Contractility and Collagen Perturbations Rapidly Impact LMNA and
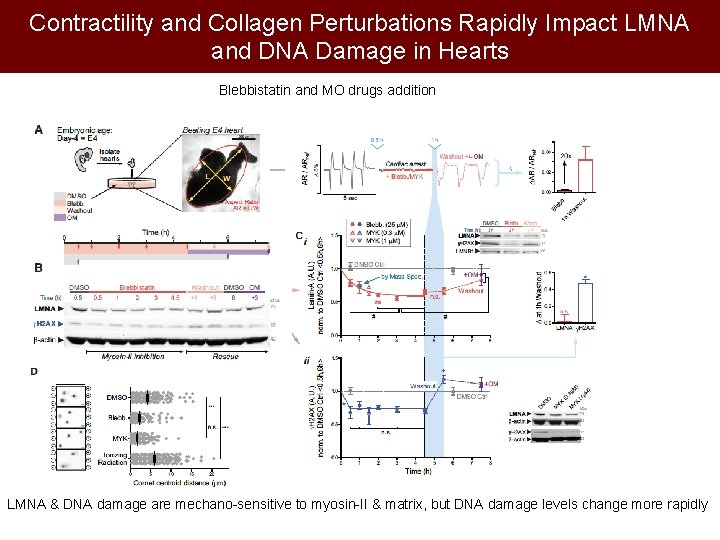
Contractility and Collagen Perturbations Rapidly Impact LMNA and DNA Damage in Hearts Blebbistatin and MO drugs addition LMNA & DNA damage are mechano-sensitive to myosin-II & matrix, but DNA damage levels change more rapidly
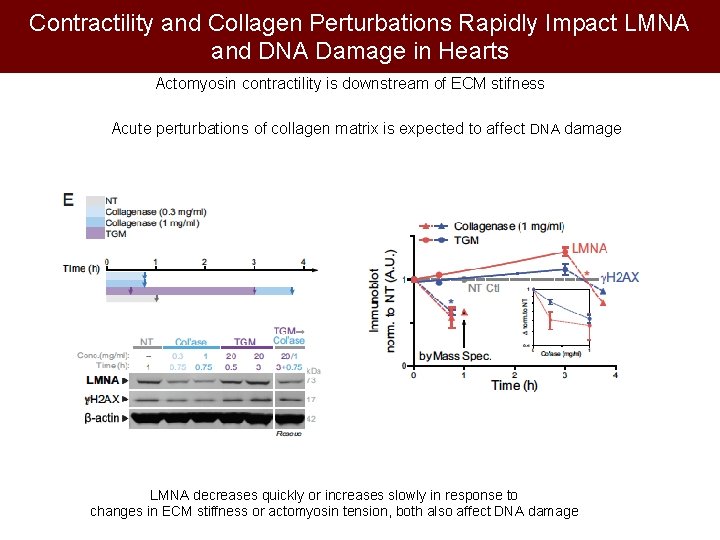
Contractility and Collagen Perturbations Rapidly Impact LMNA and DNA Damage in Hearts Actomyosin contractility is downstream of ECM stifness Acute perturbations of collagen matrix is expected to affect DNA damage LMNA decreases quickly or increases slowly in response to changes in ECM stiffness or actomyosin tension, both also affect DNA damage
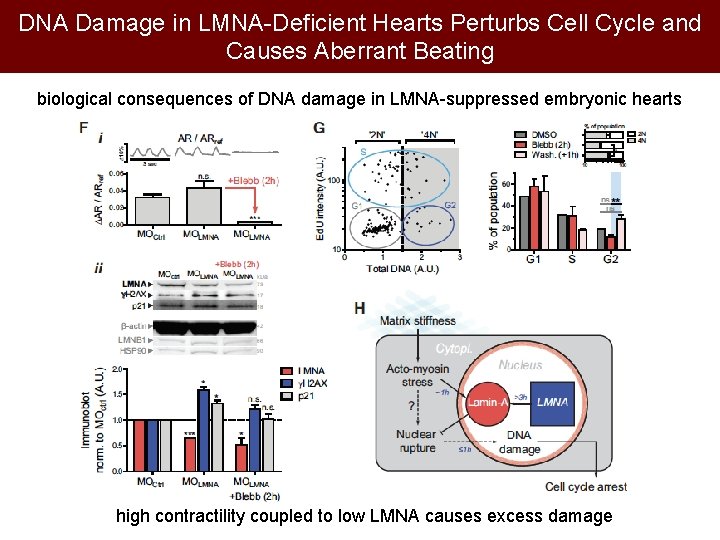
DNA Damage in LMNA-Deficient Hearts Perturbs Cell Cycle and Causes Aberrant Beating biological consequences of DNA damage in LMNA-suppressed embryonic hearts high contractility coupled to low LMNA causes excess damage
