Coordination Complexes Summary Examples of ligand substitution reactions
![Coordination Complexes - Summary Examples of ligand substitution reactions [Ni(H 2 O)6]2+(aq) + 6 Coordination Complexes - Summary Examples of ligand substitution reactions [Ni(H 2 O)6]2+(aq) + 6](https://slidetodoc.com/presentation_image_h2/37009d222c8d93989393293c206ff9e2/image-1.jpg)
Coordination Complexes - Summary Examples of ligand substitution reactions [Ni(H 2 O)6]2+(aq) + 6 NH 3(aq) [Ni(NH 3)6]2+(aq) + 6 H 2 O(l) Need to know: Crystal field theory - Inert, labile, thermodynamic stability - Structural aspects of complexes (a) cis-trans in octahedral/square planar complexes (b) Chiral forms (enantiomers) in octahedral complexes Spectrochemcial series: Cl- < H 2 O < NH 3 < CN-
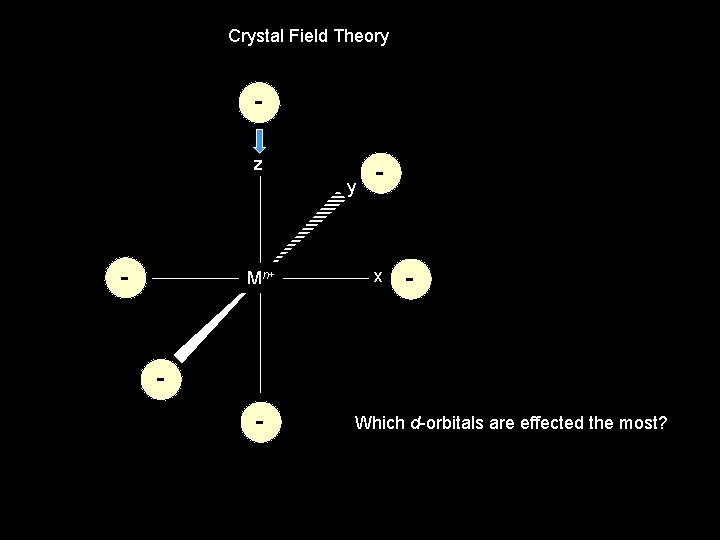
Crystal Field Theory z y - Mn+ - x - - Which d-orbitals are effected the most?
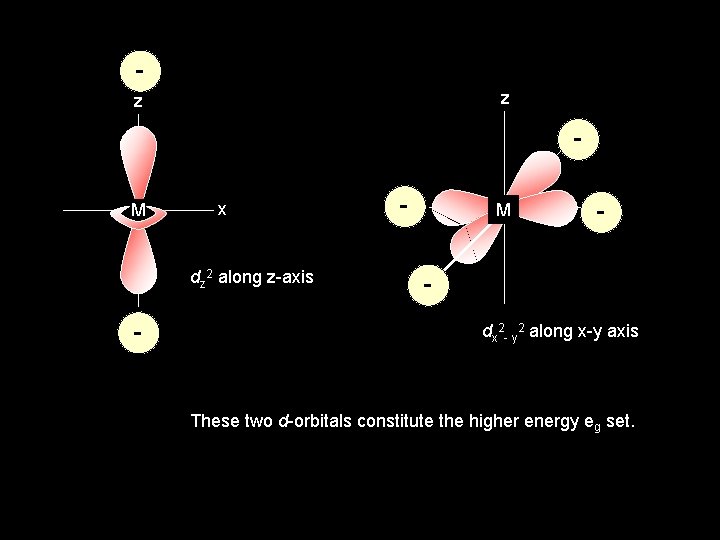
z z x- M x dz 2 along z-axis - - M x- dx 2 - y 2 along x-y axis These two d-orbitals constitute the higher energy eg set.
