Control of Analytical Variables Dr Roula Hamid MSc

Control of Analytical Variables Dr. Roula Hamid MSc Clin Biochem Central Puplic Health Laboratory QC Chemistry

Today is not the golden age of quality in healthcare laboratories. We can & should be doing the better. James O Westgard 2003

Introduction “Nice to Know”
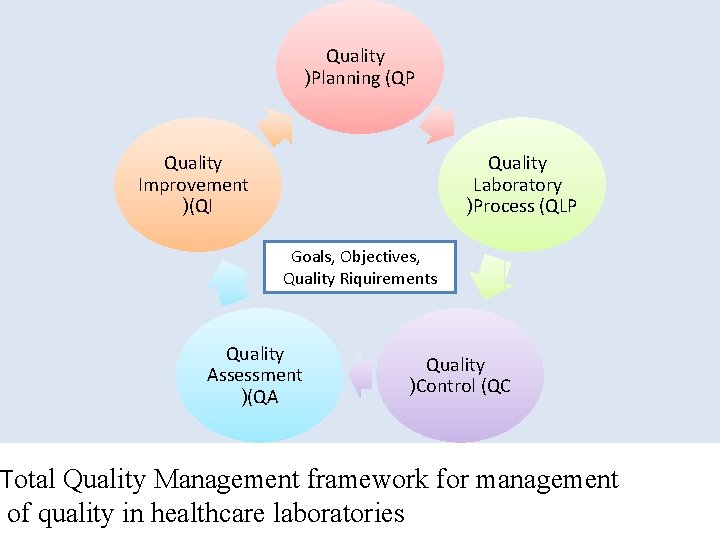
Quality )Planning (QP Quality Improvement )(QI Quality Laboratory )Process (QLP Goals, Objectives, Quality Riquirements Quality Assessment )(QA Quality )Control (QC Total Quality Management framework for management of quality in healthcare laboratories
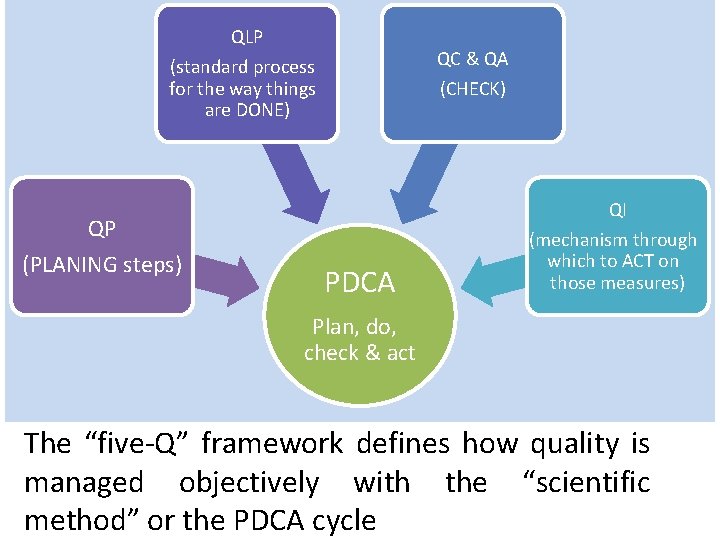
QLP QC & QA (standard process for the way things are DONE) (CHECK) QI QP (PLANING steps) PDCA (mechanism through which to ACT on those measures) Plan, do, check & act The “five-Q” framework defines how quality is managed objectively with the “scientific method” or the PDCA cycle

Control of Analytical Variables

Analytical Variables must be controlled carefully to ensure accurate measurements by analytical methods

v Documentation of analytical protocols v Monitoring technical competency v Statistical control of analytical methods v EQA v New quality initiatives

Documentation of Analytical Protocols v CLSI defines a process as a set of interrelated or interacting activities that transform input into output.

CLSI document describes the following section to be included in a laboratory procedure : A. Title: clear & concise B. Purpose or principle: e. g. this process describes how …. , C. Procedure instructions: how to do D. Related documents: listing of other procedure E. References: source of information F. Appendixes or attachments G. Auther(s): author(s) of document H. Approved signatures

Monitoring Technical Competency v Proper training of laboratory personnel to establish uniformity in technique is important.

