Conversion Factors Factors SI CONVERSIONS CONVERSIONS You must

























- Slides: 28

(Conversion Factors) Factors

SI CONVERSIONS: CONVERSIONS You must multiply the number of slices (32) by the proper conversion factor. The conversion factor should be the one that has in the denominator the same unit than the original value : 32 slices x = 4 pizzas

Practice: 1) How many millimeters are in 4 m? Determine the possible conversion factors: The two units in this problem are m and mm, if 1 m = 1, 000 mm, then • or 4 m x = 4 x 103 mm (4, 000 mm) Multiply when converting from larger to smaller units.

Practice: 2) How many meters are in 54 cm? Determine the conversion factors: The two units in this problem are m and cm, & 1 m = 100 cm, then or 54 cm x = 0. 54 m Divide when converting from smaller to larger units.

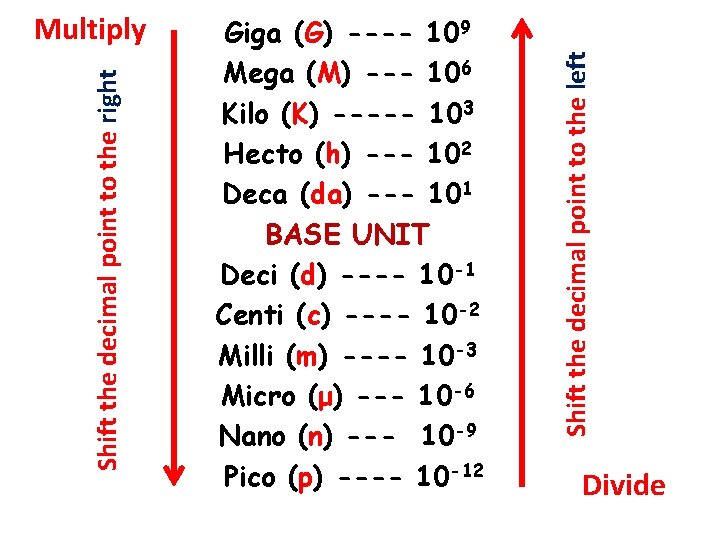
Giga (G) ----- 109 base units Mega (M) ----- 106 base units Kilo (k) ------ 103 base units Hecto (h) ----- 102 base units Deca (da) ----- 101 base units BASE UNIT (m, L, g, …. . ) Deci (d) ----- 10 -1 base units Centi (c) ----- 10 -2 base units Milli (m) ----- 10 -3 base units Micro (μ) ----- 10 -6 base units Nano (n) ----- 10 -9 base units Pico (p) ----- 10 -12 base units

Measurement and SI Units: Base units Meter (m) for length Liter (L) for volume Gram (g) for mass Second (s) for time Kelvin (K) for temperature Joule (J) for energy

Practice: 3) How many decaliters are in 3 kiloliters? Determine the possible conversion factors: The two units in this problem are da. L and k. L. 1 k. L = 102 da. L (because 103 -1 = 102 ). or 3 k. L x = 3 x 102 da. L ( 300 da. L) Multiply when converting from larger to smaller units.

Practice: 4) How many nanograms are in 25 decigrams? Determine the possible conversion factors: The two units in this problem are ng and dg. 1 dg = 108 ng (because 10 -1 -(-9) = 10 -1+9 ). or 25 dg x = 2. 5 x 109 ng Multiply when converting from larger to smaller units.

Exercises (show your work) (notebooks): I - Convert: 1) 5. 00 Km = _____ cm. 2) 250 m. L = ______ n. L. 3) 8 kg = _______ Gg. 4) 1, 000 mg = _____ kg.

Exercises (show your work) (notebooks): I - Convert: 5 5. 00 x 10 1) 5. 00 Km = _____ cm. Kilo – to - centi ---- 103 – (-2) = 105 = 5. 00 x 105 cm 1 Km = 105 cm (5, 000 cm)

Exercises (show your work) (notebooks): I - Convert: 8 2 x 10 2) 200 m. L = _____ n. L. milli – to - nano ---- 10 -3 – (-9) = 106 = 2 x 108 n. L 1 m. L = 106 n. L (200, 000 n. L)

Exercises (show your work) (notebooks): I - Convert: -6 8 x 10 3) 8 Kg = _____ Gg. Kilo – to - giga ---- 103 – 9 = 10 -6 = 8 x 10 -6 Gg 1 Kg = 10 -6 Gg (0. 000, 008 Gg)

