ti. Cl 4 → Et. Cl + Ti. Cl 3 + Al. Et 3 → Et. Ti. Cl 2 + Et 2 Al. Cl

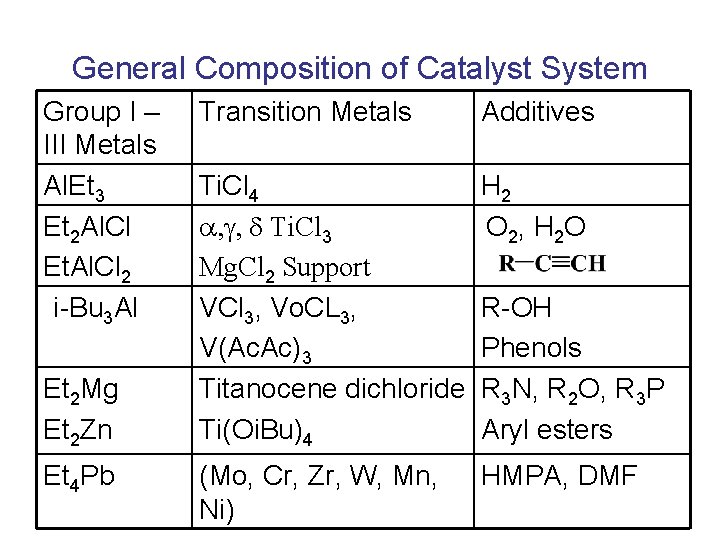
General Composition of Catalyst System Group I – III Metals Al. Et 3 Et 2 Al. Cl Et. Al. Cl 2 i-Bu 3 Al Et 2 Mg Et 2 Zn Et 4 Pb Transition Metals Additives Ti. Cl 4 , g, d Ti. Cl 3 Mg. Cl 2 Support VCl 3, Vo. CL 3, V(Ac. Ac)3 Titanocene dichloride Ti(Oi. Bu)4 H 2 O 2, H 2 O (Mo, Cr, Zr, W, Mn, Ni) HMPA, DMF R-OH Phenols R 3 N, R 2 O, R 3 P Aryl esters

Adjuvants used to control Stereochemistry Ethyl benzoate 2, 2, 6, 6 -tetramethylpiperidine Hindered amine (also antioxidant) Phenyl trimethoxy silane

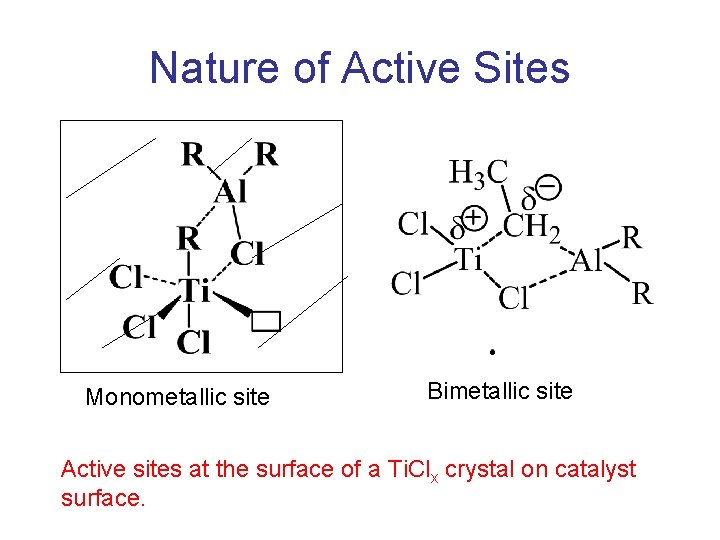
Nature of Active Sites Monometallic site Bimetallic site Active sites at the surface of a Ti. Clx crystal on catalyst surface.