Statistical control of analytical methods v Control materials v General principles of control chart v Performance characteristics of a control procedures v Westgard multirule chart v Identifying sources of analytical errors

Control materials § Specimens that are analyzed for QC purposes are known as control materials § They need to be available 1) In a stable form 2) In vial or aliquots 3) & for analysis over an extended period of time

General principles of control chart v A common method used to compare the values observed for control materials with their known values is the use of control charts
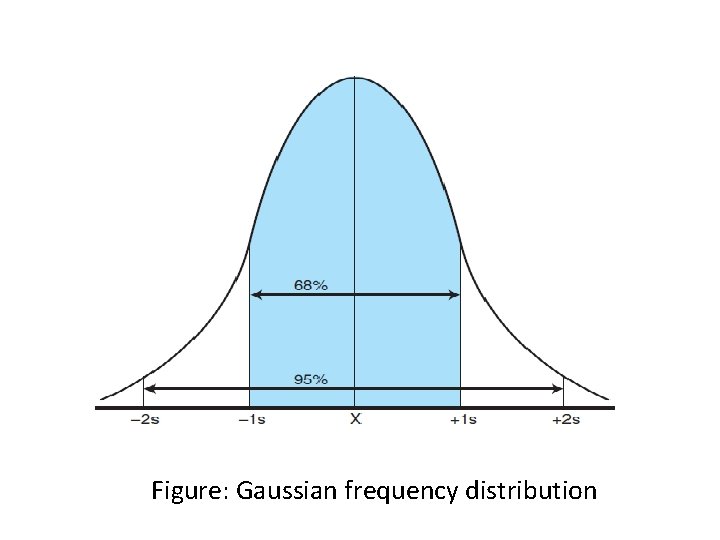
Figure: Gaussian frequency distribution
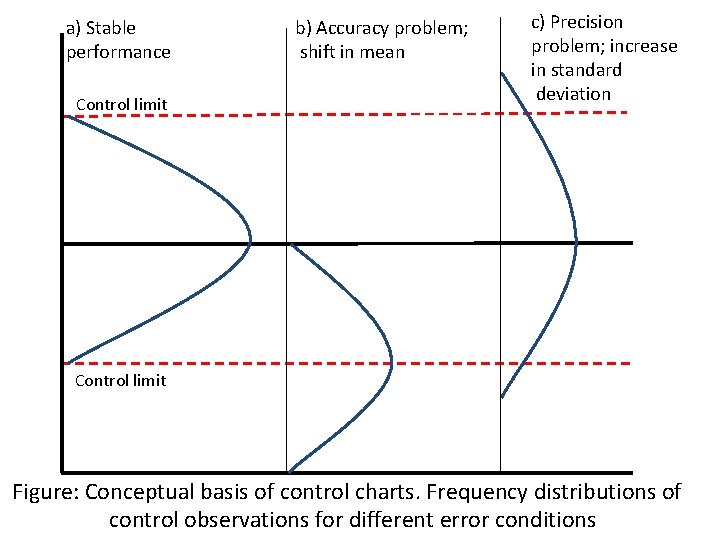
a) Stable performance Control limit b) Accuracy problem; shift in mean c) Precision problem; increase in standard deviation Control limit Figure: Conceptual basis of control charts. Frequency distributions of control observations for different error conditions
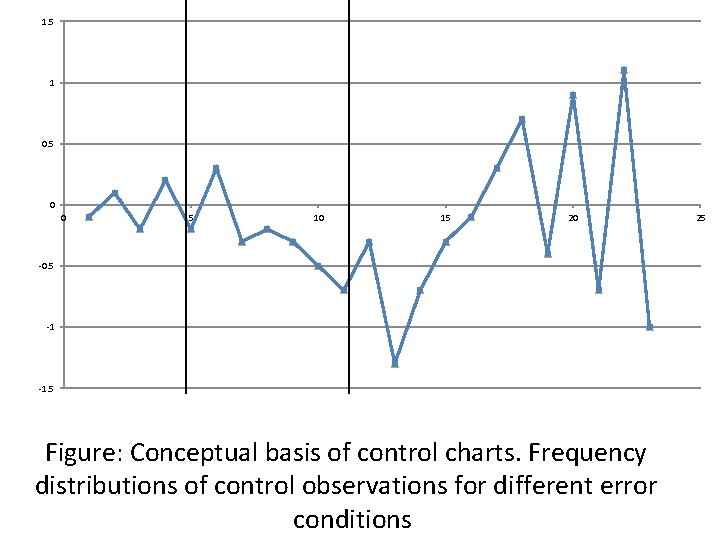
1. 5 1 0. 5 0 0 5 10 15 20 -0. 5 -1 -1. 5 Figure: Conceptual basis of control charts. Frequency distributions of control observations for different error conditions 25