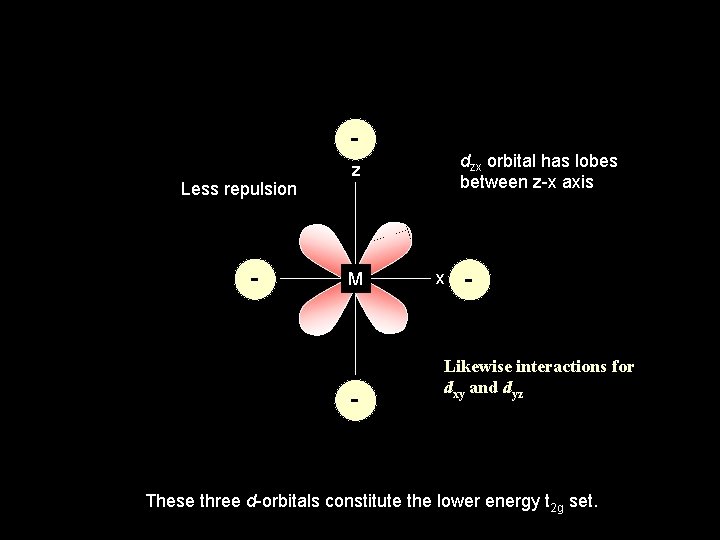
Less repulsion - dzx orbital has lobes between z-x axis z M - x - Likewise interactions for dxy and dyz These three d-orbitals constitute the lower energy t 2 g set.
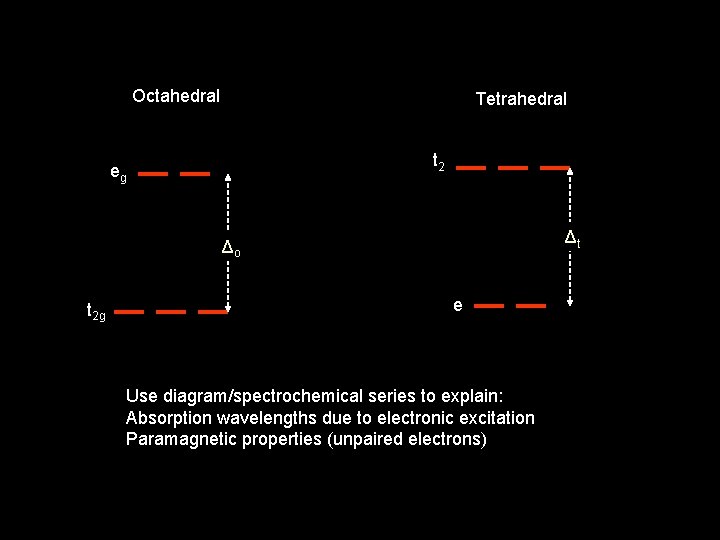
Octahedral Tetrahedral t 2 eg Δt Δo t 2 g e Use diagram/spectrochemical series to explain: Absorption wavelengths due to electronic excitation Paramagnetic properties (unpaired electrons)
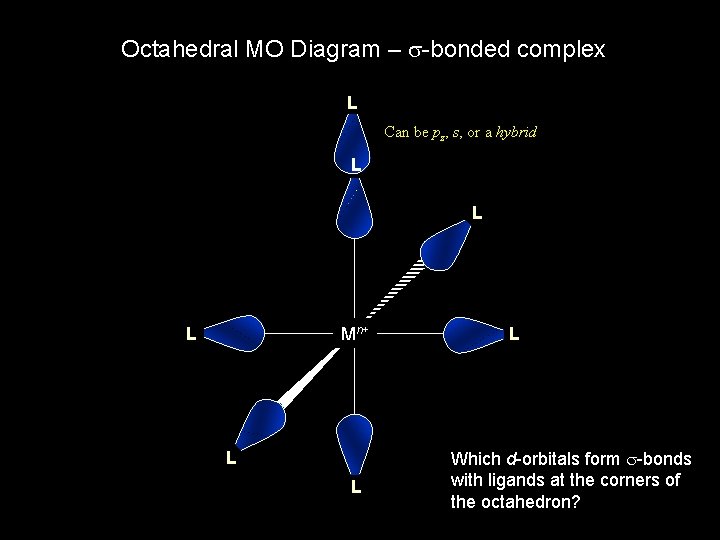
Octahedral MO Diagram – s-bonded complex L Can be pz, s, or a hybrid L L z y L Mn+ L L x L Which d-orbitals form s-bonds with ligands at the corners of the octahedron?
