Coordination Chemistry compounds made up of metals that


![Cl- outer sphere Cl- complex – charged coordination compounds [Co(NH 3)6]3+ [Co(NH 3)6]Cl 3 Cl- outer sphere Cl- complex – charged coordination compounds [Co(NH 3)6]3+ [Co(NH 3)6]Cl 3](https://slidetodoc.com/presentation_image_h2/f8bc0c213ad48089e8d100ad73440b56/image-5.jpg)


![[Co(NH 3)6]Cl 3 hexaamminecobalt(III) chloride [Cu(NH 3)4]Cl 2 tetraamminecopper(II) chloride [Cr(NH 3)4 Cl 2] [Co(NH 3)6]Cl 3 hexaamminecobalt(III) chloride [Cu(NH 3)4]Cl 2 tetraamminecopper(II) chloride [Cr(NH 3)4 Cl 2]](https://slidetodoc.com/presentation_image_h2/f8bc0c213ad48089e8d100ad73440b56/image-8.jpg)

![[Co(en)2 Br 2]I dibromobis(ethylenediamine)cobalt(III) iodide [Ru(Cl)(CN)(H 2 O)2(NH 3)2]Cl 3 diamminediaquachlorocyanoruthenium(V) chloride [Co(en)2 Br 2]I dibromobis(ethylenediamine)cobalt(III) iodide [Ru(Cl)(CN)(H 2 O)2(NH 3)2]Cl 3 diamminediaquachlorocyanoruthenium(V) chloride](https://slidetodoc.com/presentation_image_h2/f8bc0c213ad48089e8d100ad73440b56/image-10.jpg)
![When the coordination complex is negative, that is, [complex]-, the metal gets an “–ate” When the coordination complex is negative, that is, [complex]-, the metal gets an “–ate”](https://slidetodoc.com/presentation_image_h2/f8bc0c213ad48089e8d100ad73440b56/image-11.jpg)

![Na[Co. Cl 4] sodium tetrachlorocobaltate(III) tetraammineaquachloroiron(III) nitrate [Fe(NH 3)4(H 2 O)Cl](NO 3)2 Na 2[Sn(OH)6] Na[Co. Cl 4] sodium tetrachlorocobaltate(III) tetraammineaquachloroiron(III) nitrate [Fe(NH 3)4(H 2 O)Cl](NO 3)2 Na 2[Sn(OH)6]](https://slidetodoc.com/presentation_image_h2/f8bc0c213ad48089e8d100ad73440b56/image-13.jpg)
3 diamminebis(ethylenediamine)iridium(III) nitrate tetraammineplatinum(IV) hexachloroplatinate(IV) [Pt(NH 3)4] [Pt. Cl 6]2 [Ir(en)2(NH 3)2](NO 3)3 diamminebis(ethylenediamine)iridium(III) nitrate tetraammineplatinum(IV) hexachloroplatinate(IV) [Pt(NH 3)4] [Pt. Cl 6]2](https://slidetodoc.com/presentation_image_h2/f8bc0c213ad48089e8d100ad73440b56/image-14.jpg)


![[Co(NH 3)4 Cl 2]Cl green purple [Co(NH 3)4 Cl 2]Cl green purple](https://slidetodoc.com/presentation_image_h2/f8bc0c213ad48089e8d100ad73440b56/image-17.jpg)



![[Cr(H 2 O)6]Cl 3 hexaaquachromium(III) chloride [Cr(H 2 O)5 Cl]Cl 2 · H 2 [Cr(H 2 O)6]Cl 3 hexaaquachromium(III) chloride [Cr(H 2 O)5 Cl]Cl 2 · H 2](https://slidetodoc.com/presentation_image_h2/f8bc0c213ad48089e8d100ad73440b56/image-27.jpg)


![[Co(NH 3)5(ONO)]2+ [Co(NH 3)5(NO 2)]2+ pentaamminenitritocobalt(III) pentaamminenitrocobalt(III) [Co(NH 3)5(ONO)]2+ [Co(NH 3)5(NO 2)]2+ pentaamminenitritocobalt(III) pentaamminenitrocobalt(III)](https://slidetodoc.com/presentation_image_h2/f8bc0c213ad48089e8d100ad73440b56/image-31.jpg)