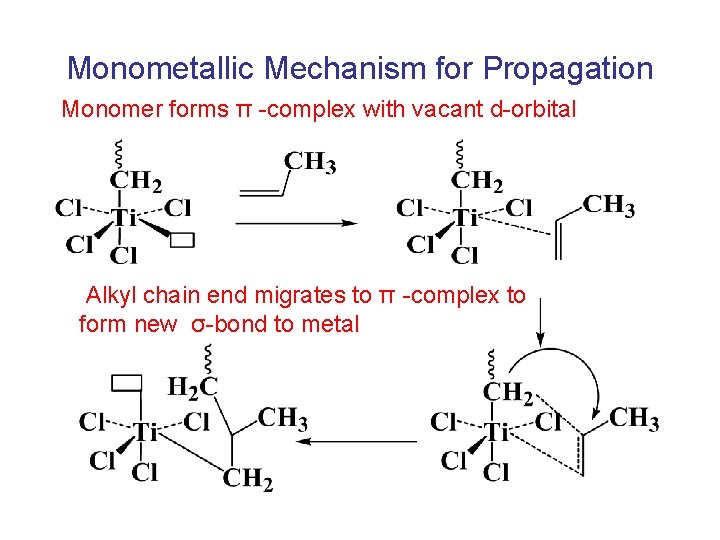
Monometallic Mechanism for Propagation Monomer forms π -complex with vacant d-orbital Alkyl chain end migrates to π -complex to form new σ-bond to metal

Monometallic Mechanism for Propagation Chain must migrate to original site to assure formation of isotactic structure If no migration occurs, syndiotactic placements will form.

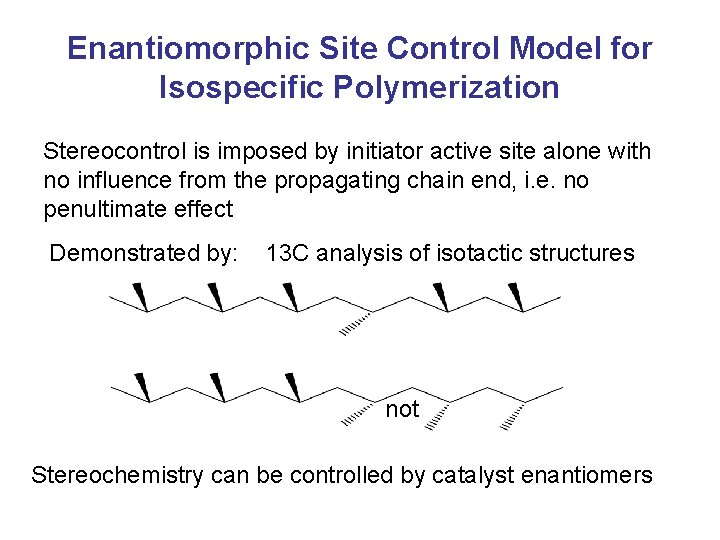
Enantiomorphic Site Control Model for Isospecific Polymerization Stereocontrol is imposed by initiator active site alone with no influence from the propagating chain end, i. e. no penultimate effect Demonstrated by: 13 C analysis of isotactic structures not Stereochemistry can be controlled by catalyst enantiomers

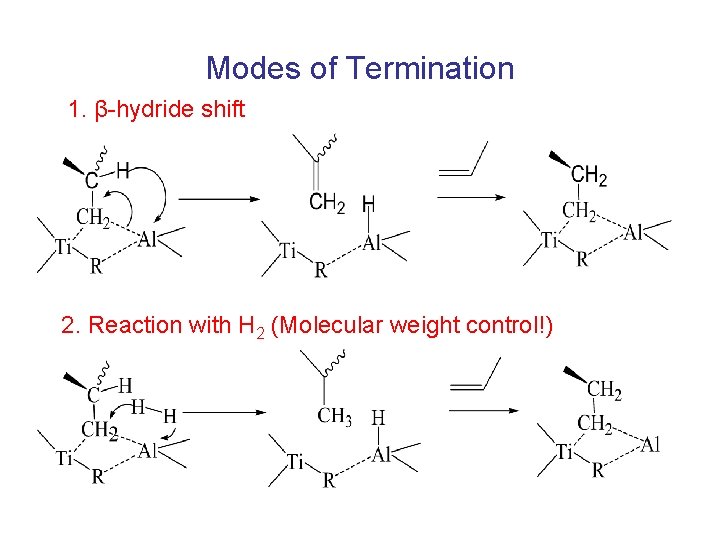
Modes of Termination 1. β-hydride shift 2. Reaction with H 2 (Molecular weight control!)

