Controls and containment in plantbased vaccine manufacturing Todd

Controls and containment in plant-based vaccine manufacturing Todd Talarico Vice President of Process Development and Clinical Manufacturing

Objective § Introduce Plant Based Biologics § Present Controls used for Manufacturing based upon: – Health Canada : Guidance Document – Submission Information for Human-Use Biological Drugs Derived from Plant Molecular Farming (PMF) – FDA: Draft Guidance for Industry: Drugs, Biologics and Medical Devices Derived from Bioengineered Plants for Use in Humans and Animals – ICH Q 7 - Good Manufacturing Practice Guide for Active Pharmaceutical Ingredients – ICH Q 5 series – Quality of Biotechnological Products – EMEA – Guideline on the Quality of Biological Active Substances Produced by Stable Transgene Expression in Higher Plants • Guideline on Good Agricultural and Collection Practice (GACP) § Focus on the process first and then containment – Familiar Principles – Principles that may be more unique to plant derived biotherapeutics and vaccines 2 © Medicago Inc. All rights reserved 2

Medicago Overview © Medicago Inc. All rights reserved Focus Vaccines & Therapeutic Proteins Manufacturing technology Transient expression in plants Vaccine technology Virus-like particles Employees ~300 Expected to grow to 500 by 2019 (commercial) Success story Innovative Vaccine Technology Potential advantages over current vaccine technologies (efficacy, cross protection) Pandemic Supply advantages Headquarters, laboratories & c. GMP facilities Quebec City, CANADA Research Triangle Park, NC, USA Corporate structure Private company Mitsubishi Tanabe Pharma Corporation (majority shareholder), PMI 3 3

Plant Platforms for the Production of Biologics • Plant cells (bioreactor) • Whole plants • Expression systems • Transgenic plants with nuclear transformation • Transient expression • Induced by plant specific bacterium or virus polynucleotide transfer • Advantages of transient expression system » No propagation through seed stock » Genetic stability of expression • Growth systems • Indoors (Greenhouses, Growth Chambers) • Outdoors with restrictions • Expression • Indoors (Greenhouses, Chambers) or Outdoors © Medicago Inc. All rights reserved 4 4

Medicago’s Technology • Plants used for production are not genetically modified and grown in a greenhouse • Use a genetically engineered Agrobacterium to drive expression of a protein in the leaves of “tobacco” plants • In the case of the Influenza vaccine, the plants produce virus like particles (VLP) containing the HA protein imbedded in a lipid bilayer when incubated under controlled conditions • The VLP are extracted from the plant tissue • The VLP are purified in a “traditional” bioprocessing scheme to produce a sterile product, suitable for injection Medicago’s Bioreactor © Medicago Inc. All rights reserved 5 5 5

Key Aspects of Medicago’s Process Control © Medicago Inc. All rights reserved • Stock maintenance – Plant seed stock maintenance system – Agrobacterium MCB/WCB maintenance • Upstream • Greenhouse • Stock to harvest • Before infiltration – Defined biological starting material • After infiltration (protein expression) • Downstream – After extraction – Purification, Formulation and Filling § Facility Control § Equipment Control § Personnel Control § Raw Material Control 6 6
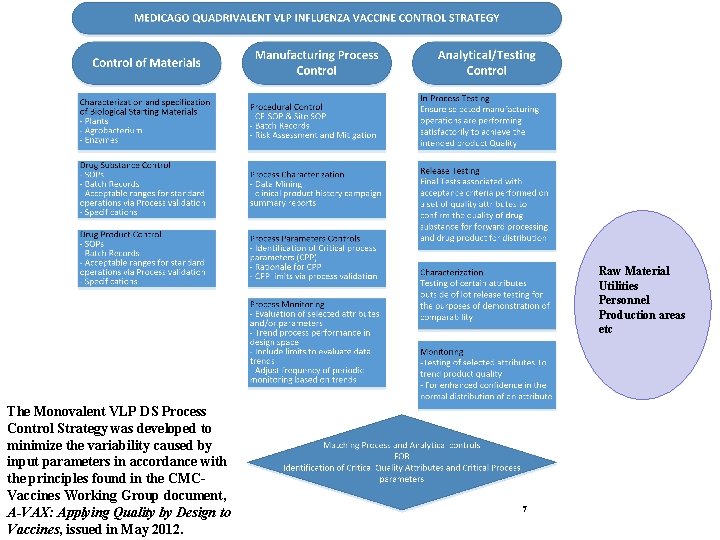
Raw Material Utilities Personnel Production areas etc The Monovalent VLP DS Process Control Strategy was developed to minimize the variability caused by input parameters in accordance with the principles found in the CMCVaccines Working Group document, A-VAX: Applying Quality by Design to Vaccines, issued in May 2012. 7