Exercises (show your work) (notebooks): I - Convert: -3 1 x 10 4) 1, 000 mg = _____ Kg. milli – to - kilo ---- 10 -3 – 3 = 10 -6 = 1 x 10 -3 Kg 1 mg = 10 -6 Kg (0. 001 Kg)

Giga (G) ---- 109 Mega (M) --- 106 Kilo (K) ----- 103 Hecto (h) --- 102 Deca (da) --- 101 BASE UNIT Deci (d) ---- 10 -1 Centi (c) ---- 10 -2 Milli (m) ---- 10 -3 Micro (μ) --- 10 -6 Nano (n) --- 10 -9 Pico (p) ---- 10 -12 Shift the decimal point to the left Shift the decimal point to the right Multiply Divide

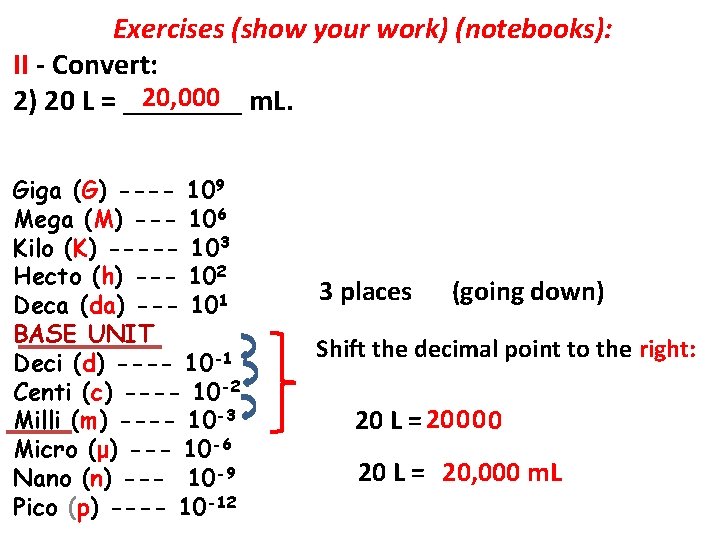
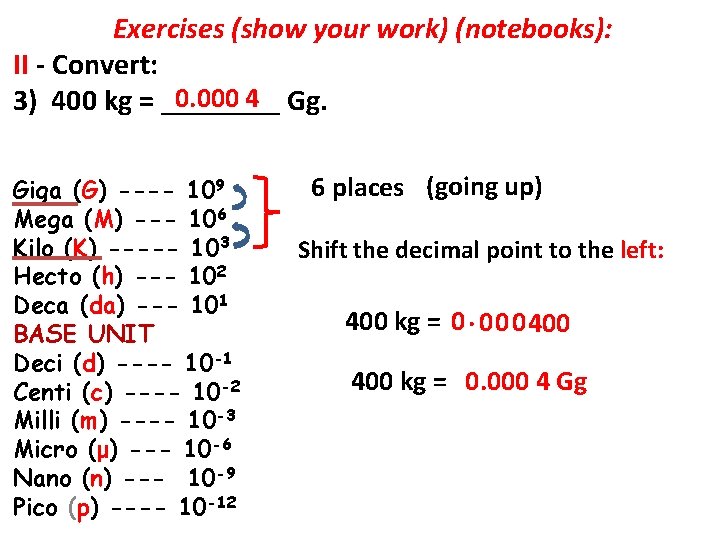
Exercises (show your work) (notebooks): II - Convert: 1) 12 hm = ____ cm. 2) 20 L = _____ m. L. 3) 400 kg = _____ Gg. 4) 1, 000 dg = _______ hg.

Exercises (show your work) (notebooks): II - Convert: 120, 000 cm. 1) 12 hm = ____ Giga (G) ---- 109 Mega (M) --- 106 Kilo (K) ----- 103 Hecto (h) --- 102 Deca (da) --- 101 BASE UNIT Deci (d) ---- 10 -1 Centi (c) ---- 10 -2 Milli (m) ---- 10 -3 Micro (μ) --- 10 -6 Nano (n) --- 10 -9 Pico (p) ---- 10 -12 4 places (going down) Shift the decimal point to the right: 12 hm = 12 0 0 12 hm = 120, 000 cm

