Contraception in obese women Prof Johannes Bitzer Former

Contraception in obese women Prof Johannes Bitzer Former President of the European Society of Contraception and Reproductive Health Editor in Chief of the European Journal of Contraception and Reproductive Health Care Member of the Executive Board of EBCOG

Definition of obesity (WHO) BMI Overweight 25 -29. 9 kg / m 2 Obese 30 -39. 9 kg / m 2 Very obese 30 -34. 9 kg / m 2 Class 1 obesity 35 -39. 9 kg / m 2 Class 2 obesity ≥ 40 kg / m 2 Class 3 obesity G. Merki

Obesity – an Increasing Problem WHO Database (accessed April 2014)
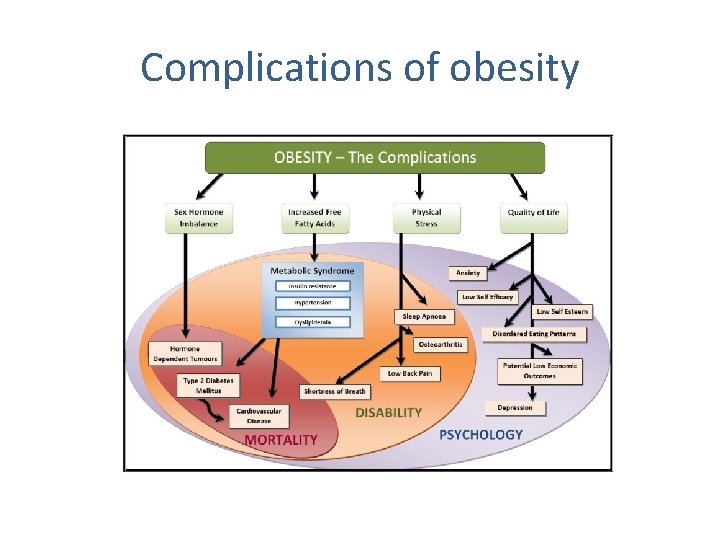
Complications of obesity

Obesity and Contraceptive Choices Concerns: • lowered efficacy of the method • increased risks associated with certain methods • further weight gain resulted from use of the method

Obesity and Contraceptive Efficacy • There are generally limited data on the effect of body weight on contraceptive effectiveness, as most studies exclude or involve only small numbers of obese subjects. • Earlier studies mainly studied on body weight • Effect of BMI studied only in more recent studies (Lopez et al, Cochrane Database Syst Rev 2013)

Obesity and Contraceptive Efficacy • Although some reports suggested increased risk of failure of COC in obese women, this has not been consistently observed in all studies. • Evidence was very contraceptive method. limited for any individual (Dinger et al, Obstet Gynecol 2011; Edelman and Jensen, Sem Reprod Med 2012; Lopez et al, Cochrane Database Syst Rev 2013)

Prolonged monitoring of ethinyl estradiol and levonorgestrel levels confirms an altered pharmacokinetic profile in obese oral contraceptives users N= 32 BMI > 30 kg/m 2 (39 kg/m 2) Pill formulation: 20 µg EE / 100 µg LNG 2 cycles Alison B. Edelman , Ganesh Cherala , Myrna Y. Munar , Barent Du. Bois , Martha Mc. Innis , Frank Z. Stanczyk , Jeffrey T. Jensen Contraception Volume 87, Issue 2 2013 220 - 226