Guidance Documents that support the plant platform § Health Canada : Guidance Document – Plant Molecular Farming (PMF) Applications: Plant-Derived Biological Drugs for Human Use (2014) § FDA: Draft Guidance for Industry: Drugs, Biologics and Medical Devices Derived from Bioengineered Plants for Use in Humans and Animals (2002) § ICH Q 7 - Good Manufacturing Practice Guide for Active Pharmaceutical Ingredients § ICH Q 5 series – Quality of Biotechnological Products § EMEA – Guideline on the Quality of Biological Active Substances Produced by Stable Transgene Expression in Higher Plants (2008) – Guideline on Good Agricultural and Collection Practice (GACP) © Medicago Inc. All rights reserved 8

Quality Requirements for Plant Derived Biologics § Meet ‘the same quality standards as drugs produced by conventional platforms’ § ‘PMF-derived drugs are subject to the same general submission requirements…as other biological drugs’ § Address ‘risks unique to PMF production platforms, particularly in the pre-harvest and harvest stages …’ – Concern about control of plants in a field 9 © Medicago Inc. All rights reserved 9
10

Stock Maintenance § All starting material must be defined – Origin and maintenance § Agrobacterium stock – Managed through the use of Master and Working Cell Banks – Based on principles defined in ICHQ 5 § Plant seed stock maintenance – Nicotiana benthamiana • Self germinating - homogeneity • Propogated during winter in Quebec (to prevent foreign pollen introduction) – Parenteral Seed Stock • Regular (5 year) regeneration with cumulative stability being monitored – Annual Seed Stock • Produced annually from parental stock • Aliquoted and stored in independent locations 11 © Medicago Inc. All rights reserved 11

Quality System (‘GMP-like’ controls) – to produce a welldefined biological starting material – – – Staff qualification and training Written procedures Incoming material specification Batch records Biological material specifications for plants and harvested material Trending, including monitoring of production consistency, environment • Characterization such as fiber content, Mycotoxins, Mold + yeast, plant virus, bacterial flora, substrate p. H, substrate conductivity – Identifying key control parameters • Product safety first • Parameters that have an impact on product attributes and yield – Other traditional oversight - Risk Assessment, Audits, Change Control – Equipment controls 12 © Medicago Inc. All rights reserved 12

Greenhouse (Upstream) § Controlled process that results in a defined biological starting material § The greenhouse environment is designated as a “controlled, not classified” environment § Quality system with QA and QC components – ‘GMP-like’ principles – Not all pharmaceutical GMP regulations can apply 13 © Medicago Inc. All rights reserved 13
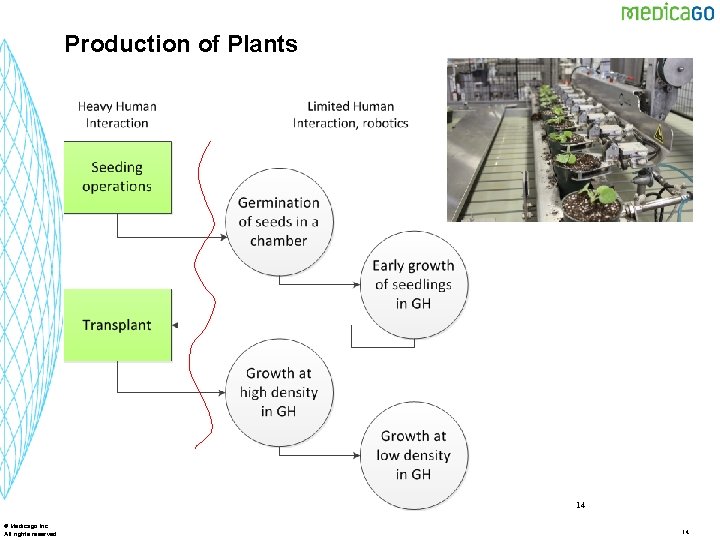
Production of Plants 14 © Medicago Inc. All rights reserved 14