Exercises (show your work) (notebooks): II - Convert: 20, 000 m. L. 2) 20 L = ____ Giga (G) ---- 109 Mega (M) --- 106 Kilo (K) ----- 103 Hecto (h) --- 102 Deca (da) --- 101 BASE UNIT Deci (d) ---- 10 -1 Centi (c) ---- 10 -2 Milli (m) ---- 10 -3 Micro (μ) --- 10 -6 Nano (n) --- 10 -9 Pico (p) ---- 10 -12 3 places (going down) Shift the decimal point to the right: 20 L = 20 0 20 L = 20, 000 m. L

Exercises (show your work) (notebooks): II - Convert: 0. 000 4 Gg. 3) 400 kg = ____ Giga (G) ---- 109 Mega (M) --- 106 Kilo (K) ----- 103 Hecto (h) --- 102 Deca (da) --- 101 BASE UNIT Deci (d) ---- 10 -1 Centi (c) ---- 10 -2 Milli (m) ---- 10 -3 Micro (μ) --- 10 -6 Nano (n) --- 10 -9 Pico (p) ---- 10 -12 6 places (going up) Shift the decimal point to the left: 400 kg = 0. 0 0 0 400 kg = 0. 000 4 Gg

Exercises (show your work) (notebooks): II - Convert: 1 4) 1, 000 dg = ____ hg. Giga (G) ---- 109 Mega (M) --- 106 Kilo (K) ----- 103 Hecto (h) --- 102 Deca (da) --- 101 BASE UNIT Deci (d) ---- 10 -1 Centi (c) ---- 10 -2 Milli (m) ---- 10 -3 Micro (μ) --- 10 -6 Nano (n) --- 10 -9 Pico (p) ---- 10 -12 3 places (going up) Shift the decimal point to the left: 1, 000 dg = 1. 000 1, 000 dg = 1 hg

Derived units are “built” from SI units: cm 2, m 2 area m 3 ; cm 3 volume g/cm 3 density Ex. How can you calculate de area of the football field? How can you calculate de volume of this classroom? How can you calculate de density of ?

Converting Units: 1 cm = 101 mm 1 cm 3 = 101 x 3 mm 3 = 103 mm 3 1 cm 2 = 101 x 2 mm 2 = 102 mm 2 (1, 000 mm 3) (100 mm 2) 1 m = 103 mm 1 m 3 = 103 x 3 mm 3 = 109 mm 3 (1, 000, 000 mm 2)

Exercises (show your work) (notebooks): III - Convert: 1) 12 cm 2 = ____ mm 2 2) 15 m 2 = _____ cm 2 3) 10 dm 3 = ____ cm 3 4) 1 m 3 = _____ mm 3

Arithmetic with units: In addition or subtraction the units should be the same: 2 kg + 3 kg = 5 kg 412 m - 12 m = 400 m So, units must be the same before adding or subtracting! 3. 001 kg + 112 g = 3. 001 kg + 0. 112 kg = 3. 113 kg 4. 314 Gm - 2 Mm = 4. 314 Gm - 0. 002 Gm = 4. 312 Gm

Arithmetic with units: In multiplication or division: the units must be multiplied or divided too: 3 m × 3 m = 9 m 2 10 kg x 9. 8 m/s 2 = 98 kg m/s 2 Sometime, units may cancel: 5 g / 10 g = 0. 5 (no units) 10. 00 m/s x 39. 37 in/m = 393. 7 in/s

Exercises (show your work) (notebooks): IV - Solve: 1) 12 cm 2 + 2. 7 cm 2 = 2) 2 m – 10 cm = 3) 2 cm x 3 cm x 6 cm = 3) 25 kg ÷ 5 kg = 4) 45 km ÷ 0. 5 h =

Temperature in SI units: The difference between the two scales (Celsius, °C & Kelvin, K) is their zero point: - The zero point in Celsius is the freezing point of water. - The zero point on the Kelvin scale is absolute zero. 00 K = - 273°C. (Absolute zero (zero K) is the coldest temperature theoretically possible). You can convert between Kelvin and Celsius by using either of the following equations: K = o. C + 273 o. C = K - 273

Temperature in SI units: Example: Express 25°C in kelvins. K = o. C + 273 K = 25 + 273 = 298 K Temperature in Fahrenheit: F = 1. 8 x o. C + 32 o. C =

Exercises (show your work) (notebooks): IV - Convert: 1) 25 o. C = _____ K. 2) 273 K = _____ o. C 3) 25 o. C = _____ K. 3) 300 K = _____ o. C