Performance characteristics of a control procedures The knowledge of performance characteristics of control procedures is necessory to select the control rules that detect relevent laboratory problems without causing too many false alarms

Westgard multirule chart v. The probability of false rejection is kept low through selection of only those rules with low v. The probabilities for error detection is improved through selection of those rules that are particularly sensitive to random & systemic errors. v. The use of multirule procedure is similar to the use of a Levey-Jennings chart, but the data interpretation is more structured.
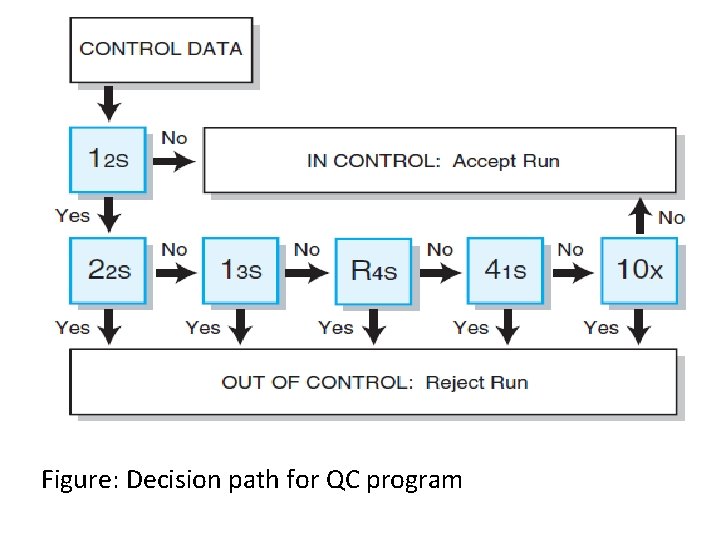
Figure: Decision path for QC program
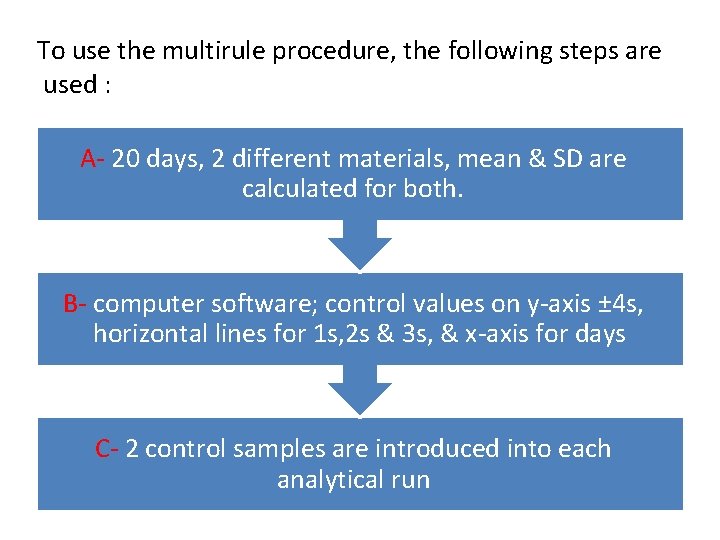
To use the multirule procedure, the following steps are used : A- 20 days, 2 different materials, mean & SD are calculated for both. B- computer software; control values on y-axis ± 4 s, horizontal lines for 1 s, 2 s & 3 s, & x-axis for days C- 2 control samples are introduced into each analytical run
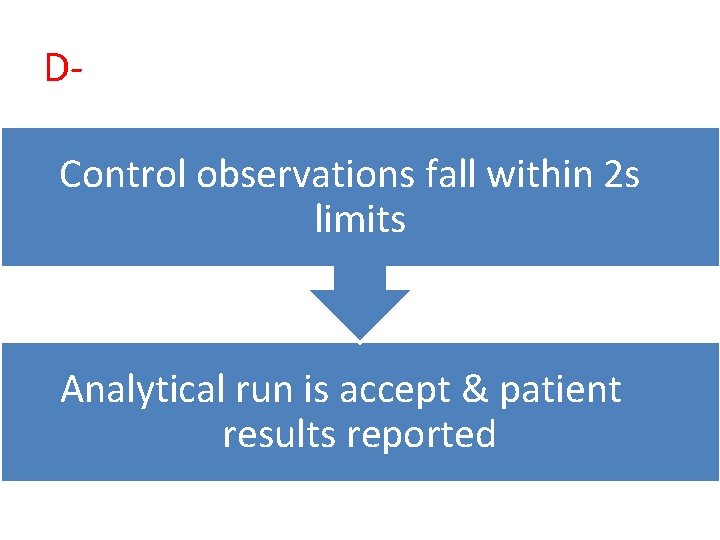
DControl observations fall within 2 s limits Analytical run is accept & patient results reported
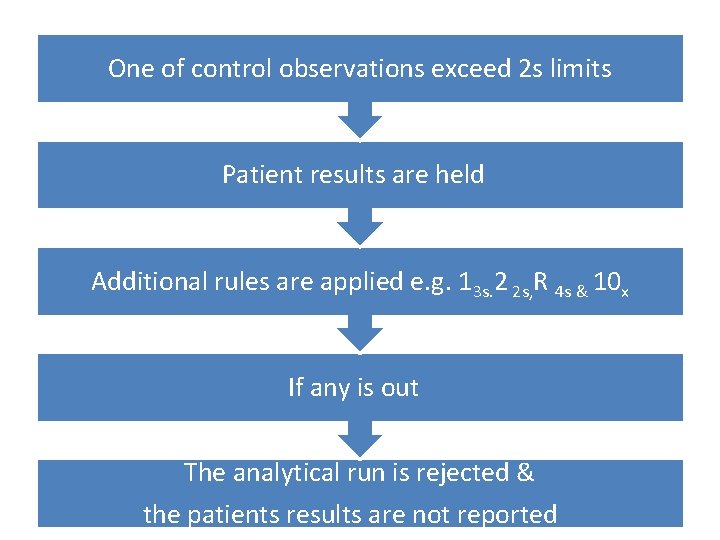
One of control observations exceed 2 s limits Patient results are held Additional rules are applied e. g. 13 s. 2 2 s, R 4 s & 10 x If any is out The analytical run is rejected & the patients results are not reported