Medicago’s Controls on Plants § All operations covered by production batch records for generation of the critical raw material § No herbicides, pesticides or fungicides § Specifications for input raw materials – Peat, fertilizer, water § Control of entry – Personnel, filtered air (not HEPA), insect monitoring, rodent exclusion and monitoring § Criteria for acceptance as plants proceed from seeding to infiltration § Routine checks for plant viruses and organisms that could produce mycotoxins § No documented cases of human viruses infecting and replicating in plants or plant viruses replicating in humans 15 © Medicago Inc. All rights reserved 15

Plant Control § Controls are in place to assure plants provide proper protein expression § The “working operating space” for many variables has been demonstrated to be large which makes for a robust production system – 20 to 50 gm plants have been used to produce the influenza vaccine with no negative impact to yield or CQA § Other variables have more impact on protein expression – Flower stage is important to control § The plant has been mapped to understand which tissues produce the most product § Kinetics of expression are determined for each strain © Medicago Inc. All rights reserved 16
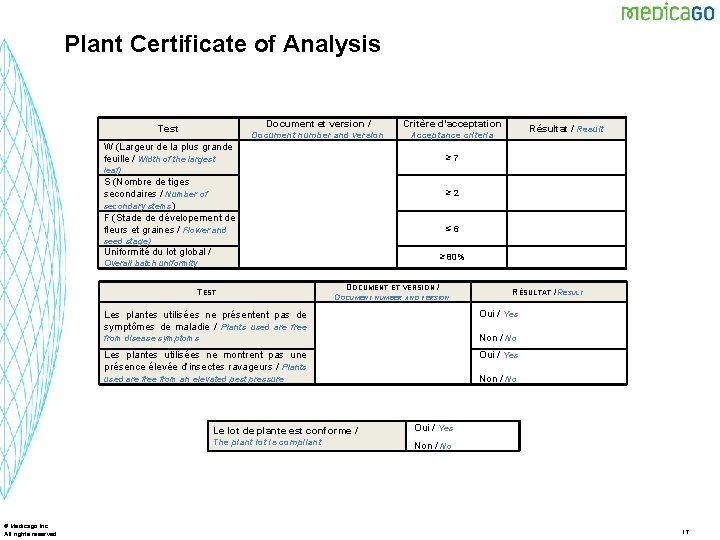
Plant Certificate of Analysis Test Document et version / Critère d’acceptation Document number and version Acceptance criteria W (Largeur de la plus grande feuille / Width of the largest Résultat / Result ≥ 7 leaf) S (Nombre de tiges secondaires / Number of secondary stems) F (Stade de dévelopement de fleurs et graines / Flower and ≥ 2 ≤ 6 seed stage) Uniformité du lot global / ≥ 80% Overall batch uniformity TEST © Medicago Inc. All rights reserved DOCUMENT ET VERSION / DOCUMENT NUMBER AND VERSION RÉSULTAT / RESULT Les plantes utilisées ne présentent pas de symptômes de maladie / Plants used are free Oui / Yes from disease symptoms Non / No Les plantes utilisées ne montrent pas une présence élevée d’insectes ravageurs / Plants Oui / Yes used are free from an elevated pest pressure Non / No Le lot de plante est conforme / Oui / Yes The plant lot is compliant Non / No 17

Downstream § After extraction – purification is similar to other biological products § Plant specific concerns – real or perceived – Impurities from plant manufacturing process • Fibres, lipids, host cell proteins • Plant secondary metabolites – Risk assessment • Consider as well the potential allergenic potential • Define their nature • Reduce to safe levels – Plant glycans (part of the glycoproteins) • Potential diffierences in glycan structure • Analysis and risk assessment – No evidence they are immunogenic – Could be beneficial to process compared with mammalian cell glycans • Product and use specific (vaccine vs long term therapeutic) 18 © Medicago Inc. All rights reserved 18