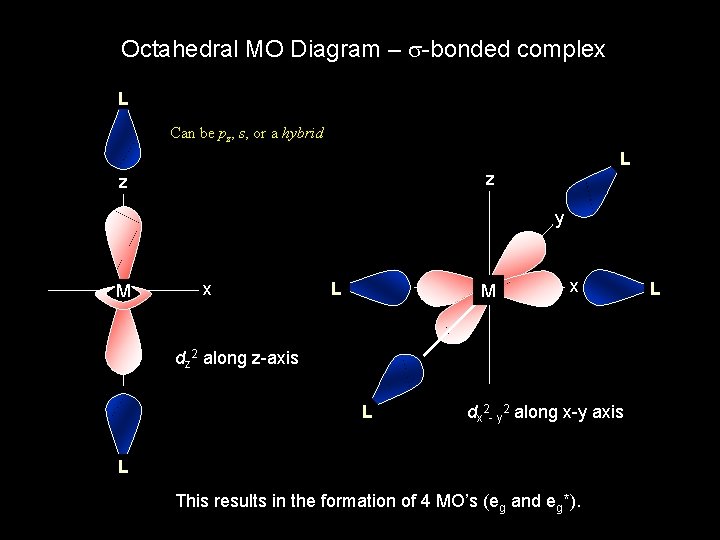
Octahedral MO Diagram – s-bonded complex L Can be pz, s, or a hybrid L z z y M x L M x dz 2 along z-axis L dx 2 - y 2 along x-y axis L This results in the formation of 4 MO’s (eg and eg*). L
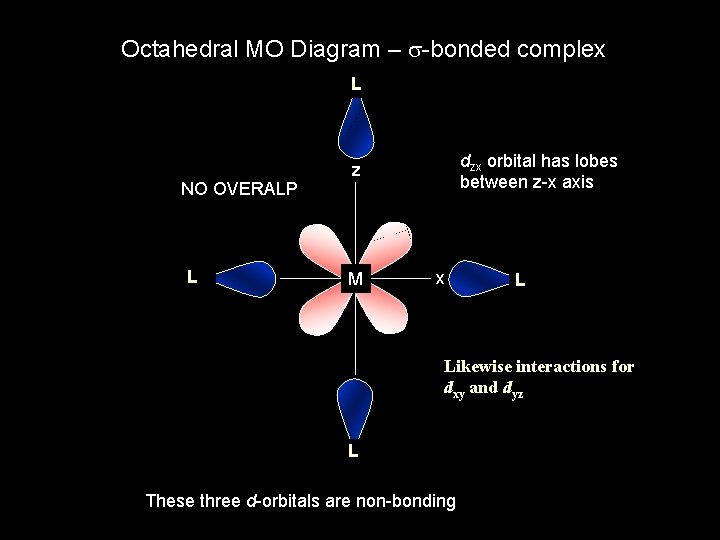
Octahedral MO Diagram – s-bonded complex L NO OVERALP L dzx orbital has lobes between z-x axis z M x L Likewise interactions for dxy and dyz L These three d-orbitals are non-bonding
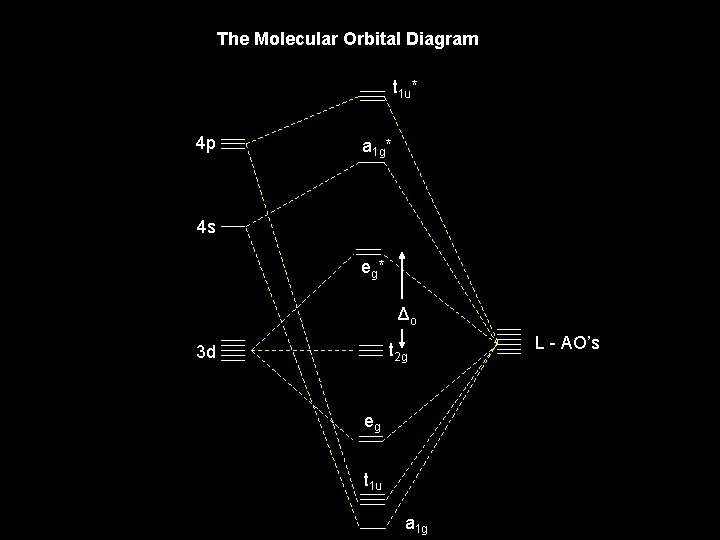
The Molecular Orbital Diagram t 1 u* 4 p a 1 g* 4 s eg * Δo t 2 g 3 d eg t 1 u a 1 g L - AO’s

Color in coordination complexes The colors are determined by Δ. Different ligands generate crystal fields of different strength. When the molecules absorb visible light, excited electrons jump from lower energy t 2 g to the higher energy eg orbital. The Δ (difference between energies of the two orbitals) is equal to the energy of the absorbed photon, and related inversely to the wavelength of the light. Weaker field ligands with smaller Δ emit light of longer λ and thus lower v. Similarly, stronger field ligands with larger Δ emit light of shorter λ and thus higher v. λ Absorbed 400 nm Violet absorbed 450 nm Blue absorbed 490 nm Blue-green absorbed 570 nm Yellow-green absorbed 580 nm Yellow absorbed 600 nm Orange absorbed 650 nm Red absorbed Color observed Green-yellow observed (λ 560 nm) Yellow observed (λ 600 nm) Red observed (λ 620 nm) Violet observed (λ 410 nm) Dark blue observed (λ 430 nm) Blue observed (λ 450 nm) Green observed (λ 520 nm)