EThe type of error is determined based on the control rule that have been violated Looking for the source of that error The problem is corrected The analysis of the entire run repeated including both control & patient samples
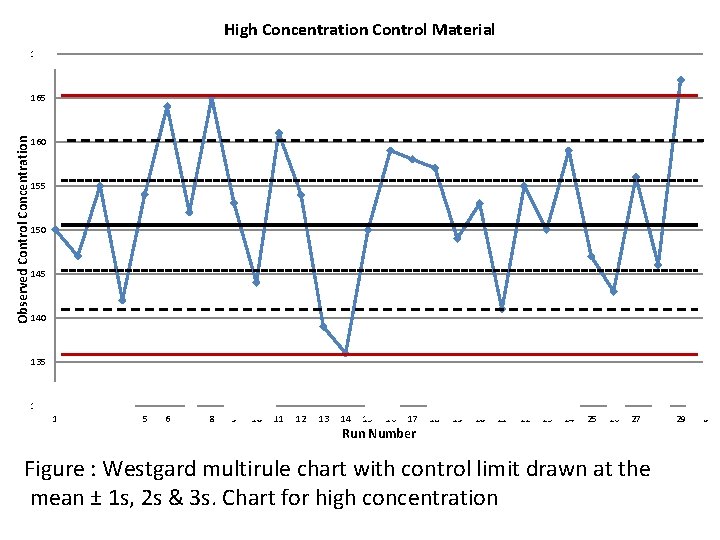
High Concentration Control Material 170 Observed Control Concentration 165 160 155 150 145 140 135 130 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 Run Number 18 19 20 21 22 23 24 25 26 27 Figure : Westgard multirule chart with control limit drawn at the mean ± 1 s, 2 s & 3 s. Chart for high concentration 28 29 30
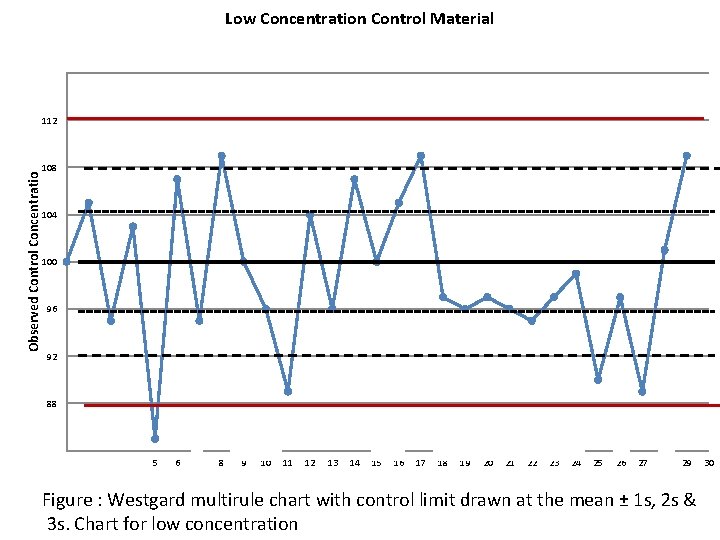
Low Concentration Control Material 116 Observed Control Concentratio 112 108 104 100 96 92 88 84 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 25 26 27 28 29 Figure : Westgard multirule chart with control limit drawn at the mean ± 1 s, 2 s & 3 s. Chart for low concentration 30

