Convection Density of Fluids Investigation 5 Part 1



























- Slides: 31

Convection Density of Fluids Investigation 5, Part 1

Setting the Scene • Observe the bottles. • What is happening? • Can you explain why the liquids behave the way they do?

Challenge of the day • There are four liquids • Layer all four liquids in a section of straw

Materials Container with 4 liquids Straw & holder Pipettes (4) Paper Towels

Using the straw • Push the angled edge of the straw over the ball of holder so it sits at a slight angle.

Using the straw • Gently squeeze a few drops of each liquid into the straw

Using the straw • Gently squeeze a few drops of each liquid into the straw Let the drops slide down the side of the straw

Using the straw • Gently squeeze a few drops of each liquid into the straw – Let the drops slide down the side of the straw

Using the straw • Gently squeeze a few drops of each liquid into the straw – Let the drops slide down the side of the straw

Directions • Record all trials using colored pencils on the “Liquid Layers” worksheet • Circle the correct layering diagram • Answer the question for Part 2

Discussion • What do you think caused the salt solutions to layer this way?

Discussion • The liquids are made with water and salt • How is it that some salt solutions can be heavy and some light if they are made with the same materials? Breakpoint…

Bottles • Observe the bottles – How are these 2 bottles like the straw challenge? – How are they different?

Ponder this! Which is heavier? Gold or Feathers

Weight and volume • Cup of beans and the cup of foam cubes • Which do you think is heavier? • Think about this: – Cups are the same size and full – How can one be heavier than the other?

Mass vs Volume Mass • The amount of stuff in an object • You find mass by measuring the weight Volume • Is how much space a sample of matter occupies • Measured using graduated cylinders

Density • The amount of matter in a volume of material • Density = mass / volume • Ratio between the mass of matter and its volume • Equal volumes of material – One is heavier than the other – Heavier material is denser

Consider salt solution • Do you think density played a role in the layering activity?

Determining Density • Will these 2 identical volumes of liquids have the same mass? • Make a prediction!

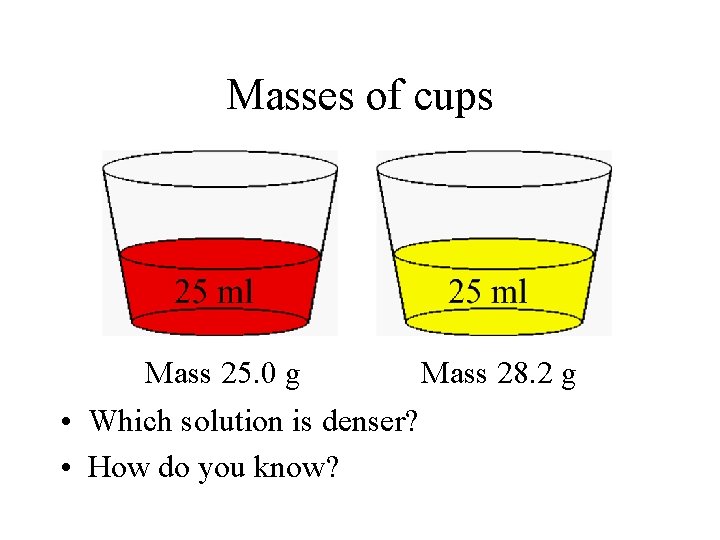
Masses of cups Mass 25. 0 g Mass 28. 2 g • Which solution is denser? • How do you know?

Discuss with group • Which solution would layer on top of the other? • What does this tell you about how liquids of different densities interact? (Which one will be on top? )

Masses and Volumes • On the “Liquid Layers” • Write the masses and volumes Red Green Yellow Blue Mass 25. 0 g 26. 5 g 28. 2 g 30. 0 g Volume 25 ml

Densities of colored salt solutions • Turn to “Calculating Density” • Transfer data from”Liquid Layers” page • Calculate the density (D = M / V) and answer the questions at the bottom

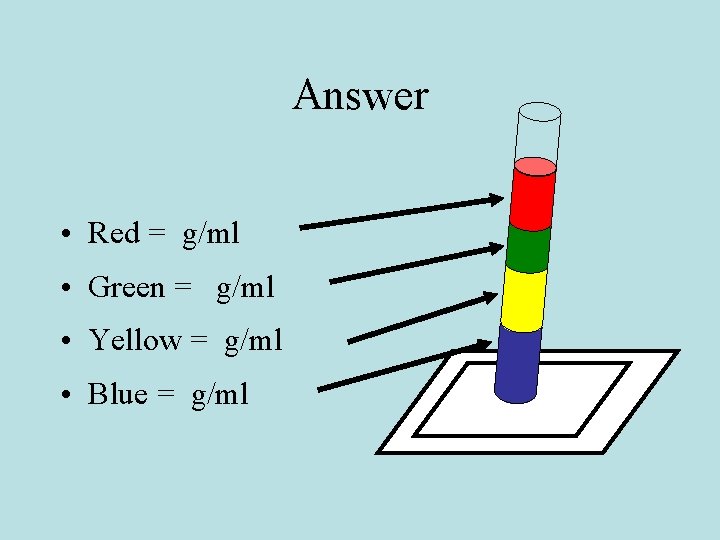
Answer • Red = g/ml • Green = g/ml • Yellow = g/ml • Blue = g/ml

• Density of water is 1. 0 g/ml • Pine wood has a density of 0. 60 g/cc. Will it float or sink in water? • Styrene plastic has a density of 1. 7 g/cc. Will it float or sink in the red saltwater? • What general statement can you make about floating and sinking in water?

Revisiting density bottles • Observe the bottles again • Explain those observations in terms of density

Revisiting density bottles The red liquid is less dense than the clear liquid The clear liquid is denser than the red liquid The clear liquid is less dense than the red liquid The red liquid is denser than the clear liquid

Review: Density • Density is a physical property of matter. • The density of any object can be calculated if you know its volume and its mass • Volume can be measured in milliliters (ml) or cubic centimeters (cc). The unit most often used is cubic centimeters, but milliliters can be used with fluids. 1 ml=1 cc • Mass can be determined by weighing. • Density is mass per unit volume. • Always include the units with your calculations

Density Problem • I have two samples of salt water. Sample 1 is 45 ml, and its mass is 54 g. Sample 2 is 120 ml and its mass is 132 g. Which sample is denser?

Density Practice • Complete : -Density Identities -What is Density? -How is Density Measured?

Reading • Read “Density” on page 27 of the Green Resource Book • Answer questions for homework