![Both enantiomers of [Co(en)3]3+ Δ-tris(ethylenediamine)cobalt(III) ( look down C 3 axis; turn right, drills Both enantiomers of [Co(en)3]3+ Δ-tris(ethylenediamine)cobalt(III) ( look down C 3 axis; turn right, drills](https://slidetodoc.com/presentation_image_h2/f8bc0c213ad48089e8d100ad73440b56/image-33.jpg)
- Slides: 33

Coordination Chemistry – compounds made up of metals that are covalently bound to other molecules or ions Ligands – molecules (or ions) that donate both electrons to the metal and form a coordinate covalent bond – covalent bond formed between a ligand metal where both electrons in the covalent bond originate from the ligand

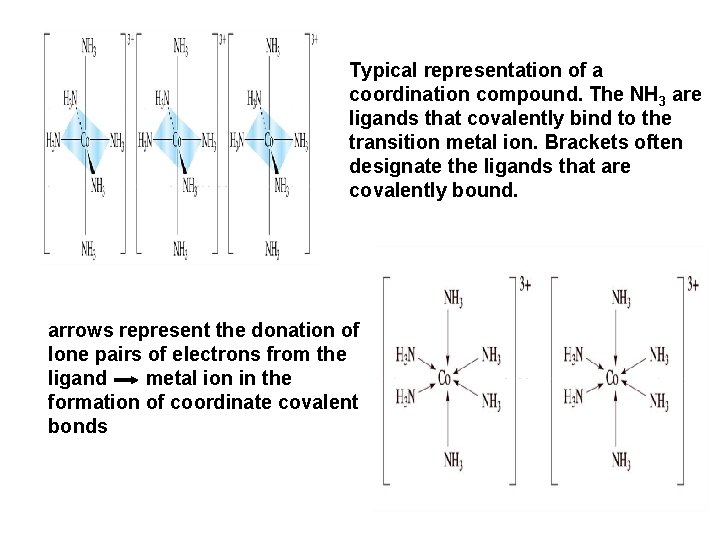
Typical representation of a coordination compound. The NH 3 are ligands that covalently bind to the transition metal ion. Brackets often designate the ligands that are covalently bound. arrows represent the donation of lone pairs of electrons from the ligand metal ion in the formation of coordinate covalent bonds

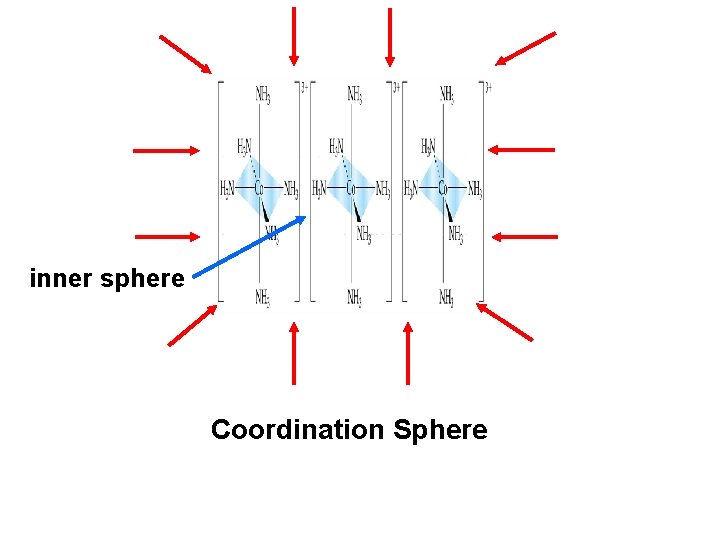
Coordination Compound – compounds made up of ligands that are covalently bound to a transition metal Coordination Number – number of covalent bonds to the central transition metal element Coordination Sphere – area where the ligands are attached to the transition metal

inner sphere Coordination Sphere
![Cl outer sphere Cl complex charged coordination compounds CoNH 363 CoNH 36Cl 3 Cl- outer sphere Cl- complex – charged coordination compounds [Co(NH 3)6]3+ [Co(NH 3)6]Cl 3](https://slidetodoc.com/presentation_image_h2/f8bc0c213ad48089e8d100ad73440b56/image-5.jpg)
Cl- outer sphere Cl- complex – charged coordination compounds [Co(NH 3)6]3+ [Co(NH 3)6]Cl 3