Identifying sources of analytical errors Alerted to a control problem Inspection OR Checklist Analytical methods Equipments Reagents Specimens ERRORS Systemic Random

Systemic Errors Random Errors Impure calibration materials Improper preparation of calibrating solution Erroneous set points & assigned values Unstable calibrating solutions Lack of reproducibility in the pipetting of samples & reagents Dissolving of reagents tablets & mixing of sample & reagents Lack of stability of temperature baths, timing regulation, & photometric & other sensors Contaminated solutions Inadequate calibration technique Nonlinear or unstable calibration function Inadequate sample blank Unstable reagent blanks

EQA Procedures used to compare the performance of different laboratories (EQA)

IQC & EQA are complementary IQC For daily • monitoring of accuracy For daily • monitoring of precision EQA Maintenance of • long term accuracy of the analytical methods

Features of External Quality Assessment Programs § EQA program available to the clinical laboratories by professional societies & manufactures of control materials § All the participating laboratories analyzing the same lot of control material § Results are tabulated periodically & sent to the sponsering group for data analysis § The reports often includes extensive data analysis, statesical sumaries & plots

§ The mean of values of all laboratories is taken as the true or correct value & is used for comparision with the indivisual laboratory’s mean § Different approaches for data anaalysis e. g. t-test, SDI, Youden plots & Levey-Jennings plots

New quality initiatives The six sigma process Lean production ISO 9000

The six sigma process § The six sigma control is an evolution in quality management § 6 sigma or 6 SD of process variation should fit within the tolerance limits for the process
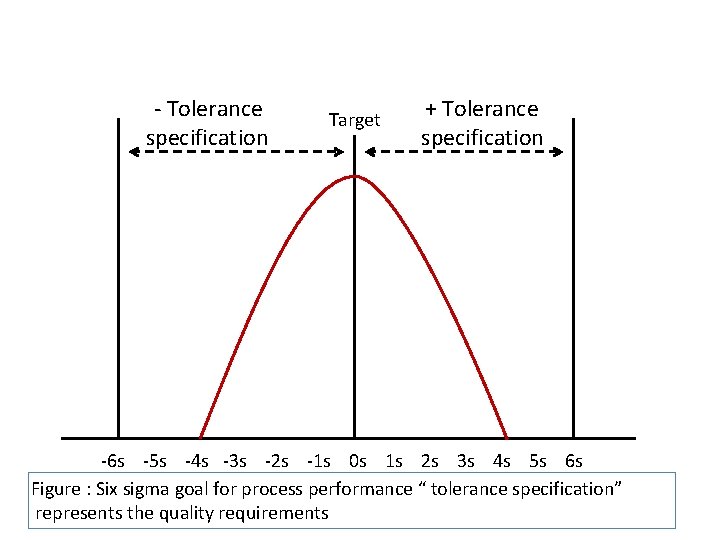
- Tolerance specification Target + Tolerance specification -6 s -5 s -4 s -3 s -2 s -1 s 0 s 1 s 2 s 3 s 4 s 5 s 6 s Figure : Six sigma goal for process performance “ tolerance specification” represents the quality requirements