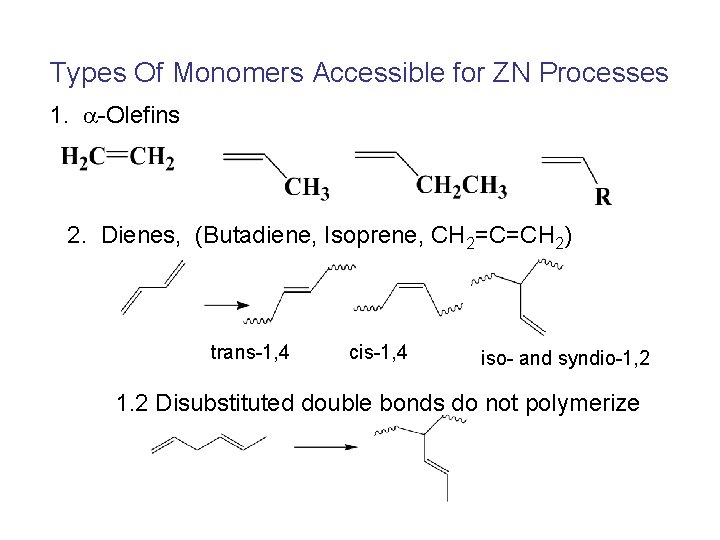
Types Of Monomers Accessible for ZN Processes 1. -Olefins 2. Dienes, (Butadiene, Isoprene, CH 2=C=CH 2) trans-1, 4 cis-1, 4 iso- and syndio-1, 2 1. 2 Disubstituted double bonds do not polymerize

Ethylene-Propylene Diene Rubber (EPDM) S. Cesca, Macromolecular Reviews, 10, 1 -231 (1975) Catalyst soluble in hydrocarbons Continuous catalyst addition required to maintain activity Rigid control of monomer feed ratio required to assure incorporation of propylene and diene monomers

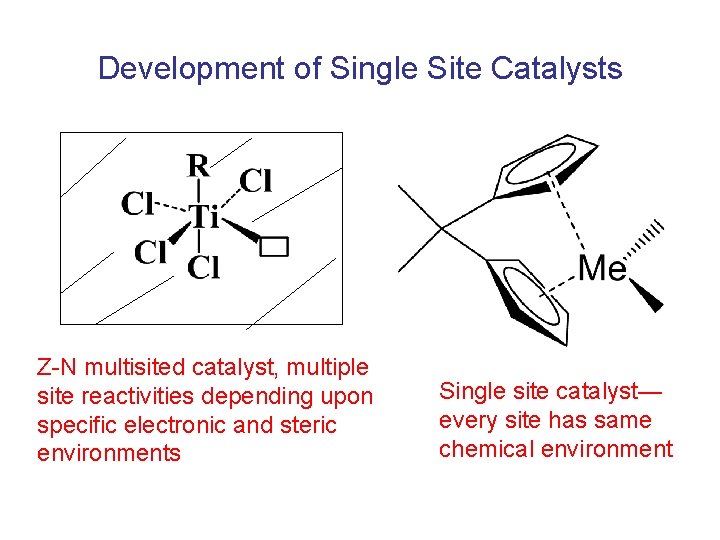
Development of Single Site Catalysts Z-N multisited catalyst, multiple site reactivities depending upon specific electronic and steric environments Single site catalyst— every site has same chemical environment

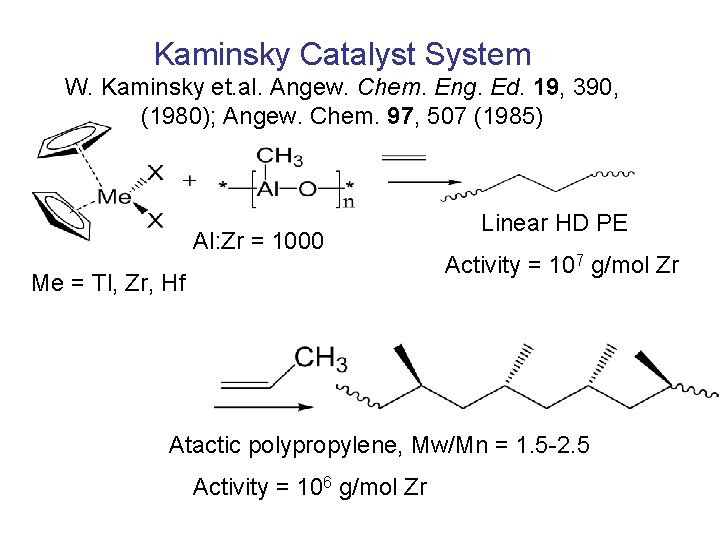
Kaminsky Catalyst System W. Kaminsky et. al. Angew. Chem. Eng. Ed. 19, 390, (1980); Angew. Chem. 97, 507 (1985) Al: Zr = 1000 Me = Tl, Zr, Hf Linear HD PE Activity = 107 g/mol Zr Atactic polypropylene, Mw/Mn = 1. 5 -2. 5 Activity = 106 g/mol Zr