![CoNH 36Cl 3 hexaamminecobaltIII chloride CuNH 34Cl 2 tetraamminecopperII chloride CrNH 34 Cl 2 [Co(NH 3)6]Cl 3 hexaamminecobalt(III) chloride [Cu(NH 3)4]Cl 2 tetraamminecopper(II) chloride [Cr(NH 3)4 Cl 2]](https://slidetodoc.com/presentation_image_h2/f8bc0c213ad48089e8d100ad73440b56/image-8.jpg)
[Co(NH 3)6]Cl 3 hexaamminecobalt(III) chloride [Cu(NH 3)4]Cl 2 tetraamminecopper(II) chloride [Cr(NH 3)4 Cl 2] NO 3 tetraamminedichlorochromium(III) nitrate [Ni(CO)4] tetracarbonylnickel(0)

![Coen2 Br 2I dibromobisethylenediaminecobaltIII iodide RuClCNH 2 O2NH 32Cl 3 diamminediaquachlorocyanorutheniumV chloride [Co(en)2 Br 2]I dibromobis(ethylenediamine)cobalt(III) iodide [Ru(Cl)(CN)(H 2 O)2(NH 3)2]Cl 3 diamminediaquachlorocyanoruthenium(V) chloride](https://slidetodoc.com/presentation_image_h2/f8bc0c213ad48089e8d100ad73440b56/image-10.jpg)
[Co(en)2 Br 2]I dibromobis(ethylenediamine)cobalt(III) iodide [Ru(Cl)(CN)(H 2 O)2(NH 3)2]Cl 3 diamminediaquachlorocyanoruthenium(V) chloride
![When the coordination complex is negative that is complex the metal gets an ate When the coordination complex is negative, that is, [complex]-, the metal gets an “–ate”](https://slidetodoc.com/presentation_image_h2/f8bc0c213ad48089e8d100ad73440b56/image-11.jpg)
When the coordination complex is negative, that is, [complex]-, the metal gets an “–ate” ending Zn �zincate Ni �nickelate V �vanadate (drop “–ium” ending) Pt � platinate Cr � chromate Co � cobaltate

Some metals get the historical Latin name with an “–ate” ending (complex is negative, [complex]- ) Cu �cuprate Fe �ferrate Ag �argentate Sn �stannate Au �aurate Pb �plumbate
![NaCo Cl 4 sodium tetrachlorocobaltateIII tetraammineaquachloroironIII nitrate FeNH 34H 2 OClNO 32 Na 2SnOH6 Na[Co. Cl 4] sodium tetrachlorocobaltate(III) tetraammineaquachloroiron(III) nitrate [Fe(NH 3)4(H 2 O)Cl](NO 3)2 Na 2[Sn(OH)6]](https://slidetodoc.com/presentation_image_h2/f8bc0c213ad48089e8d100ad73440b56/image-13.jpg)
Na[Co. Cl 4] sodium tetrachlorocobaltate(III) tetraammineaquachloroiron(III) nitrate [Fe(NH 3)4(H 2 O)Cl](NO 3)2 Na 2[Sn(OH)6] sodium hexahydroxostannate(IV)
3 diamminebis(ethylenediamine)iridium(III) nitrate tetraammineplatinum(IV) hexachloroplatinate(IV) [Pt(NH 3)4] [Pt. Cl 6]2](https://slidetodoc.com/presentation_image_h2/f8bc0c213ad48089e8d100ad73440b56/image-14.jpg)
[Ir(en)2(NH 3)2](NO 3)3 diamminebis(ethylenediamine)iridium(III) nitrate tetraammineplatinum(IV) hexachloroplatinate(IV) [Pt(NH 3)4] [Pt. Cl 6]2

