COORDINATION COMPOUNDS MODULE 26 Agitha R Menon PGTChemistry


![Examples K 2[Co(NH 3)2 Cl 4] potassium diamminetetrachloridocobaltate(II) [Co(NH 3)4 Cl 2]Cl tetraamminedichloridocobalt(III) chloride Examples K 2[Co(NH 3)2 Cl 4] potassium diamminetetrachloridocobaltate(II) [Co(NH 3)4 Cl 2]Cl tetraamminedichloridocobalt(III) chloride](https://slidetodoc.com/presentation_image_h/02f5347b0f0d2d8ced4101dd57d65a61/image-5.jpg)

![Examples triamminetriaquachromium(III) chloride [Cr(NH 3)3(H 2 O)3 ] Cl 3 Dichloridobis(ethane-1, 2 -diamine)cobalt(III)ion [Co. Examples triamminetriaquachromium(III) chloride [Cr(NH 3)3(H 2 O)3 ] Cl 3 Dichloridobis(ethane-1, 2 -diamine)cobalt(III)ion [Co.](https://slidetodoc.com/presentation_image_h/02f5347b0f0d2d8ced4101dd57d65a61/image-7.jpg)

- Slides: 8

CO-ORDINATION COMPOUNDS MODULE 2/6 Agitha R Menon PGT(Chemistry) AECS, Indore

IUPAC Nomenclature of Coordination Complexes RULES FOR NAMING CO-ORDINATION COMPOUNDS 1. Cation is written first then the anion. 2. Ligands should be written in alphabetical order before metal ion. Prefix (di, tri, tetra) indicates number of each ligand. When the names of the ligands include a numerical prefix, then the terms, bis, tris, tetrakis are used. These are placed within parentheses( ) § 3. No prefix will be given for counter ions. 4. Oxidation state of metal ion should be written in parentheses( ) in Roman Numerals 5. If complex ion is an anion, metal name should end with the suffix -ate and if it is a cation, then original name of the metal is retained.

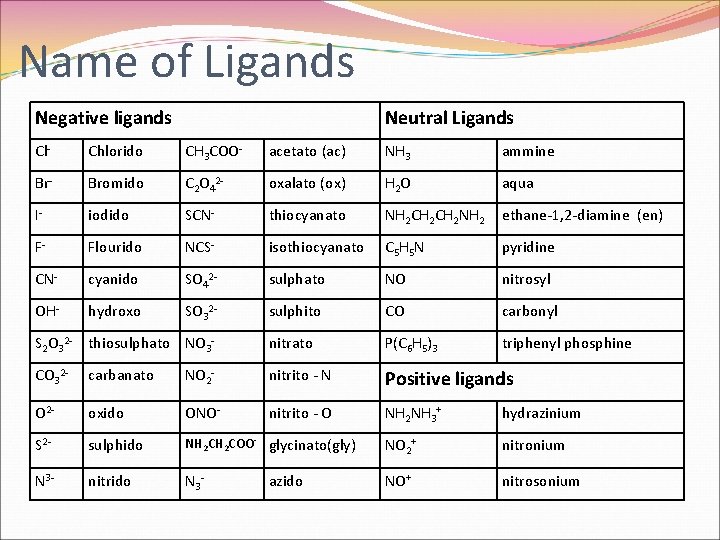
Name of Ligands Negative ligands Neutral Ligands Cl- Chlorido CH 3 COO- acetato (ac) NH 3 ammine Br- Bromido C 2 O 42 - oxalato (ox) H 2 O aqua I- iodido SCN- thiocyanato NH 2 CH 2 NH 2 ethane-1, 2 -diamine (en) F- Flourido NCS- isothiocyanato C 5 H 5 N pyridine CN- cyanido SO 42 - sulphato NO nitrosyl OH- hydroxo SO 32 - sulphito CO carbonyl S 2 O 32 - thiosulphato NO 3 - nitrato P(C 6 H 5)3 triphenyl phosphine CO 32 - carbanato NO 2 - nitrito - N Positive ligands O 2 - oxido ONO- nitrito - O NH 2 NH 3+ hydrazinium S 2 - sulphido NH 2 COO- glycinato(gly) NO 2+ nitronium N 3 - nitrido N 3 - NO+ nitrosonium azido