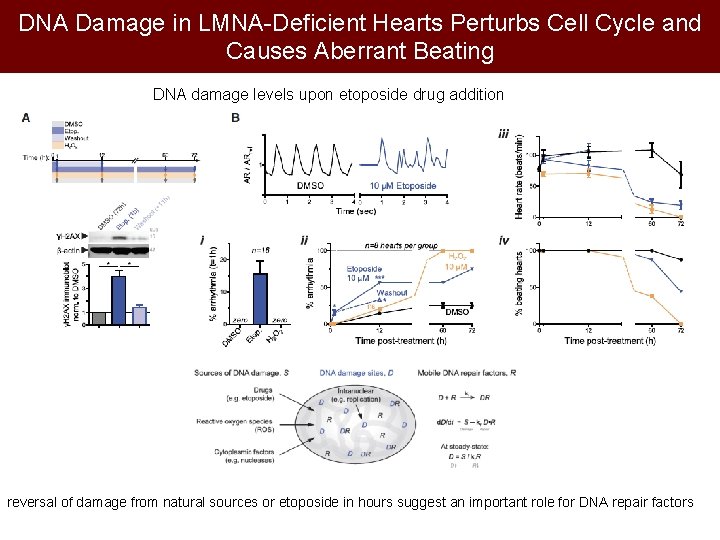
DNA Damage in LMNA-Deficient Hearts Perturbs Cell Cycle and Causes Aberrant Beating DNA damage levels upon etoposide drug addition reversal of damage from natural sources or etoposide in hours suggest an important role for DNA repair factors
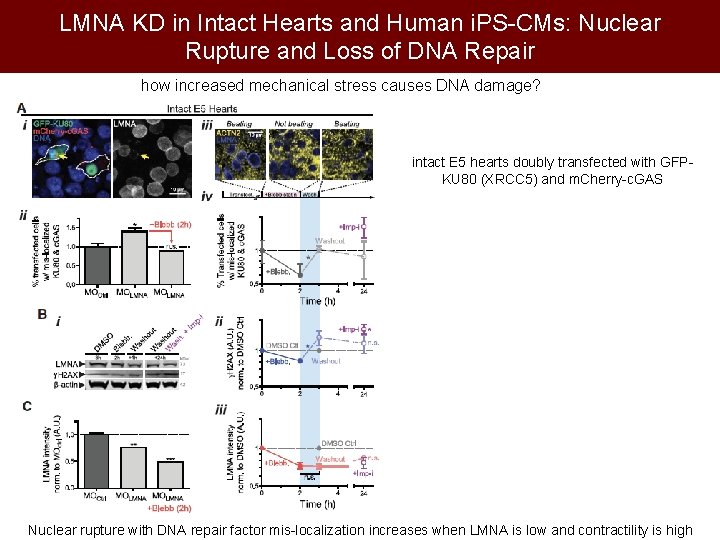
LMNA KD in Intact Hearts and Human i. PS-CMs: Nuclear Rupture and Loss of DNA Repair how increased mechanical stress causes DNA damage? intact E 5 hearts doubly transfected with GFPKU 80 (XRCC 5) and m. Cherry-c. GAS Nuclear rupture with DNA repair factor mis-localization increases when LMNA is low and contractility is high
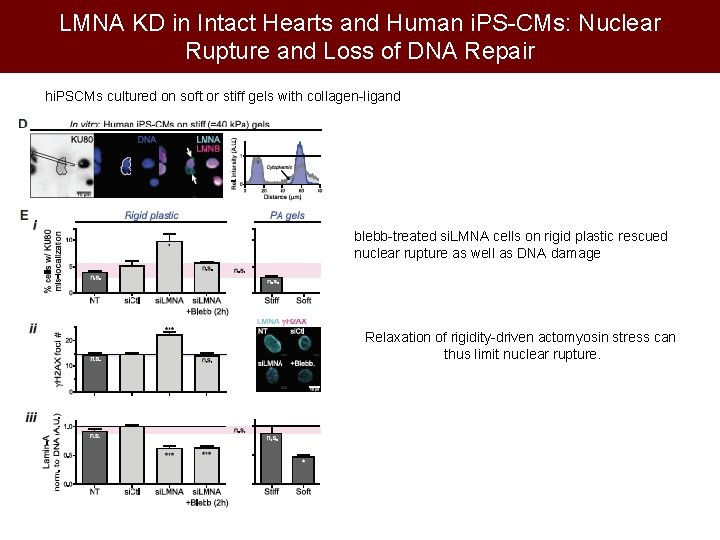
LMNA KD in Intact Hearts and Human i. PS-CMs: Nuclear Rupture and Loss of DNA Repair hi. PSCMs cultured on soft or stiff gels with collagen-ligand blebb-treated si. LMNA cells on rigid plastic rescued nuclear rupture as well as DNA damage Relaxation of rigidity-driven actomyosin stress can thus limit nuclear rupture.