Alfred Werner 1866 - 1919

Co. Cl 3 ∙ 4 NH 3 2 distinct compounds Co. Cl 3 ∙ 4 NH 3 + 1 Ag. NO 3 [Co(NH 3)4 Cl 2]Cl 1 Ag. Cl
![CoNH 34 Cl 2Cl green purple [Co(NH 3)4 Cl 2]Cl green purple](https://slidetodoc.com/presentation_image_h2/f8bc0c213ad48089e8d100ad73440b56/image-17.jpg)
[Co(NH 3)4 Cl 2]Cl green purple

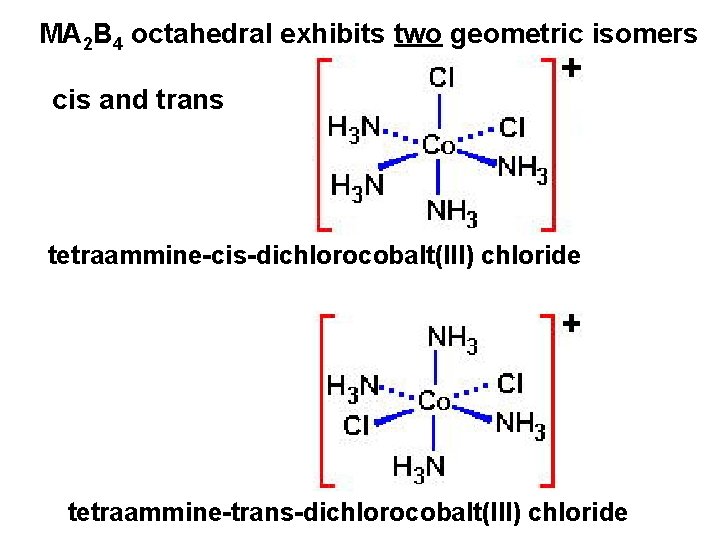
Various types of coordination compound isomers 1. Geometric isomers – ligands that differ only in spatial arrangement

MA 2 B 4 octahedral exhibits two geometric isomers cis and trans tetraammine-cis-dichlorocobalt(III) chloride tetraammine-trans-dichlorocobalt(III) chloride

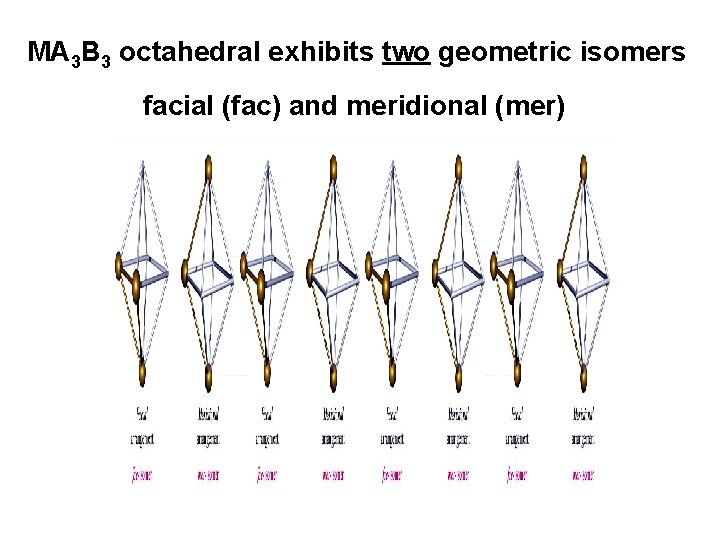
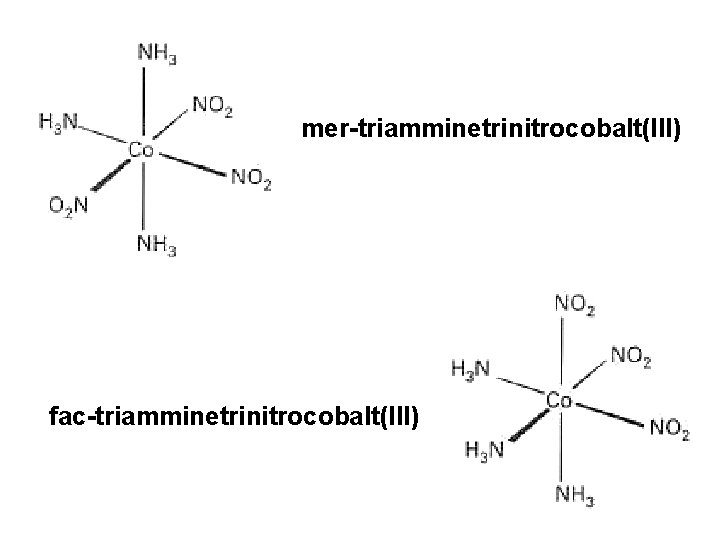
MA 3 B 3 octahedral exhibits two geometric isomers facial (fac) and meridional (mer)