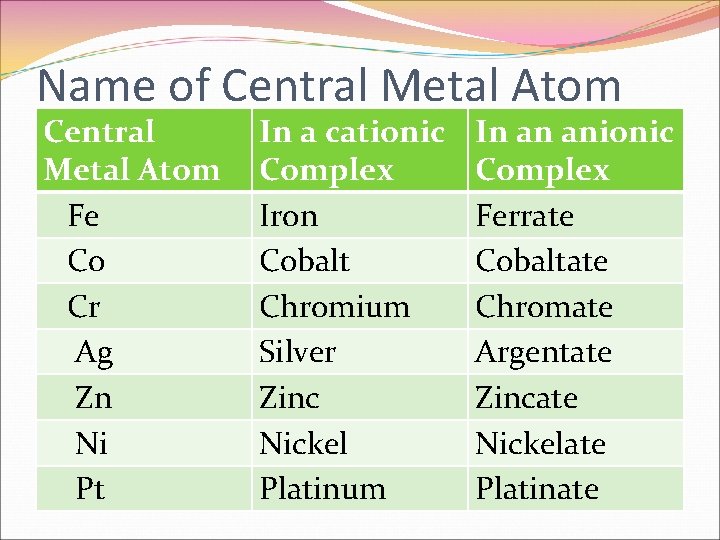
Name of Central Metal Atom Fe Co Cr Ag Zn Ni Pt In a cationic Complex Iron Cobalt Chromium Silver Zinc Nickel Platinum In an anionic Complex Ferrate Cobaltate Chromate Argentate Zincate Nickelate Platinate
![Examples K 2CoNH 32 Cl 4 potassium diamminetetrachloridocobaltateII CoNH 34 Cl 2Cl tetraamminedichloridocobaltIII chloride Examples K 2[Co(NH 3)2 Cl 4] potassium diamminetetrachloridocobaltate(II) [Co(NH 3)4 Cl 2]Cl tetraamminedichloridocobalt(III) chloride](https://slidetodoc.com/presentation_image_h/02f5347b0f0d2d8ced4101dd57d65a61/image-5.jpg)
Examples K 2[Co(NH 3)2 Cl 4] potassium diamminetetrachloridocobaltate(II) [Co(NH 3)4 Cl 2]Cl tetraamminedichloridocobalt(III) chloride [Pt(NH 3)2 Cl(NO 2)] Diamminechloridonitrito-N-platinum(II)

Nomenclature of Coordination Complexes RULES FOR WRITING FORMULAE OF CO-ORDINATION COMPOUNDS 1. The central atom is listed first. 2. The ligands are then listed in alphabetical order. 3. The formula for the entire coordination entity is enclosed in square brackets [ ]. 4. When ligands are polyatomic, their formulae are enclosed in parentheses( ). 5. There should be no space between the ligands and the metal within a coordination sphere. 6. The formula of a charged coordination entity is to be written with the residual as a right superscript with the number before the sign.
![Examples triamminetriaquachromiumIII chloride CrNH 33H 2 O3 Cl 3 Dichloridobisethane1 2 diaminecobaltIIIion Co Examples triamminetriaquachromium(III) chloride [Cr(NH 3)3(H 2 O)3 ] Cl 3 Dichloridobis(ethane-1, 2 -diamine)cobalt(III)ion [Co.](https://slidetodoc.com/presentation_image_h/02f5347b0f0d2d8ced4101dd57d65a61/image-7.jpg)
Examples triamminetriaquachromium(III) chloride [Cr(NH 3)3(H 2 O)3 ] Cl 3 Dichloridobis(ethane-1, 2 -diamine)cobalt(III)ion [Co. Cl 2(en)2]+ Potassium trioxalatochromate(III) K 3[Cr(C 2 O 4)3] diamminesilver(I) dicyanidoargentate(I) [Ag(NH 3)2][Ag(CN)2]

References: 1. NCERT Class XII Chemistry Vol 1 2. Google images