![Problems 1. (a) When water ligands in [Ti(H 2 O)6]3+ are replaced by CN- Problems 1. (a) When water ligands in [Ti(H 2 O)6]3+ are replaced by CN-](http://slidetodoc.com/presentation_image_h2/37009d222c8d93989393293c206ff9e2/image-11.jpg)
Problems 1. (a) When water ligands in [Ti(H 2 O)6]3+ are replaced by CN- ligands to give [Ti(CN)6]3 -, the maximum absorption shifts from 500 nm to 450 nm. Is this shift in the expected direction? Explain. What color do you expect to observe for this ion? CN- is a stronger field ligand than H 2 O. Therefore the energy separation between the t 2 g and eg levels is greater. Ti 3+ is a d 1 metal ion. Do = hc/l. If D is larger, l is smaller. [Ti(CN)6]3 eg [Ti(H 2 O)6]3+ eg A(lmax) = 500 nm Solution appears red A(lmax) = 450 nm Do Do Solution appears yellow t 2 g
![(b) The [Fe(H 2 O)6]3+ ion has a pale purple color, and the [Fe(CN)6]3 (b) The [Fe(H 2 O)6]3+ ion has a pale purple color, and the [Fe(CN)6]3](http://slidetodoc.com/presentation_image_h2/37009d222c8d93989393293c206ff9e2/image-12.jpg)
(b) The [Fe(H 2 O)6]3+ ion has a pale purple color, and the [Fe(CN)6]3 - ion has a ruby red color. What are the approximate wavelengths of the maximum absorption for each ion? Is the shift of wavelength in the expected direction? Explain. CN- is a stronger field ligand than H 2 O. Therefore the energy separation between the t 2 g and eg levels is greater. Fe 3+ is a d 5 metal ion. Do = hc/l. If D is larger, l is smaller.

- Slides: 13