mer-triamminetrinitrocobalt(III) fac-triamminetrinitrocobalt(III)

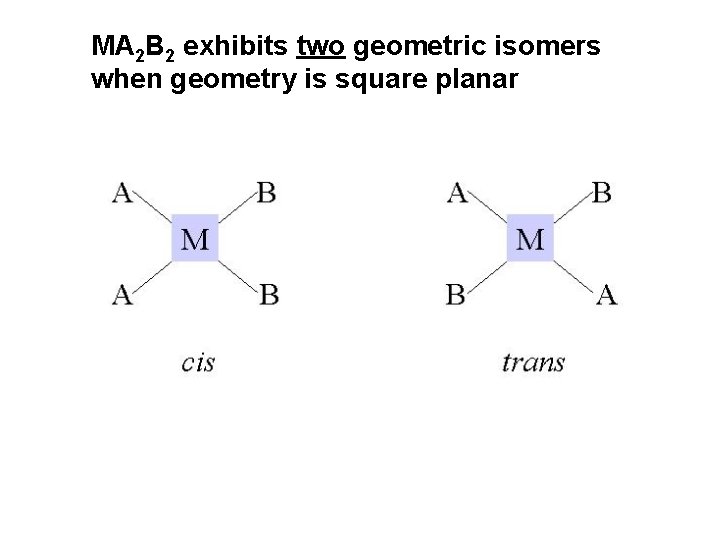
MA 2 B 2 exhibits two geometric isomers when geometry is square planar

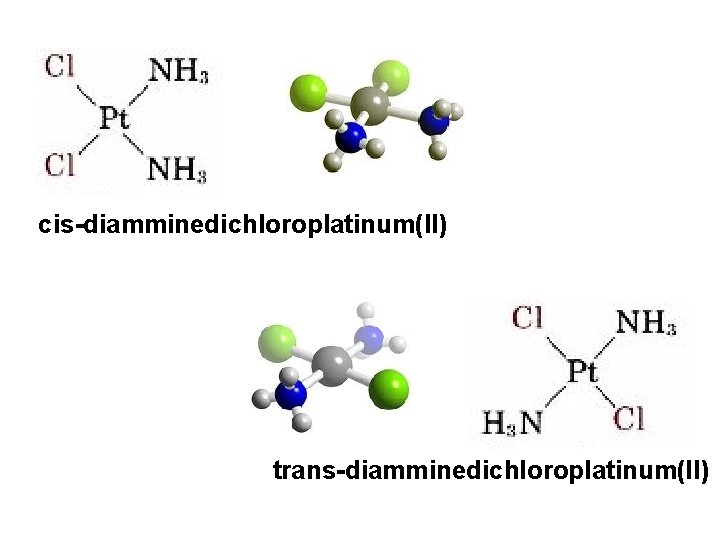
cis-diamminedichloroplatinum(II) trans-diamminedichloroplatinum(II)


2. Coordination isomers – both cation and anion are coordination complexes but vary in ligands or central element [Co(NH 3)6] [Cr(CN)6] vs. [Cr(NH 3)6] [Co(CN)6] both have same empirical formula: Co. Cr(NH 3)6(CN)6 [Co(NH 3)5(CN)] [Cr(NH 3)(CN)5] vs. [Co(NH 3)6] [Cr(CN)6]