Efficiency of hormonal contraceptive in obese women Comparison to a historical control group of normal weight women Altered pharmacokinetic parameters in obese women • • > AUC > Half-life < Clearance Later steady-state Edelmann et al. : Contraception Volume 87, Issue 2 2013 220 - 226
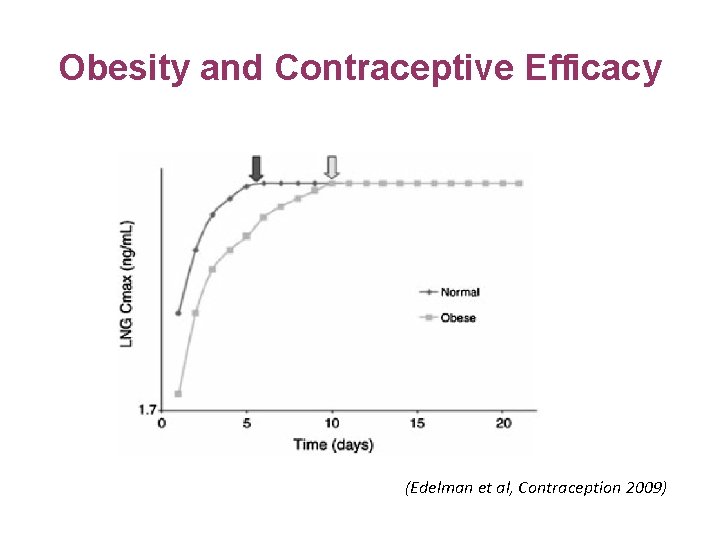
Obesity and Contraceptive Efficacy (Edelman et al, Contraception 2009)
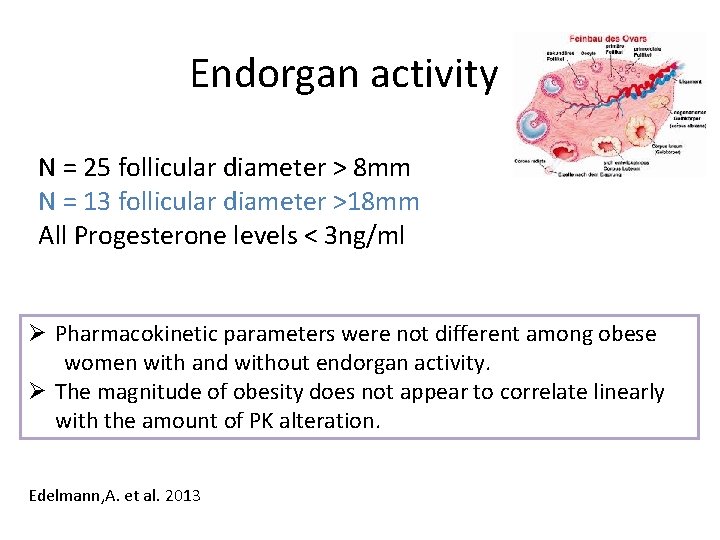
Endorgan activity N = 25 follicular diameter > 8 mm N = 13 follicular diameter >18 mm All Progesterone levels < 3 ng/ml Ø Pharmacokinetic parameters were not different among obese women with and without endorgan activity. Ø The magnitude of obesity does not appear to correlate linearly with the amount of PK alteration. Edelmann, A. et al. 2013

Conclusion • Obesity alters PK parameters of COC even more than originally documented (CL, AUC, t 1/2) • Majority of obese women had poor endorgan suppression, however PK parameters and endorgan suppression do not correlate • A longer t ½ translates into a longer time to achieve steady state (difficult after 7 - day pill free interval and when initiating CHC) Open Questions form these very qualified data • • • Should obese women use 10 days additional protection when starting COC ? Would a long-cycle be more efficient ? Would a dosage of 30 mcg. EE/150 LNG be of advantage Edelman, A. et al. Contraception 2013

Obesity and Contraceptive Efficacy • Efficacy of combined contraceptive patch may be lowered in women weighing 90 kg or above.
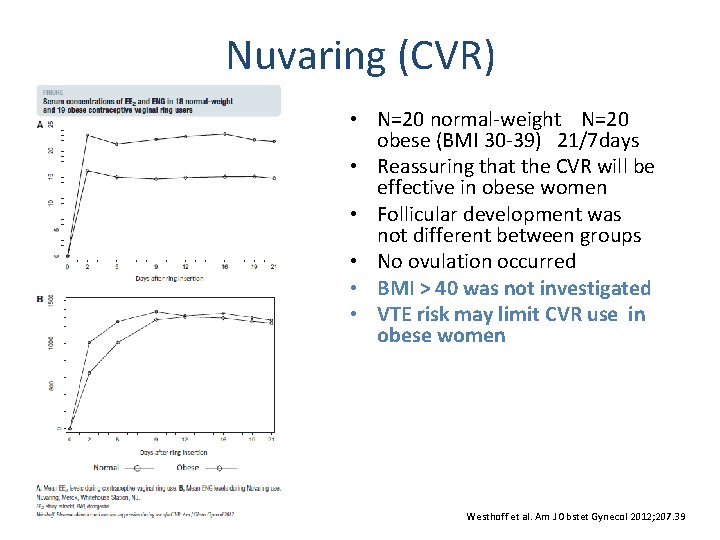
Nuvaring (CVR) • N=20 normal-weight N=20 obese (BMI 30 -39) 21/7 days • Reassuring that the CVR will be effective in obese women • Follicular development was not different between groups • No ovulation occurred • BMI > 40 was not investigated • VTE risk may limit CVR use in obese women Westhoff et al. Am J Obstet Gynecol 2012; 207. 39

