Controlling Gene Expression SBI 4 U Biology Gene

Controlling Gene Expression SBI 4 U Biology

Gene Expression • Follows the Central Dogma: DNA RNA Protein • Most eukaryotes are diploid; they have two copies of every gene in each cell. • So, if that’s the case, why aren’t skin cells making blood proteins? Or brain cells making digestive enzymes? • It’s a question of regulatory control: not every gene gets transcribed…
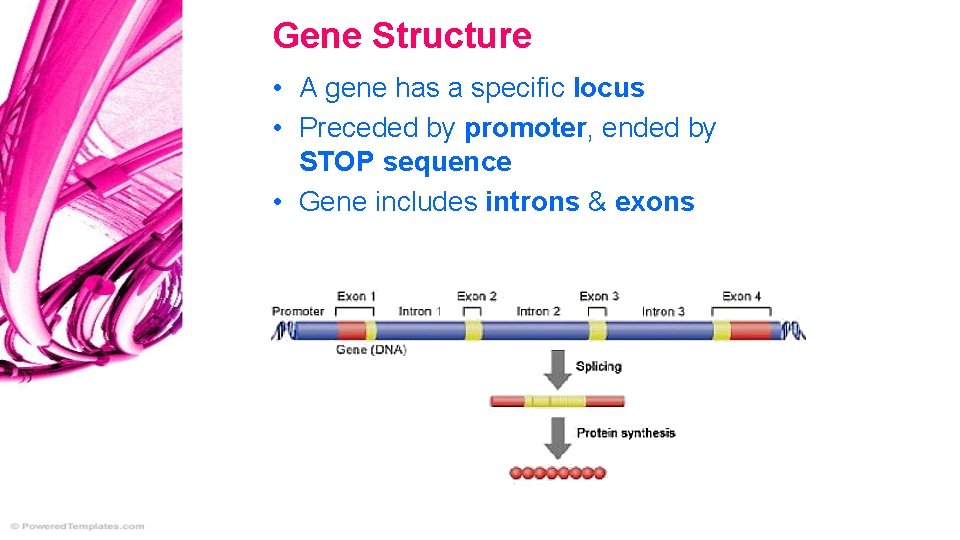
Gene Structure • A gene has a specific locus • Preceded by promoter, ended by STOP sequence • Gene includes introns & exons

Levels of Control • Four levels: transcriptional, posttranscriptional, transport, translational.

Regulators, Operators, Promoters • RNA polymerase binds at a Promoter • Often have a Regulator gene, influencing binding • The regulator gene’s product binds at an Operator sequence, blocks promoter.
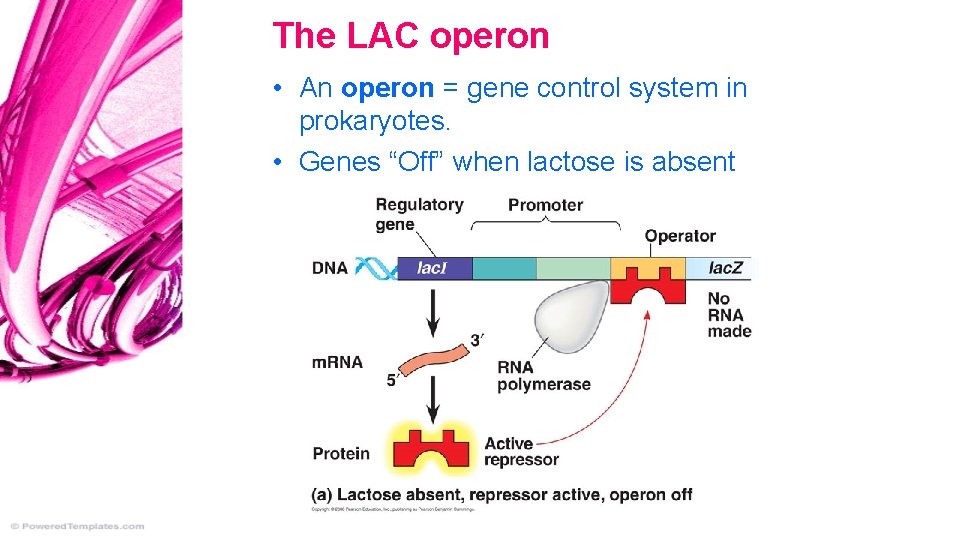
The LAC operon • An operon = gene control system in prokaryotes. • Genes “Off” when lactose is absent
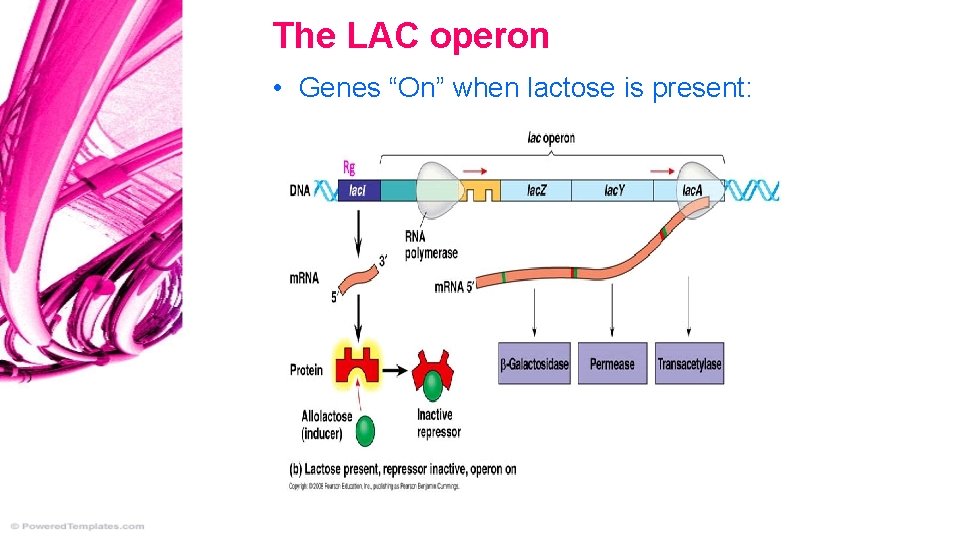
The LAC operon • Genes “On” when lactose is present:

Transcriptional Control • In nucleus • What gets transcribed into m. RNA, and what doesn’t • Some genes are inactivated & never transcribed; ‘junk’ DNA (SINES, LINES) Post-Transcriptional Control • In nucleus • Editing of Introns by sn. RNA spliceosomes • Some m. RNA’s are degraded at this point!

Transport Control • • In nucleus & cytoplasm What goes into the cyto, what doesn’t Non-shuttling proteins block transport Some products stay in nucleus, i. e. histones, r. RNA, RNA polymerase… Translational Control • In cytoplasm; Golgi, ER, free ribosome… • Inhibitor molecules bind to some m. RNA, inactivating it; no translation. • Often by a feedback mechanism, i. e. ferritin protein = only when Fe 3+ high
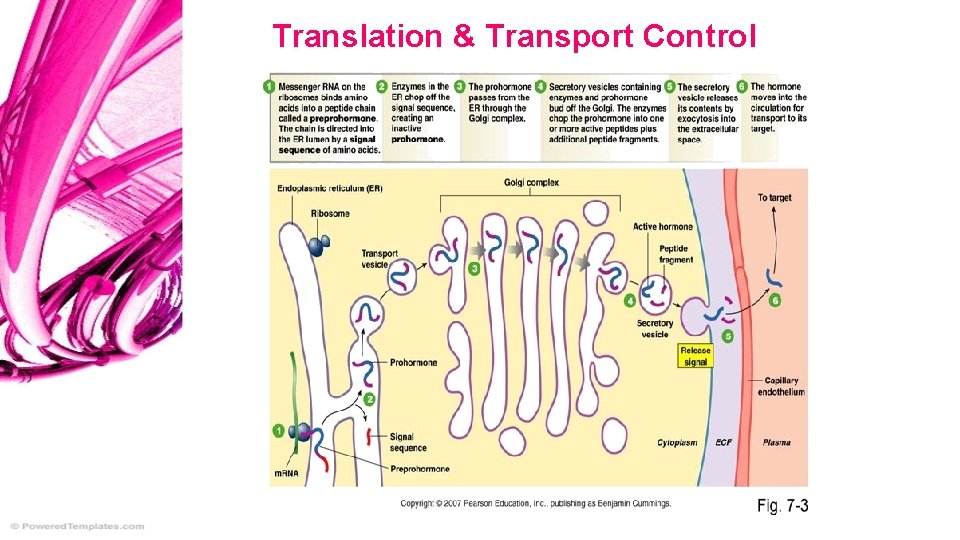
Translation & Transport Control

Post-Translational Modification • The translated polypeptide may need some ‘tweaking’ to make it functional: • Enzymatic activation: i. e. insulin is made as a single chain, which is cleaved into 2, held together by disulphide bonds • Environmental factors: i. e. , pepsinogen is converted to pepsin by HCl. • Structural modification: creating the 2 o, 3 o or 4 o structures of proteins; i. e. hemoglobin = 4 chains, held together by Fe 3+ • Glycosylation: Adding sugars to protein; i. e. , glycoproteins aid in cell identity. • Methylation: activation by adding CH 3
- Slides: 11