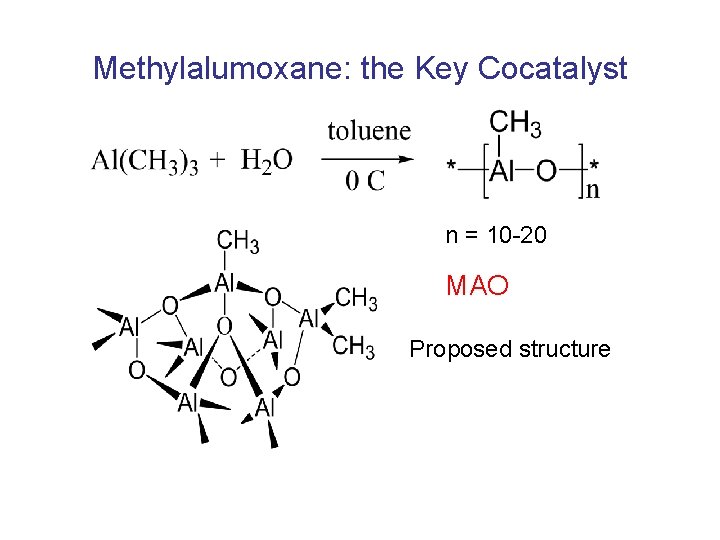
Methylalumoxane: the Key Cocatalyst n = 10 -20 MAO Proposed structure

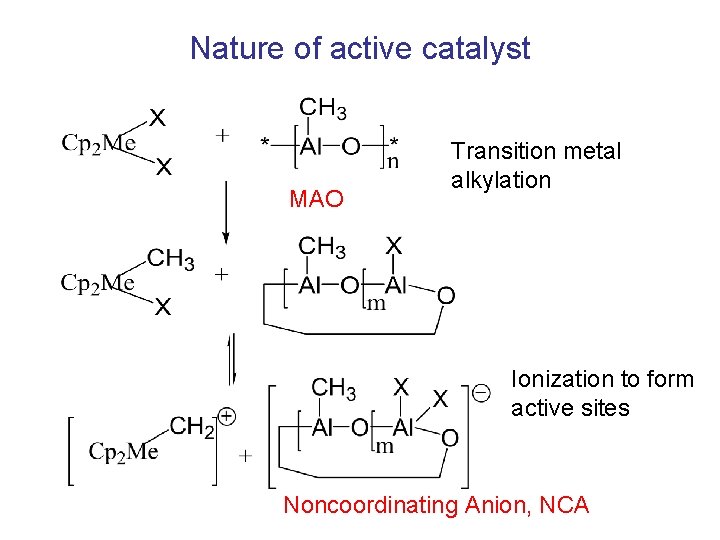
Nature of active catalyst MAO Transition metal alkylation Ionization to form active sites Noncoordinating Anion, NCA

Homogeneous Z-N Polymerization Advantages: High Catalytic Activity Impressive control of stereochemistry Well defined catalyst precursors Design of Polymer microstructures, including chiral polymers Disadvantages: Requires large excess of Aluminoxane (counter-ion) Higher tendency for chain termination: β-H elimination, etc. Limited control of molecular weight distribution

Evolution of single site catalysts Date 1950’s Metallocene Stereo control Performance None Moderate Mw PE Some comonomer incorporation Early 1980’s None High MW PE Better comonomer incorporation