Process Validation (PPQ) § Master validation plan outlines methods § Data mining performed on 3 production process generations § Data from > 20 than runs used to set CPP ranges based on CQA of vaccine § Secondary dataset for current process compared back to original data mine § These data will be used to set the acceptable ranges for the consistency lots used for Phase 3 manufacture § Agreement on proposal to the Agency for consistency lots § Obtained buy in for number of lots per strain and number of runs to generate drug substance § Three fills planned with CMO to validate filling process § Matrixed based on number of units filled © Medicago Inc. All rights reserved 19

CPPs for the Production Process § 5 CPP around filtration operations § 3 CPP around chromatography § 1 CPP for virus inactivation § Quality mandated CPP for filtration (2) and virus inactivation (1) – for example – Sterility § Moving forward some CPP may be eliminated by incorporating PAT or reducing risk © Medicago Inc. All rights reserved 20

Containment – Material and Process § Clarification and downstream operations are similar to traditional bioprocessing – Use of disposables – Where stainless steel is used, cleaning validation is traditional • Look for plant based compounds (nicotine, chlorophyll, phenolics) – Closed systems as we approach drug substance • Single use bag technology – VLPs pass through a 0. 2 µ filter – Facility design utilizes typical classifications § Greenhouse and VLP (or protein) extraction are the areas that are dissimilar to traditional bioreactor based biopharmaceutical operations 21 © Medicago Inc. All rights reserved 21

Process Challenges from a “Contaminant” Perspective § A greenhouse is not an aseptic environment § Efficient operations require recycling of materials – Water, fertilizer § Biological raw materials – No animal derived components – Many not be “low bioburden or sterile” § Agrobacterium is Gram negative – Endotoxin (but of low pyrogenicity) § Human interaction in an open system with critical raw materials – Plants § A VLP is much like a virus from a surface biochemical point of view – Hard to get a process to have high levels of viral removal/inactivation 22 © Medicago Inc. All rights reserved 22
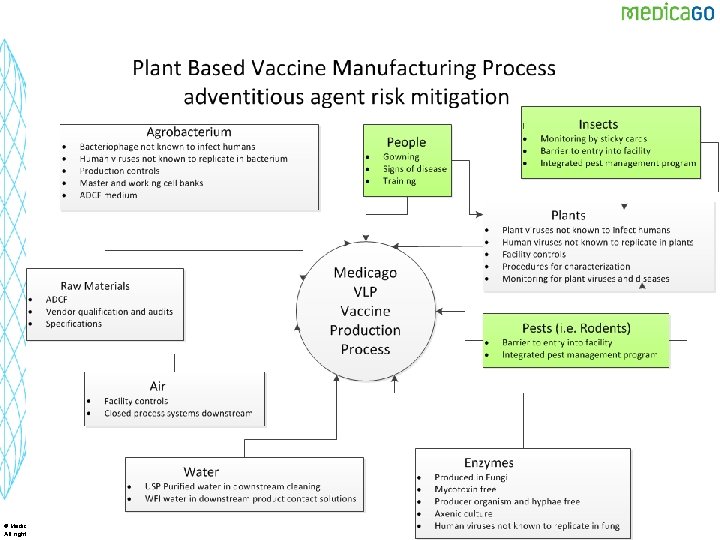
23 © Medicago Inc. All rights reserved 23

Bioburden Clearance in the Process § Centrifugation and microfiltration early in the process reduce bioburden § Sterilizing grade filter removes Agrobacterium early in the process and reduces all bioburden prior to process intermediate entrance into downstream operations § Additional microfiltration protects chromatography columns and reduces any bioburden load § Sterile filtration prior to fill of bulk drug substance and again prior to fill to syringes or vials © Medicago Inc. All rights reserved 24

Viral Risk § Plants are considered as a low risk for introduction of adventitious agents – Human viruses have not been shown to replicate in plants – Plant viruses are not known to infect human cells – Plants act as a barrier to virus introduction into the process § No animal derived components used in the process § Largest risk is introduction of an adventitious agent into the process by human, rodent or insect. § Regulators worry about new threats emerging based upon past experience in biological processes § Plant based production system presents a low risk of AA introduction when the proper controls are in place 25 © Medicago Inc. All rights reserved 25