3. Hydrate isomers – water is interchanged from inner sphere to outer sphere [Cr(H 2 O)6]Cl 3 - purple [Cr(H 2 O)5 Cl]Cl 2 · H 2 O - blue-green [Cr(H 2 O)4 Cl 2]Cl · 2 H 2 O - dark green
![CrH 2 O6Cl 3 hexaaquachromiumIII chloride CrH 2 O5 ClCl 2 H 2 [Cr(H 2 O)6]Cl 3 hexaaquachromium(III) chloride [Cr(H 2 O)5 Cl]Cl 2 · H 2](https://slidetodoc.com/presentation_image_h2/f8bc0c213ad48089e8d100ad73440b56/image-27.jpg)
[Cr(H 2 O)6]Cl 3 hexaaquachromium(III) chloride [Cr(H 2 O)5 Cl]Cl 2 · H 2 O pentaaquachlorochromium(III) chloride hydrate [Cr(H 2 O)4 Cl 2]Cl · 2 H 2 O tetraaqua-cis-dichlorochromium(III) chloride dihydrate


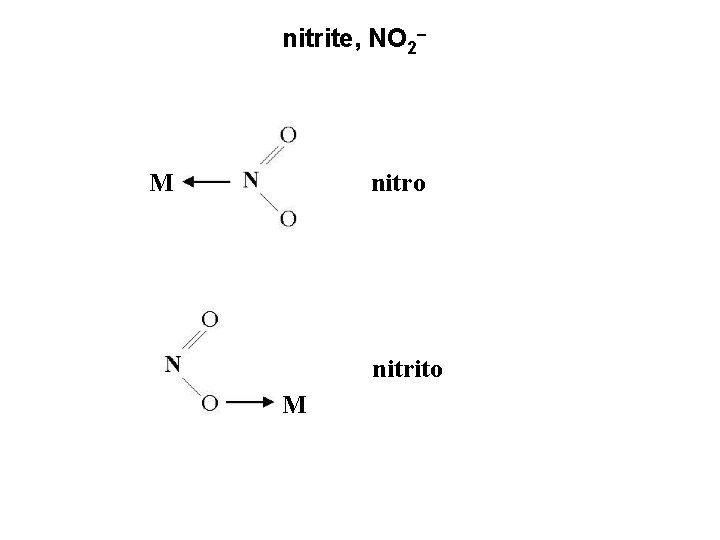
4. Linkage isomers – a ligand can coordinate to a metal in more than one way ambidentate – ligands that have the ability to coordinate to a metal through different atoms thiocyanate, SCN and nitrite, NO 2 M-SCN named S-thiocyanato or thiocyanato M-NCS named N-thiocyanato or isothiocyanato

nitrite, NO 2 M nitro nitrito M
![CoNH 35ONO2 CoNH 35NO 22 pentaamminenitritocobaltIII pentaamminenitrocobaltIII [Co(NH 3)5(ONO)]2+ [Co(NH 3)5(NO 2)]2+ pentaamminenitritocobalt(III) pentaamminenitrocobalt(III)](https://slidetodoc.com/presentation_image_h2/f8bc0c213ad48089e8d100ad73440b56/image-31.jpg)
[Co(NH 3)5(ONO)]2+ [Co(NH 3)5(NO 2)]2+ pentaamminenitritocobalt(III) pentaamminenitrocobalt(III)

5. Optical isomers – complex is chiral and will rotate a plane of polarized light all tris-chelates are D 3 and thus chiral
![Both enantiomers of Coen33 ΔtrisethylenediaminecobaltIII look down C 3 axis turn right drills Both enantiomers of [Co(en)3]3+ Δ-tris(ethylenediamine)cobalt(III) ( look down C 3 axis; turn right, drills](https://slidetodoc.com/presentation_image_h2/f8bc0c213ad48089e8d100ad73440b56/image-33.jpg)
Both enantiomers of [Co(en)3]3+ Δ-tris(ethylenediamine)cobalt(III) ( look down C 3 axis; turn right, drills into ground - Δ ) λ-tris(ethylenediamine)cobalt(III) ( look down C 3 axis; turn left, drills into ground - λ )