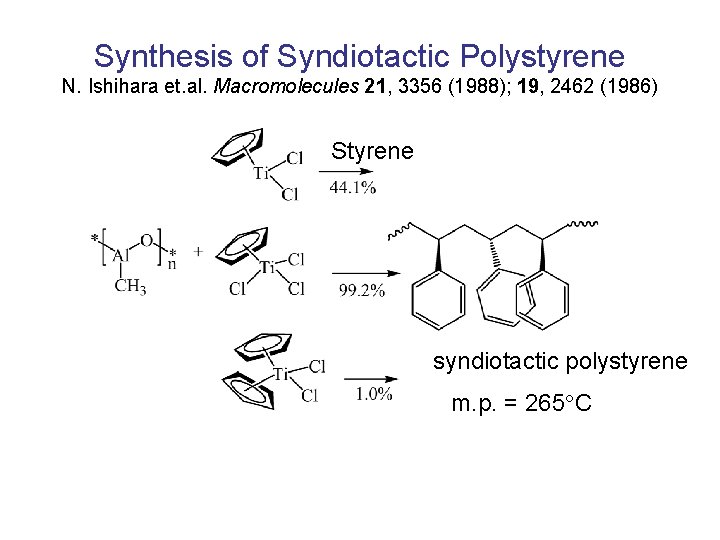
Synthesis of Syndiotactic Polystyrene N. Ishihara et. al. Macromolecules 21, 3356 (1988); 19, 2462 (1986) Styrene syndiotactic polystyrene m. p. = 265 C

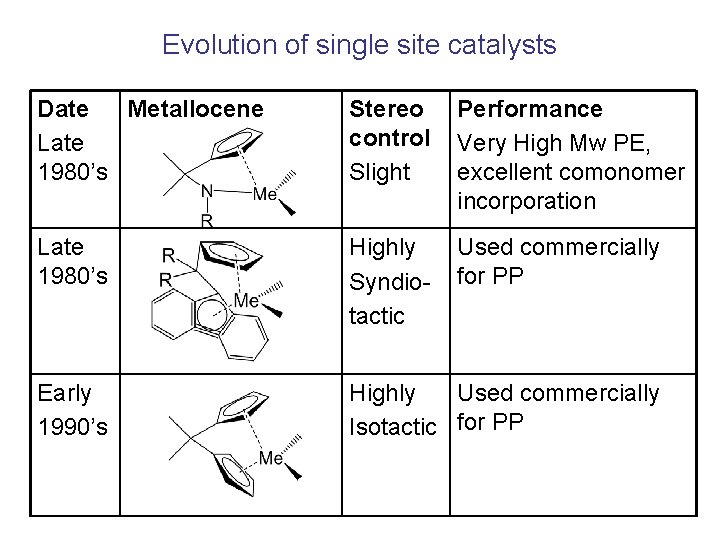
Evolution of single site catalysts Date Metallocene Late 1980’s Stereo control Slight Performance Very High Mw PE, excellent comonomer incorporation Late 1980’s Highly Syndiotactic Used commercially for PP Early 1990’s Highly Used commercially Isotactic for PP

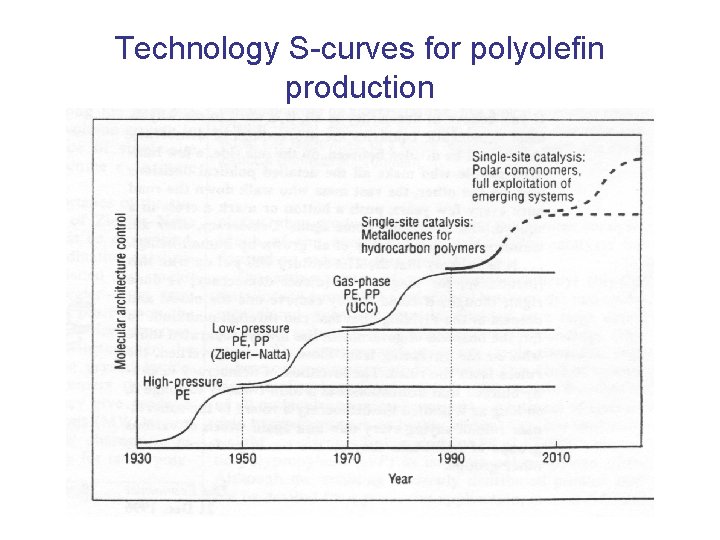
Technology S-curves for polyolefin production