Viral Clearance § Many negotiations with the Agency to move from safety testing to inactivation § Inclusion of a UVC treatment unit operation in the process § Development work performed to identify the optimal dose to inactivate a variety of viruses with minimal impact to the product § Added bonus of spiroplasma and mycoplasma inactivation © Medicago Inc. All rights reserved 26
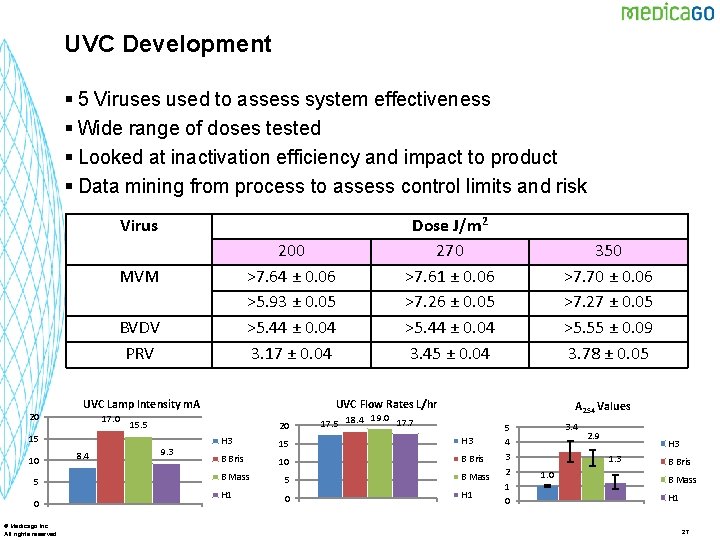
UVC Development § 5 Viruses used to assess system effectiveness § Wide range of doses tested § Looked at inactivation efficiency and impact to product § Data mining from process to assess control limits and risk Virus 200 >7. 64 ± 0. 06 >5. 93 ± 0. 05 >5. 44 ± 0. 04 3. 17 ± 0. 04 MVM BVDV PRV 20 UVC Lamp Intensity m. A 17. 0 10 5 0 © Medicago Inc. All rights reserved 8. 4 20 9. 3 350 >7. 70 ± 0. 06 >7. 27 ± 0. 05 >5. 55 ± 0. 09 3. 78 ± 0. 05 UVC Flow Rates L/hr 15. 5 15 Dose J/m 2 270 >7. 61 ± 0. 06 >7. 26 ± 0. 05 >5. 44 ± 0. 04 3. 45 ± 0. 04 A 254 Values 19. 0 17. 7 17. 5 18. 4 H 3 15 H 3 B Bris 10 B Bris B Mass 5 B Mass H 1 0 H 1 5 4 3 2 1 0 3. 4 2. 9 H 3 1. 0 B Bris B Mass H 1 27

UVC Viral Inactivation § Process validation completed – – Scaled down model equivalent to manufacturing process Rationale documented to support chosen studies (Risk based) Used the development data to set boundries for validation study Duplicate runs with three viruses using two HA types at a single UVC dose – Robustness studies to assess processing and equipment extremes at chosen UVC dose – Re-use of flow path § Regulatory – Negotiation for required steps and clearance levels – Required parallel AA testing for a period to establish safety – Interested in product impact-immunogenicity for each strain © Medicago Inc. All rights reserved 28

Validation: Normal Operating Conditions • MMV spike • B Phuket and H 1 California strains • Conclusion: Clearance equivalent to previous experimental results, complete inactivation © Medicago Inc. All rights reserved Run 1 H 1 California Log Clearance >7. 88 ± 0. 07 Run 2 H 1 California Run 1 B Phuket Run 2 B Phuket >7. 86± 0. 05 >7. 74 ± 0. 05 >7. 76± 0. 07 29 29

Summary § We at Medicago have developed our strategy for containment and control based upon Canadian, ICH and FDA guidances to the industry § Quality Systems and GMP principles cover the entire process including the plant ‘biological starting material’ § The use of plants as “bioreactors” results in containment that differs from traditional biotech operations § Downstream operations are similar to those used in other biotech processes and result in efficient and timely removal of bacterial microorganisms and host cells § Process validation and QBD principles apply to processes producing biotherapeutics and vaccines from plants as they do to other substrates § While plants may be inherently safe with regard to expansion of human pathogens, the regulatory agencies have concerns with raw materials and vectors that may introduce adventitious agents into plant derived products 30 © Medicago Inc. All rights reserved 30
- Slides: 30