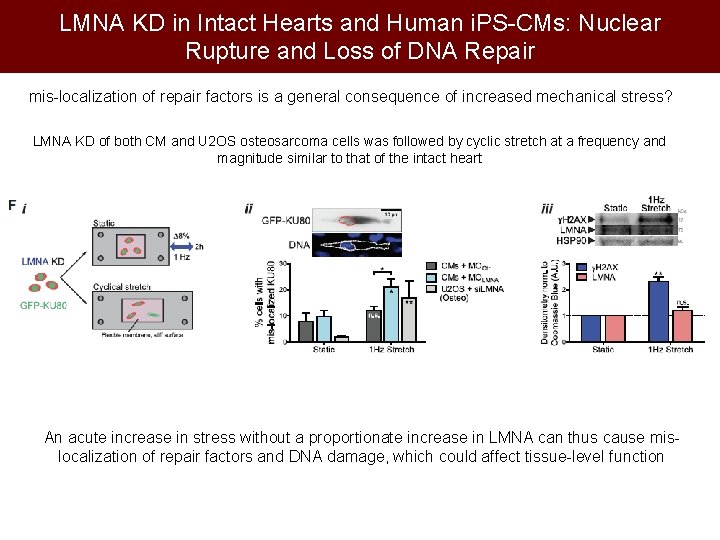
LMNA KD in Intact Hearts and Human i. PS-CMs: Nuclear Rupture and Loss of DNA Repair mis-localization of repair factors is a general consequence of increased mechanical stress? LMNA KD of both CM and U 2 OS osteosarcoma cells was followed by cyclic stretch at a frequency and magnitude similar to that of the intact heart An acute increase in stress without a proportionate increase in LMNA can thus cause mislocalization of repair factors and DNA damage, which could affect tissue-level function
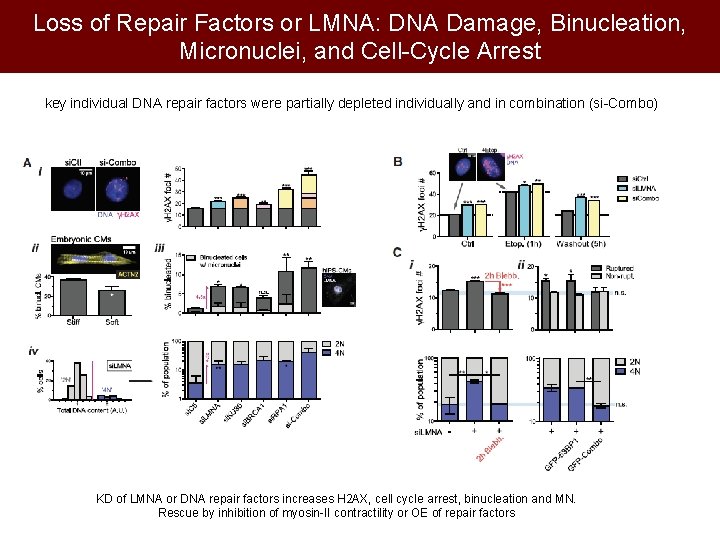
Loss of Repair Factors or LMNA: DNA Damage, Binucleation, Micronuclei, and Cell-Cycle Arrest key individual DNA repair factors were partially depleted individually and in combination (si-Combo) KD of LMNA or DNA repair factors increases H 2 AX, cell cycle arrest, binucleation and MN. Rescue by inhibition of myosin-II contractility or OE of repair factors
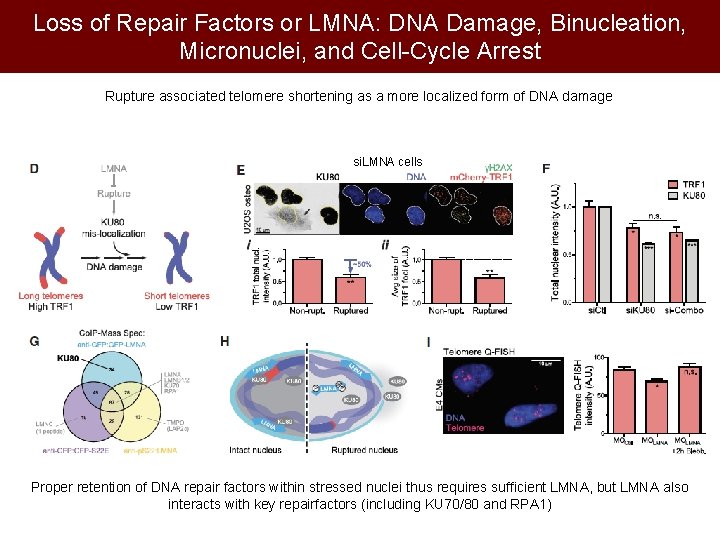