Progestin-only methods Efficiacy and safety in obese women
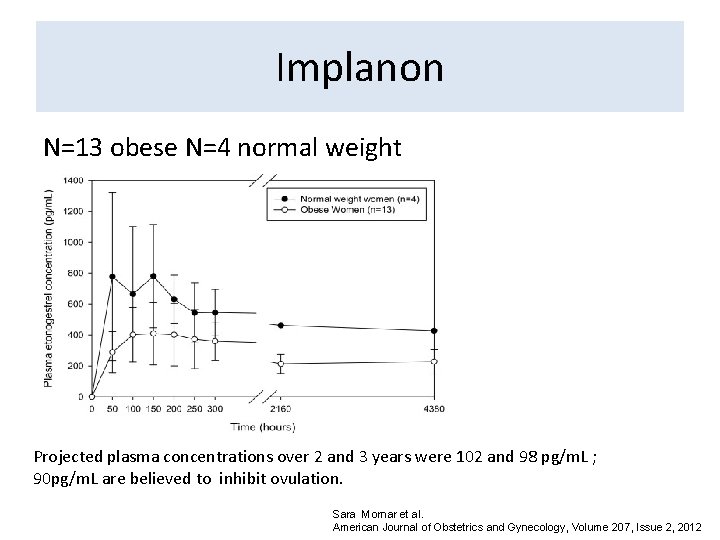
Implanon N=13 obese N=4 normal weight Projected plasma concentrations over 2 and 3 years were 102 and 98 pg/m. L ; 90 pg/m. L are believed to inhibit ovulation. Sara Mornar et al. American Journal of Obstetrics and Gynecology, Volume 207, Issue 2, 2012

Implanon • N= 1168 Implanon users→ 35 % obese • N= 4200 IUD users → 35 % obese Pregnancies in all women Implanon 1 1 IUD 12 4 obese women Efficiency of Implanon may be reduced with obesity. Earlier removal is recommended in women > 80 kg Xu, H. Obstetrics and Gynecology July 2012

Implanon Conclusion ESC Recommendation • The implant is a safe option for obese women • However with regard to effectiveness: Consider early removal after 24 months (? ) in obese women

DMPA subcutaneously DMPA listed as class 1 for obese adult women from WHO. Study • 12 weekly 104 mg DMPA sc for 6 months 12 week MPA level Group 1 BMI > 40 kg/m 2 n=5 498 pg/ml** Group 2 BMI = 30 -39 kg/m 2 n=5 672 pg/ml Group 3 BMI < 25 kg/m 2 n=5 707 pg/ml* * Contraceptive threshold 200 pg/ml; ** 1 woman one level 182 pg/ml P. Segall-Gutierrrez et al, Contraception 81, 2010

DMPA subcutaneously Conclusion In very obese women a shortening of the interval between injection has to be considered P. Segall-Gutierrrez et al, Contraception 81, 2010

DMPA ESC Conclusion • DMPA im and sc are effective methods in obese women • There may be a negative effect on insulin resistance , without clear evidence about the clinical relevance • In comparison to CHC benefits are higher for obese women • Consider shortening of the interval between injections in very obese women

POP Desogestrel 75 mcg • No increased risk for VTE or ATE • Minimal impact on lipids and glucose metabolism • At present no data indicate a decreased efficiacy in obese women • But no data means: be careful !! Might not be efficient in obese women.

Obesity and Contraceptive Efficacy • Reduced efficacy of levonorgestrel and ulipristal emergency contraceptive pills in obese women. • Failure rate increased by more than 3 x in obese women (p<0. 0001), more so for LNG than UPA (Glasier et al, Contraception 2011)

Safety of contraceptive methods in obese women

Obesity and Contraceptive Safety • All methods of contraception are not considered contraindicated in obese women according to the World Health Organisation Medical Eligibility Criteria, although the UK Medical Eligibility Criteria (UKMEC) classifies BMI >=35 kg/m 2 as category 3 for using combined hormonal contraception (CHC).