Lean production § It is a quality process that is focused on creating more value by eliminating activities that are considered waste § e. g. Lean team at Saint Mary’s Hospital used lean production to improve the efficiency of its paper ordering system for lab work in their ICU.
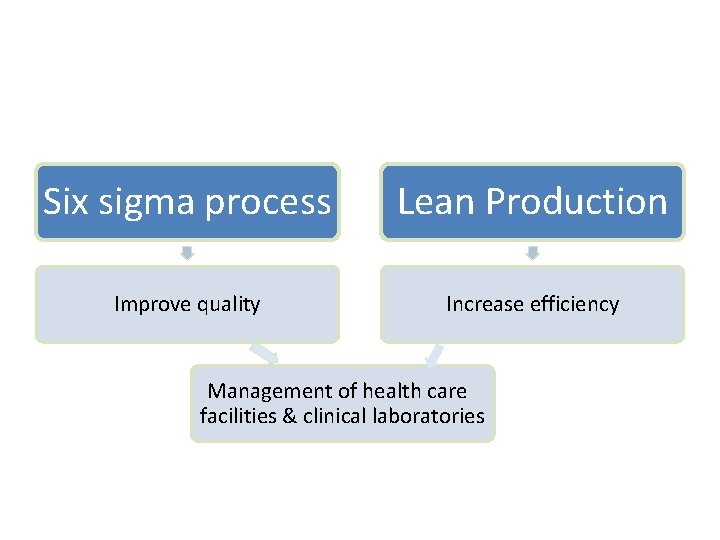
Six sigma process Lean Production Improve quality Increase efficiency Management of health care facilities & clinical laboratories

ISO 9000 § The International Standard Organization (ISO) has developed the ISO 9000 standards § It is a set of 4 standards (ISO 9001 -9004) enacted to ensure quality management & QA. § ISO 9000 represents an international consequence on the essential features of a QS to ensure the effective operation of an organization

Joint Committee for traceability in Laboratory Medicine The traceability of values assigned to calibrators &/or control materials must be assured through available reference measurement procedures &/or available reference materials of a higher order
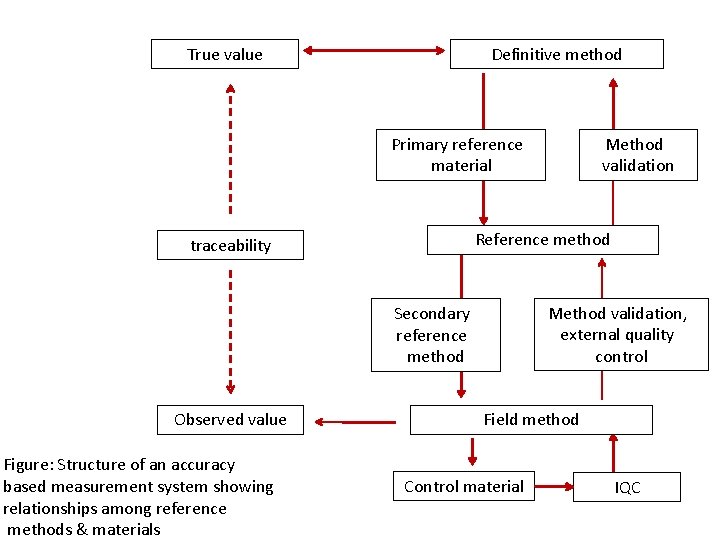
True value Definitive method Primary reference material Reference method traceability Method validation, external quality control Secondary reference method Observed value Figure: Structure of an accuracy based measurement system showing relationships among reference methods & materials Method validation Field method Control material IQC

References Burtis, C. A. , Ashwood, E. R. & Br uns, D. E. Fundamentals of Clinical Chemistry. 2008. . 6 th. ed. SAUNDERS ELSEVIER. P: 249 -262. Arneson, W. & Brichell, J. Clinical Chemistry ‘A Laboratory Perspective’. 2007. F. A. Davis Company. P: 53 -72. Westgard, J. O. Internal Quality Control: Planning & Implementation Strategies. 2003. Ann Clin Biochem. 40; 593 -611.

Thank you
- Slides: 42