Loss of Repair Factors or LMNA: DNA Damage, Binucleation, Micronuclei, and Cell-Cycle Arrest Rupture associated telomere shortening as a more localized form of DNA damage si. LMNA cells Proper retention of DNA repair factors within stressed nuclei thus requires sufficient LMNA, but LMNA also interacts with key repairfactors (including KU 70/80 and RPA 1)
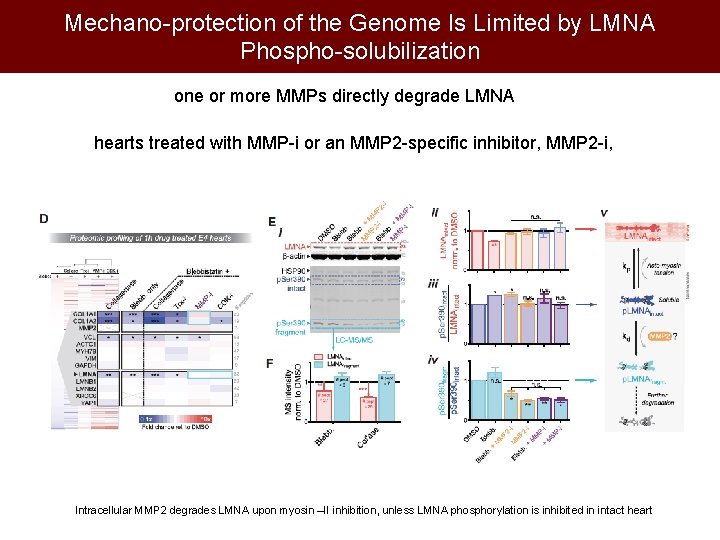
Mechano-protection of the Genome Is Limited by LMNA Phospho-solubilization one or more MMPs directly degrade LMNA hearts treated with MMP-i or an MMP 2 -specific inhibitor, MMP 2 -i, Intracellular MMP 2 degrades LMNA upon myosin –II inhibition, unless LMNA phosphorylation is inhibited in intact heart
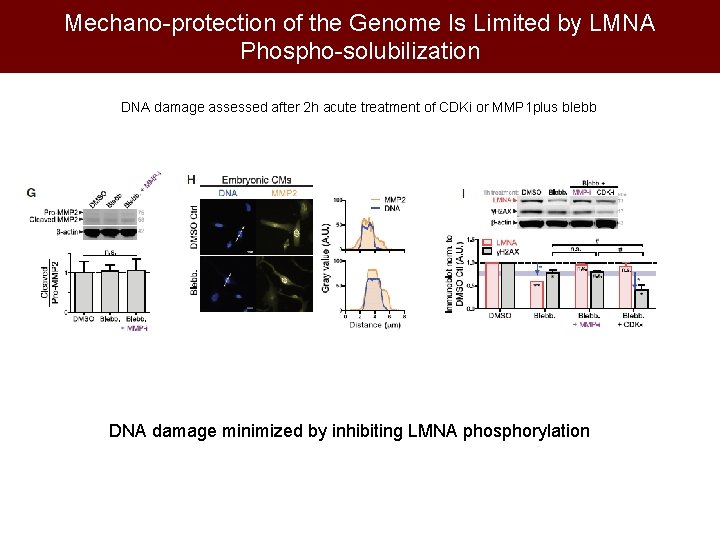
Mechano-protection of the Genome Is Limited by LMNA Phospho-solubilization DNA damage assessed after 2 h acute treatment of CDKi or MMP 1 plus blebb DNA damage minimized by inhibiting LMNA phosphorylation

conclusions
- Slides: 13