Obesity and Contraceptive Safety • The main risk of concern for using CHC is venous thromboembolism (VTE), particularly in the first year after initiation. • The risk of VTE is relatively higher with newer generation pills although the absolute difference is probably small. • Data are awaited to indicate whether the recently introduced combined oral contraceptive containing natural oestradiol is associated with lower thromboembolic risk.

Obesity and Contraceptive Safety • The risk of transdermal CHC is not lower compared to combined oral contraceptives. Raymond et al, Obstet Gynecol 2012

Obesity and Contraceptive Safety • The risk of transdermal CHC is not lower compared to combined oral contraceptives. • Historical national registry based cohort study in Denmark: • Users of transdermal patches had adjusted relative risk of 2. 3 (1. 0 to 5. 2) of VTE vs users of combined oral contraceptives containing levonorgestrel (Lidegaard et al, BMJ 2012) • May be related to the higher area under the curve of estrogen exposure in patch users vs COC users

Obesity and Contraceptive Safety • CHC should not be prescribed to obese women with additional cardiovascular risk factors such as hypertension, diabetes, migraine, smoking or inherited thrombophilia. • The effect of CHC on glucose and lipid metabolism in obese women without other medical complications has not been adequately studied. • Progestogen-only contraception is generally considered safe in obese women.
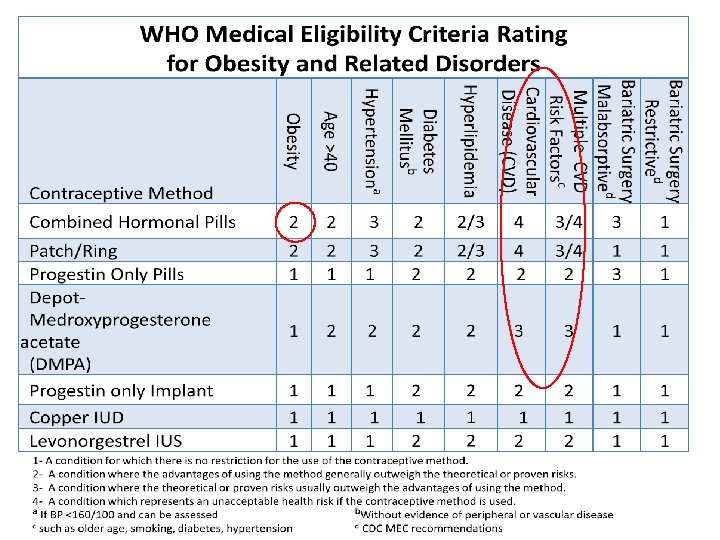

Obesity and Contraceptive Safety – Nonhormonal methods • There is generally no efficacy or safety concern for use of intrauterine contraception and barrier methods in obese women • May be more technical difficulties in insertion of intrauterine devices or fitting of diaphragm. • Laparoscopic sterilisation may be associated with higher anaesthetic and surgical risks and difficulties in obese women • Hysteroscopic sterilisation may provide an alternative if available.

Contraceptive and Weight Gain • Four trials with a placebo or no intervention group: no evidence of causal association between combination contraceptives and weight change. • Also, most comparisons of different CHCs showed no substantial difference in weight or difference in discontinuation rates due to weight change. • Available evidence is insufficient to determine the effect of CHC on weight, but no large effect is evident (Gallo et al, Cochrane Database Syst Rev 2014)

Contraceptive and Weight Gain • Limited evidence of weight gain when using progestinonly contraceptives. • Some differences in weight change with POC compared to no hormonal contraceptive. • Actual mean weight gain was low for 6 to 12 months, i. e. , less than 2 kg for most studies. • More weight gain was noted at two and three years, but was generally similar for the study groups. People may gain weight over time regardless of contraceptive use. (Lopez et al, Cochrane Database Syst Rev 2013)

Contraceptive and Weight Gain Summary: • There is no robust evidence to support a causal relationship between hormonal contraception and weight gain although this is a common belief.

Copper–IUD and LNG-releasing device ESC recommendation • Metabolic effects are none or minimal and efficiency is not reduced with obesity • Both methods are highly recommended after contraindications have been taken into consideration

Concluding remarks • Generally, risks of pregnancy >> risks of using hormonal contraception in obese women, although obesity is a significant risk factor for VTE and cardiovascular diseases • Women with additional risk factors may consider avoiding combined hormonal contraceptives • Non-oestrogen-containing contraceptive generally safer & acceptable options methods are • No adequate scientific evidence that hormonal contraceptives aggravates weight gain
- Slides: 36